Anthrax
Medical Information
Anthrax is rare in the United States. However, clinicians should be vigilant for cases in travelers from countries with higher prevalence rates and in workers who handle animal products from areas where anthrax is endemic (i.e., found regularly). Employers and clinicians should also keep in mind the possibility of intentional anthrax releases or other deliberate exposures (e.g., bioterrorism).
This page discusses three forms of anthrax that result from three routes through which someone can be exposed to Bacillus anthracis (BA) or its spores:
- cutaneous, or skin, infection from contact exposure;
- infection associated with inhalation exposure (i.e., breathing in the bacteria or spores); and
- gastrointestinal infection associated with ingestion of contaminated materials, such as raw or undercooked meat from an animal infected with anthrax.
Two other types of anthrax, oropharyngeal (mouth and throat) anthrax and meningeal (membranes covering the brain and spinal cord) anthrax, are secondary complications of the three main types of anthrax listed above. Less common than the gastrointestinal form, oropharyngeal anthrax is also associated with the ingestion of contaminated meat, but it tends to be milder than the classic gastrointestinal disease with a better prognosis.1, 2 Meningeal anthrax is a rare complication of anthrax (typically cutaneous anthrax) in which the bacteria spread to the central nervous system. Despite intensive antibiotic therapy, meningeal anthrax is almost always fatal.
Injection anthrax, has been identified among heroin-injecting drug users in northern Europe. However, this type of anthrax has not been reported in the U.S. Injection anthrax is not generally associated with work tasks that lead to this type of occupational exposure.
Exposure and Infection
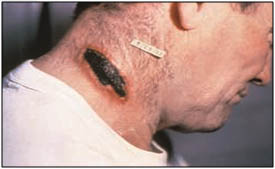
Photo Credit: CDC
Infection with BA can cause cutaneous anthrax. Lesions, like on this patient’s neck, are marked by a typical black eschar.
- Most anthrax infections are cutaneous and result from BA spores entering a cut or abrasion on the skin, such as when handling contaminated wool, animal hides, leather, or hair products of infected animals. In some instances, such as when working with active bacteria in laboratories, contact with the active bacteria may cause infection.
- Inhalation anthrax can occur when a person inhales airborne spores generated during the processing of animal hides, wool, or hair contaminated with the spores. In laboratories, processes that aerosolize BA or its spores or activities that are not conducted with appropriate controls (e.g., in biosafety cabinets) can lead to exposure. There is also risk of exposure occurring from a bioterrorist/criminal event involving the release of BA spores.
- Gastrointestinal anthrax can occur after consuming contaminated meat and other food.
The amount of BA it takes to infect someone and cause anthrax infection varies depending upon the species (e.g., human, type of animal) exposed and the route of exposure.
- The median infectious dose (ID50), the average amount of BA it takes to infect someone and cause anthrax, is estimated at 8,000 to 50,0000 spores via the inhalation route of exposure, but is unknown for cutaneous and ingestion exposure.
- The estimated median lethal dose (LD50), the average amount of BA it takes to kill a person who is exposed, ranges from 10 or fewer spores for cutaneous anthrax to 2,500-55,000 spores for inhalation anthrax.3
- Estimates of ID50 and LD50 for cutaneous and gastrointestinal anthrax are not well understood, though they are likely higher than for inhalation anthrax.4
Once in the body, BA spores can germinate (the bacteria transition out of the spore form and begin to multiply) and cause an infection. However, there are many factors that determine the severity of the infection, including the number of spores germinating and releasing active bacteria (i.e., bacteria that can grow and multiply) and toxins into the body. A healthy person's immune system is typically able to fight off small amounts of the bacteria, particularly when the route of exposure is cutaneous. In these cases, a person exposed to small amounts of BA is unlikely to develop symptoms of infection. In some cases, BA spores remain dormant and do not germinate to cause an infection.
When BA spores are used as a biological weapon by terrorists or other criminals, BA must be grown, concentrated, dried, and milled into a powder of fine particles that can be released into the air. People who are exposed can inhale the particles into their lungs. Particle size and other characteristics of the BA spores can affect how they are deposited within a person's respiratory system and their potential for causing infection.
Symptoms
Other Names for Anthrax
- Woolsorter's disease
- Ragpicker's disease
- Cumberland disease
- Maladi Charbon
- Malignant pustule
- Malignant carbuncle
- Milbrand
- Splenic fever
Symptoms of anthrax vary depending on the route of exposure and site or type of infection, but usually occur within seven days. However, symptoms can take anywhere from one day to more than two months to appear.5
- Cutaneous anthrax begins as an itchy bump or a group of small blisters. A painless ulcer (a skin sore) then appears with a black center. Skin sores may also have a black appearance when they form dry scabs called "eschars." In the majority of cases, the sore(s) appears on the face, neck, arms, or hands. Less than one percent of appropriately treated cutaneous anthrax cases result in death.6
- Initially, inhalation anthrax may resemble a common cold with fever, chills, headache, body ache, and cough. Later, there may be chest discomfort, shortness of breath, and confusion or dizziness. Nausea, vomiting, or stomach pains; drenching sweats; and extreme tiredness may also be present. Case-fatality rates for inhalation anthrax have varied from 85 percent in historical cases to 45 percent among victims of the 2001 bioterrorist attack despite appropriate antibiotics and supportive care.7
- Symptoms of gastrointestinal anthrax may include fever and chills, swelling of the neck and glands in the neck, sore throat, painful swallowing, hoarseness, nausea and vomiting (with or without blood), diarrhea, headache, red eyes and face, stomach ache, fainting, and swelling of the stomach. Gastrointestinal anthrax infection can spread to the bloodstream, and may result in death. While the case-fatality rate of gastrointestinal anthrax is unknown, the CDC estimates that between 25 and 60 percent of people with this form of the disease will die.
Diagnosis
Anthrax can be diagnosed by isolating BA from the blood, skin lesions, or respiratory secretions, or by measuring specific antibodies in the blood of people who are suspected cases.
The CDC Anthrax Diagnosis page provides information on diagnostic procedures for anthrax.
The CDC Laboratory Testing for Anthrax: Frequently Asked Questions page provides information on specimen collection and shipping, and a list of laboratories approved for BA testing.
Medical Management and Countermeasures
Antibiotic and antitoxin therapy are available for treating patients with anthrax in or out of a hospital setting. Antibiotic treatment targets all types of BA infection. Antitoxin treatment helps eliminate the toxins BA release into an infected person's body, rather than targeting the bacteria causing the disease. Antitoxin therapy is useful alone and in combination with other treatment options.
Hospitalization for anthrax depends on the severity of the case and the need for supportive treatment, such as continuous fluid drainage and mechanical ventilation.
For prevention of anthrax after a known exposure, a course of oral antibiotics, such as ciprofloxacin or doxycycline, is typically prescribed for up to 60 days. Patient medical history, other patient characteristics, and route of exposure influence the antibiotic treatment selected. In general, the sooner treatment is started, the better the prognosis (outcome).
A licensed anthrax vaccine is available for use in humans with a 92.5 percent efficacy (effectiveness) for protection against cutaneous and inhalation anthrax.8 However, it is generally not available for the general public and is only approved by the Food and Drug Administration for high-risk adults before exposure.
The CDC's Advisory Committee on Immunization Practices (ACIP) recommends vaccination against BA for the following populations at risk for occupational exposure:
- Persons handling animals or animal products where standards are insufficient to prevent exposure to BA spores;
- Veterinarians and other persons considered to be at high risk for BA exposure if they handle potentially infected animals in research settings or in areas with a high incidence of enzootic (disease affecting animals) anthrax cases;
- Laboratory workers at risk for repeated exposure to BA spores;
- Military personnel determined by the Department of Defense (DoD) to have a serious risk for exposure to aerosolized BA spores; and
- Environmental investigators and remediation workers whose occupation may require repeated entry into areas contaminated with BA spores.
ACIP advises against vaccination for workers involved in routine processing of clinical specimens or environmental swabs in general diagnostic laboratories.9 The Committee also does not recommend pre-vaccination of workers in postal processing facilities (due to the rapid detection systems allowing post-exposure therapy to begin immediately) or for emergency responders and other response and recovery workers.
Workers outside of those whom ACIP recommends get the anthrax vaccine prior to exposure may be offered the vaccine after exposure, such as following a biological agent attack involving BA.
In February 2014, the CDC convened panels of anthrax experts to review and update guidelines for anthrax post-exposure prophylaxis and treatment. Significant changes include more details on critical care and clinical procedures and other new antimicrobial alternatives, including favored antimicrobial drug treatment for suspected anthrax meningitis.
The CDC Vaccine Information Statement provides more information on vaccination against anthrax.
The CDC Anthrax Web pages provide more information on medical care and laboratory testing for anthrax.
1 Navacharoen, N., Sirisanthana, T., Navacharoen, W., & Ruckphaopunt, K. (1985). Oropharyngeal Anthrax. The Journal of Laryngology & Otology, 99(12), 1293-1295.
2 Dixon, T.C., Meselson, M., Guillemin, J., and Hanna, P.C., "Anthrax," N Engl J Med, 341:815-826 (1999).
3 Biosafety in Microbiological and Biomedical Laboratories, 5th Edition. National Institute of Health (NIH), Centers for Disease Control and Prevention (CDC), U.S. Department of Health and Human Services (HHS) (2009), 123.
4 Anthrax. Center for Infectious Disease Research and Policy (CIDRAP), University of Minnesota.
5 "Symptoms," Centers for Disease Control and Prevention (CDC), U.S. Department of Health and Human Services (HHS).
6 "Anthrax (Bacillus anthracis), 2010 Case Definition," Centers for Disease Control and Prevention (CDC), U.S. Department of Health and Human Services (HHS).
7 "Symptoms," Centers for Disease Control and Prevention (CDC), U.S. Department of Health and Human Services (HHS).
8 Anthrax. Vaccines, Blood and Biologics, Food and Drug Administration (FDA), U.S. Department of Health and Human Services (HHS).
9 Biosafety in Microbiological and Biomedical Laboratories (BMBL), 5th Edition. Centers for Disease Control and Prevention (CDC), U.S. Department of Health and Human Services (HHS) (2009).

