[Federal Register: February 28, 2006 (Volume 71, Number 39)][Rules and Regulations] [Page 10099-10385]
From the Federal Register Online via GPO Access [wais.access.gpo.gov]
[DOCID:fr28fe06-25]
-----------------------------------------------------------------------
Part II
Department of Labor
-----------------------------------------------------------------------
Occupational Safety and Health Administration
-----------------------------------------------------------------------
29 CFR Parts 1910, 1915, et al.
Occupational Exposure to Hexavalent Chromium; Final Rule
-----------------------------------------------------------------------
DEPARTMENT OF LABOR
Occupational Safety and Health Administration
29 CFR Parts 1910, 1915, 1917, 1918, and 1926
[Docket No. H054A]
RIN 1218-AB45
Occupational Exposure to Hexavalent Chromium
AGENCY: Occupational Safety and Health Administration (OSHA),
Department of Labor.
ACTION: Final rule.
-----------------------------------------------------------------------
SUMMARY: The Occupational Safety and Health Administration (OSHA) is
amending the existing standard which limits occupational exposure to
hexavalent chromium (Cr(VI)). OSHA has determined based upon the best
evidence currently available that at the current permissible exposure
limit (PEL) for Cr(VI), workers face a significant risk to material
impairment of their health. The evidence in the record for this
rulemaking indicates that workers exposed to Cr(VI) are at an increased
risk of developing lung cancer. The record also indicates that
occupational exposure to Cr(VI) may result in asthma, and damage to the
nasal epithelia and skin.
The final rule establishes an 8-hour time-weighted average (TWA)
exposure limit of 5 micrograms of Cr(VI) per cubic meter of air (5
[mu]g/m\3\). This is a considerable reduction from the previous PEL of
1 milligram per 10 cubic meters of air (1 mg/10 m\3\, or 100 [mu]g/
m\3\) reported as CrO3, which is equivalent to a limit of 52
[mu]g/m\3\ as Cr(VI). The final rule also contains ancillary provisions
for worker protection such as requirements for exposure determination,
preferred exposure control methods, including a compliance alternative
for a small sector for which the new PEL is infeasible, respiratory
protection, protective clothing and equipment, hygiene areas and
practices, medical surveillance, recordkeeping, and start-up dates that
include four years for the implementation of engineering controls to
meet the PEL.
The final standard separately regulates general industry,
construction, and shipyards in order to tailor requirements to the
unique circumstances found in each of these sectors.
The PEL established by this rule reduces the significant risk posed
to workers by occupational exposure to Cr(VI) to the maximum extent
that is technologically and economically feasible.
DATES: This final rule becomes effective on May 30, 2006. Start-up
dates for specific provisions are set in Sec. 1910.1026(n) for general
industry; Sec. 1915.1026(l) for shipyards; and Sec. 1926.1126(l) for
construction. However, affected parties do not have to comply with the
information collection requirements in the final rule until the
Department of Labor publishes in the Federal Register the control
numbers assigned by the Office of Management and Budget (OMB).
Publication of the control numbers notifies the public that OMB has
approved these information collection requirements under the Paperwork
Reduction Act of 1995.
ADDRESSES: In compliance with 28 U.S.C. 2112(a), the Agency designates
the Associate Solicitor for Occupational Safety and Health, Office of
the Solicitor, Room S-4004, U.S. Department of Labor, 200 Constitution
Avenue, NW., Washington, DC 20210, as the recipient of petitions for
review of these standards.
FOR FURTHER INFORMATION CONTACT: Mr. Kevin Ropp, Director, OSHA Office
of Communications, Room N-3647, U.S. Department of Labor, 200
Constitution Avenue, NW., Washington, DC 20210; telephone (202) 693-
1999.
SUPPLEMENTARY INFORMATION: The following table of contents lays out the
structure of the preamble to the final standards. This preamble
contains a detailed description of OSHA's legal obligations, the
analyses and rationale supporting the Agency's determination, including
a summary of and response to comments and data submitted during the
rulemaking.
I. General
II. Pertinent Legal Authority
III. Events Leading to the Final Standard
IV. Chemical Properties and Industrial Uses
V. Health Effects
A. Absorption, Distribution, Metabolic Reduction and Elimination
1. Deposition and Clearance of Inhaled Cr(VI) From the
Respiratory Tract
2. Absorption of Inhaled Cr(VI) Into the Bloodstream
3. Dermal Absorption of Cr(VI)
4. Absorption of Cr(VI) by the Oral Route
5. Distribution of Cr(VI) in the Body
6. Metabolic Reduction of Cr(VI)
7. Elimination of Cr(VI) From the Body
8. Physiologically-Based Pharmacokinetic Modeling
9. Summary
B. Carcinogenic Effects
1. Evidence From Chromate Production Workers
2. Evidence From Chromate Pigment Production Workers
3. Evidence From Workers in Chromium Plating
4. Evidence From Stainless Steel Welders
5. Evidence From Ferrochromium Workers
6. Evidence From Workers in Other Industry Sectors
7. Evidence From Experimental Animal Studies
8. Mechanistic Considerations
C. Non-Cancer Respiratory Effects
1. Nasal Irritation, Nasal Tissue Ulcerations and Nasal Septum
Perforations
2. Occupational Asthma
3. Bronchitis
4. Summary
D. Dermal Effects
E. Other Health Effects
VI. Quantitative Risk Assessment
A. Introduction
B. Study Selection
1. Gibb Cohort
2. Luippold Cohort
3. Mancuso Cohort
4. Hayes Cohort
5. Gerin Cohort
6. Alexander Cohort
7. Studies Selected for the Quantitative Risk Assessment
C. Quantitative Risk Assessments Based on the Gibb Cohort
1. Environ Risk Assessments
2. National Institute for Occupational Safety and Health (NIOSH)
Risk Assessment
3. Exponent Risk Assessment
4. Summary of Risk Assessments Based on the Gibb Cohort
D. Quantitative Risk Assessments Based on the Luippold Cohort
E. Quantitative Risk Assessments Based on the Mancuso, Hayes,
Gerin, and Alexander Cohorts
1. Mancuso Cohort
2. Hayes Cohort
3. Gerin Cohort
4. Alexander Cohort
F. Summary of Risk Estimates Based on Gibb, Luippold, and
Additional Cohorts
G. Issues and Uncertainties
1. Uncertainty With Regard to Worker Exposure to Cr(VI)
2. Model Uncertainty, Exposure Threshold, and Dose Rate Effects
3. Influence of Smoking, Race, and the Healthy Worker Survivor
Effect
4. Suitability of Risk Estimates for Cr(VI) Exposures in Other
Industries
H. Conclusions
VII. Significance of Risk
A. Material Impairment of Health
1. Lung Cancer
2. Non-Cancer Impairments
B. Risk Assessment
1. Lung Cancer Risk Based on the Gibb Cohort
2. Lung Cancer Risk Based on the Luippold Cohort
3. Risk of Non-Cancer Impairments
C. Significance of Risk and Risk Reduction
VIII. Summary of the Final Economic Analysis and Regulatory
Flexibility Analysis
IX. OMB Review Under the Paperwork Reduction Act of 1995
X. Federalism
XI. State Plans
XII. Unfunded Mandates
XIII. Protecting Children from Environmental Health and Safety Risks
XIV. Environmental Impacts
XV. Summary and Explanation of the Standards
(a) Scope
(b) Definitions
(c) Permissible Exposure Limit (PEL)
(d) Exposure Determination
(e) Regulated Areas
(f) Methods of Compliance
(g) Respiratory Protection
(h) Protective Work Clothing and Equipment
(i) Hygiene Areas and Practices
(j) Housekeeping
(k) Medical Surveillance
(l) Communication of Chromium (VI) Hazards to Employees
(m) Recordkeeping
(n) Dates
XVI. Authority and Signature
XVII. Final Standards
I. General
This final rule establishes a permissible exposure limit (PEL) of 5
micrograms of Cr(VI) per cubic meter of air (5 [mu]g/m\3\) as an 8-hour
time-weighted average for all Cr(VI) compounds. After consideration of
all comments and evidence submitted during this rulemaking, OSHA has
made a final determination that a PEL of 5 [mu]g/m\3\ is necessary to
reduce the significant health risks posed by occupational exposures to
Cr(VI); it is the lowest level that is technologically and economically
feasible for industries impacted by this rule. A full explanation of
OSHA's rationale for establishing this PEL is presented in the
following preamble sections: V (Health Effects), VI (Quantitative Risk
Assessment), VII (Significance of Risk), VIII (Summary of the Final
Economic Analysis and Regulatory Flexibility Analysis), and XV (Summary
and Explanation of the Standard, paragraph (c), Permissible Exposure
Limit).
OSHA is establishing three separate standards covering occupational
exposures to Cr(VI) for: general industry (29 CFR 1910.1026); shipyards
(29 CFR 1915.1026), and construction (29 CFR 1926.1126). In addition to
the PEL, these three standards include ancillary provisions for
exposure determination, methods of compliance, respiratory protection,
protective work clothing and equipment, hygiene areas and practices,
medical surveillance, communication of Cr(VI) hazards to employees,
recordkeeping, and compliance dates. The general industry standard has
additional provisions for regulated areas and housekeeping. The Summary
and Explanation section of this preamble (Section XV, paragraphs (d)
through (n)) includes a full discussion of the basis for including
these provisions in the final standards.
Several major changes were made to the October 4, 2004 proposed
rule as a result of OSHA's analysis of comments and data received
during the comment periods and public hearings. The major changes are
summarized below and are fully discussed in the Summary and Explanation
section of this preamble (Section XV)
Scope. As proposed, the standards apply to occupational exposures
to Cr(VI) in all forms and compounds with limited exceptions. OSHA has
made a final determination to exclude from coverage of these final
standards exposures that occur in the application of pesticides
containing Cr(VI) (e.g., the treatment of wood with preservatives).
These exposures are already covered by the Environmental Protection
Agency. OSHA is also excluding exposures to portland cement and
exposures in work settings where the employer has objective data
demonstrating that a material containing chromium or a specific
process, operation, or activity involving chromium cannot release
dusts, fumes, or mists of Cr(VI) in concentrations at or above 0.5
[mu]g/m\3\ under any expected conditions of use. OSHA believes that the
weight of evidence in this rulemaking demonstrates that the primary
risk in these two exposure scenarios can be effectively addressed
through existing OSHA standards for personal protective equipment,
hygiene, hazard communication and the PELs for portland cement or
particulates not otherwise regulated (PNOR).
Permissible Exposure Limit. OSHA proposed a PEL of 1 [mu]g/m\3\ but
has now determined that a PEL 5 [mu]g/m\3\ is the lowest level that is
technologically and economically feasible.
Exposure Determination. OSHA did not include a provision for
exposure determination in the proposed shipyard and construction
standards, reasoning that the obligation to meet the proposed PEL would
implicitly necessitate performance-based monitoring by the employer to
ensure compliance with the PEL. However, OSHA was convinced by
arguments presented during the rulemaking that an explicit requirement
for exposure determination is necessary to ensure that employee
exposures are adequately characterized. Therefore OSHA has included a
provision for exposure determination for general industry, shipyards
and construction in the final rule. In order to provide additional
flexibility in characterizing employee exposures, OSHA is allowing
employers to choose between a scheduled monitoring option and a
performance-based option for making exposure determinations.
Methods of Compliance. Under the proposed rule employers were to
use engineering and work practice controls to achieve the proposed PEL
unless the employer could demonstrate such controls are not feasible.
In the final rule, OSHA has retained this exception but has added a
provision that only requires employers to use engineering and work
practice controls to reduce or maintain employee exposures to 25 [mu]g/
m\3\ when painting aircraft or large aircraft parts in the aerospace
industry to the extent such controls are feasible. The employer must
then supplement those engineering controls with respiratory protection
to achieve the PEL. As discussed more fully in the Summary of the Final
Economic Analysis and Regulatory Flexibility Analysis (Section VIII)
and the Summary and Explanation (Section XV) OSHA has determined that
this is the lowest level achievable through the use of engineering and
work practice controls alone for these limited operations.
Housekeeping. In the proposed rule, cleaning methods such as
shoveling, sweeping, and brushing were prohibited unless they were the
only effective means available to clean surfaces contaminated with
Cr(VI). The final standard has modified this prohibition to make clear
only dry shoveling, sweeping and brushing are prohibited so that
effective wet shoveling, sweeping, and brushing would be allowed. OSHA
is also adding a provision that allows the use of compressed air to
remove Cr(VI) when no alternative method is feasible.
Medical Surveillance. As proposed and continued in these final
standards, medical surveillance is required to be provided to employees
experiencing signs or symptoms of the adverse health effects associated
with Cr(VI) exposure or exposed in an emergency. In addition, for
general industry, employees exposed above the PEL for 30 or more days a
year were to be provided medical surveillance. In the final standard,
OSHA has changed the trigger for medical surveillance to exposure above
the action level (instead of the PEL) for 30 days a year to take into
account the existing risks at the new PEL. This provision has also been
extended to the standards for shipyards and construction since those
employers now will be required to perform an exposure determination and
thus will be able to determine which employees are exposed above the
action level 30 or more days a year.
Communication of Hazards. In the proposed standard, OSHA specified
the sign for the demarcation of regulated areas in general industry and
the label for contaminated work clothing or equipment and Cr(VI)
contaminated waste and debris. The proposed standard also listed the
various elements to be covered for employee training. In order to
simplify requirements under this section of the final standard and
reduce confusion between this standard and the Hazard Communication
Standard, OSHA has removed the requirement for special signs and labels
and the specification of employee training elements. Instead, the final
standard requires that signs, labels and training be in accordance with
the Hazard Communication Standard (29 CFR 1910.1200). The only
additional training elements required in the final rule are those
related specifically to the contents of the final Cr(VI) standards.
While the final standards have removed language in the communication of
hazards provisions to make them more consistent with OSHA's existing
Hazard Communication Standard, the employers obligation to mark
regulated areas (where regulated areas are required), to label Cr(VI)
contaminated clothing and wastes, and to train on the hazards of Cr(VI)
have not changed.
Recordkeeping. In the proposed standards for shipyards and
construction there were no recordkeeping requirements for exposure
records since there was not a requirement for exposure determination.
The final standard now requires exposure determination for shipyards
and construction and therefore, OSHA has also added provisions for
exposure records to be maintained in these final standards. In keeping
with its intent to be consistent with the Hazard Communication
Standard, OSHA has removed the requirement for training records in the
final standards.
Dates. In the proposed standard, the effective date of the standard
was 60 days after the publication date; the start-up date for all
provisions except engineering controls was 90 days after the effective
date; and the start-up date for engineering controls was two years
after the effective date. OSHA believes that it is appropriate to allow
additional time for employers, particularly small employers, to meet
the requirements of the final rule. The effective and start-up dates
have been extended as follows: the effective date for the final rule is
changed to 90 days after the publication date; the start-up date for
all provisions except engineering controls is changed to 180 days after
the effective date for employers with 20 or more employees; the start-
up date for all provisions except engineering controls is changed to
one year after the effective date for employers with 19 or fewer
employees; and the start-up date for engineering controls is changed to
four years after the effective date for all employers.
II. Pertinent Legal Authority
The purpose of the Occupational Safety and Health Act, 29 U.S.C.
651 et seq. ("the Act") is to,
* * * assure so far as possible every working man and woman in the
nation safe and healthful working conditions and to preserve our
human resources. 29 U.S.C. 651(b).
To achieve this goal Congress authorized the Secretary of Labor
(the Secretary) to promulgate and enforce occupational safety and
health standards. 29 U.S.C. 654(b) (requiring employers to comply with
OSHA standards), 655(a) (authorizing summary adoption of existing
consensus and federal standards within two years of the Act's
enactment), and 655(b) (authorizing promulgation, modification or
revocation of standards pursuant to notice and comment).
The Act provides that in promulgating health standards dealing with
toxic materials or harmful physical agents, such as this standard
regulating occupational exposure to Cr(VI), the Secretary,
* * * shall set the standard which most adequately assures, to the
extent feasible, on the basis of the best available evidence that no
employee will suffer material impairment of health or functional
capacity even if such employee has regular exposure to the hazard
dealt with by such standard for the period of his working life. 29
U.S.C. Sec. 655(b)(5).
The Supreme Court has held that before the Secretary can promulgate
any permanent health or safety standard, she must make a threshold
finding that significant risk is present and that such risk can be
eliminated or lessened by a change in practices. Industrial Union
Dept., AFL-CIO v. American Petroleum Institute, 448 U.S. 607, 641-42
(1980) (plurality opinion) ("The Benzene case"). The Court further
observed that what constitutes "significant risk" is "not a
mathematical straitjacket" and must be "based largely on policy
considerations." The Benzene case, 448 U.S. at 655. The Court gave the
example that if,
* * * the odds are one in a billion that a person will die from
cancer * * * the risk clearly could not be considered significant.
On the other hand, if the odds are one in one thousand that regular
inhalation of gasoline vapors that are 2% benzene will be fatal, a
reasonable person might well consider the risk significant. * * *
Id.
OSHA standards must be both technologically and economically
feasible. United Steelworkers v. Marshall, 647 F.2d 1189, 1264 (D.C.
Cir. 1980) ("The Lead I case"). The Supreme Court has defined
feasibility as "capable of being done." American Textile Mfrs. Inst.
v. Donovan, 425 U.S. 490, 509 (1981) ("The Cotton dust case"). The
courts have further clarified that a standard is technologically
feasible if OSHA proves a reasonable possibility,
* * * within the limits of the best available evidence * * * that
the typical firm will be able to develop and install engineering and
work practice controls that can meet the PEL in most of its
operations. See The Lead I case, 647 F.2d at 1272.
With respect to economic feasibility, the courts have held that a
standard is feasible if it does not threaten massive dislocation to or
imperil the existence of the industry. See The Lead case, 647 F.2d at
1265. A court must examine the cost of compliance with an OSHA standard
"in relation to the financial health and profitability of the industry
and the likely effect of such costs on unit consumer prices." Id.
[The] practical question is whether the standard threatens the
competitive stability of an industry, * * * or whether any intra-
industry or inter-industry discrimination in the standard might
wreck such stability or lead to undue concentration. Id. (citing
Industrial Union Dept., AFL-CIO v. Hodgson, 499 F.2d 467 (D.C. Cir.
1974)).
The courts have further observed that granting companies reasonable
time to comply with new PEL's may enhance economic feasibility. Id.
While a standard must be economically feasible, the Supreme Court has
held that a cost-benefit analysis of health standards is not required
by the Act because a feasibility analysis is. The Cotton dust case, 453
U.S. at 509. Finally, unlike safety standards, health standards must
eliminate risk or reduce it to the maximum extent that is
technologically and economically feasible. See International Union,
United Automobile, Aerospace & Agricultural Implement Workers of
America, UAW v. OSHA, 938 F.2d 1310, 1313 (D.C. Cir. 1991); Control of
Hazardous Energy Sources (Lockout/Tagout), Final rule; supplemental
statement of reasons, (58 FR 16612, March 30, 1993).
III. Events Leading to the Final Standard
OSHA's previous standards for workplace exposure to Cr(VI) were
adopted in 1971, pursuant to section 6(a) of the Act, from a 1943
American National Standards Institute (ANSI) recommendation originally
tissues (36 FR at 10466, 5/29/71; Ex. 20-3). OSHA's general industry
standard set a permissible exposure limit (PEL) of 1 mg chromium
trioxide per 10 m\3\ air in the workplace (1 mg/10 m\3\
CrO3) as a ceiling concentration, which corresponds to a
concentration of 52 [mu]g/m\3\ Cr(VI). A separate rule promulgated for
the construction industry set an eight-hour time-weighted-average PEL
of 1 mg/10 m3 CrO3, also equivalent to 52 [mu]g/
m\3\ Cr(VI), adopted from the American Conference of Governmental
Industrial Hygienists (ACGIH) 1970 Threshold Limit Value (TLV) (36 FR
at 7340, 4/17/71).
Following the ANSI standard of 1943, other occupational and public
health organizations evaluated Cr(VI) as a workplace and environmental
hazard and formulated recommendations to control exposure. The ACGIH
first recommended control of workplace exposures to chromium in 1946,
recommending a time-weighted average Maximum Allowable Concentration
(later called a Threshold Limit Value) of 100 [mu]g/m\3\ for chromic
acid and chromates as Cr2O3 (Ex. 5-37), and later
classified certain Cr(VI) compounds as class A1 (confirmed human)
carcinogens in 1974. In 1975, the NIOSH Criteria for a Recommended
Standard recommended that occupational exposure to Cr(VI) compounds
should be limited to a 10-hour TWA of 1 [mu]g/m\3\, except for some
forms of Cr(VI) then believed to be noncarcinogenic (Ex. 3-92). The
National Toxicology Program's First Annual Report on Carcinogens
identified calcium chromate, chromium chromate, strontium chromate, and
zinc chromate as carcinogens in 1980 (Ex. 35-157).
During the 1980s, regulatory and standards organizations came to
recognize Cr(VI) compounds in general as carcinogens. The Environmental
Protection Agency (EPA) Health Assessment Document of 1984 stated that,
* * * using the IARC [International Agency for Research on Cancer]
classification scheme, the level of evidence available for the
combined animal and human data would place hexavalent chromium (Cr
VI) compounds into Group 1, meaning that there is decisive evidence
for the carcinogenicity of those compounds in humans (Ex. 19-1, p.
7-107).
In 1988 IARC evaluated the available evidence regarding Cr(VI)
carcinogenicity, concluding in 1990 that
* * * [t]here is sufficient evidence in humans for the
carcinogenicity of chromium[VI] compounds as encountered in the
chromate production, chromate pigment production and chromium
plating industries, [and] sufficient evidence in experimental
animals for the carcinogenicity of calcium chromate, zinc chromates,
strontium chromate and lead chromates (Ex. 18-3, p. 213).
In September 1988, NIOSH advised OSHA to consider all Cr(VI)
compounds as potential occupational carcinogens (Ex. 31-22-22). ACGIH
now classifies water-insoluble and water-soluble Cr(IV) compounds as
class A1 carcinogens (Ex. 35-207). Current ACGIH standards include
specific 8-hour time-weighted average TLVs for calcium chromate (1
[mu]g/m3), lead chromate (12 [mu]g/m3), strontium
chromate (0.5 [mu]g/m3), and zinc chromates (10 [mu]g/
m3), and generic TLVs for water soluble (50 [mu]g/
m3) and insoluble (10 [mu]g/m3) forms of
hexavalent chromium not otherwise classified, all measured as chromium
(Ex. 35-207).
In July 1993, OSHA was petitioned for an emergency temporary
standard to reduce occupational exposures to Cr(VI) compounds (Ex. 1).
The Oil, Chemical, and Atomic Workers International Union (OCAW) and
Public Citizen's Health Research Group (Public Citizen), citing
evidence that occupational exposure to Cr(VI) increases workers' risk
of lung cancer, petitioned OSHA to promulgate an emergency temporary
standard to lower the PEL for Cr(VI) compounds to 0.5 [mu]g/
m3 as an eight-hour time-weighted average (TWA). Upon review
of the petition, OSHA agreed that there was evidence of increased
cancer risk from exposure to Cr(VI) at the existing PEL, but found that
the available data did not show the "grave danger" required to
support an emergency temporary standard (Ex. 1-C). The Agency therefore
denied the request for an emergency temporary standard, but initiated
Section 6(b)(5) rulemaking and began performing preliminary analyses
relevant to the rule.
In 1997, Public Citizen petitioned the United States Court of
Appeals for the Third Circuit to compel OSHA to complete rulemaking
lowering the standard for occupational exposure to Cr(VI). The Court
denied Public Citizen's request, concluding that there was no
unreasonable delay and dismissed the suit. Oil, Chemical and Atomic
Workers Union and Public Citizen Health Research Group v. OSHA, 145
F.3d 120 (3rd Cir. 1998). Afterwards, the Agency continued its data
collection and analytic efforts on Cr(VI) (Ex. 35-208, p. 3). In 2002,
Public Citizen again petitioned the Court to compel OSHA to commence
rulemaking to lower the Cr(VI) standard (Ex. 31-24-1). Meanwhile on
August 22, 2002, OSHA published a Request for Information on Cr(VI) to
solicit additional information on key issues related to controlling
exposures to Cr(VI) (FR 67 at 54389), and on December 4, 2002 announced
its intent to proceed with developing a proposed standard (Ex. 35-306).
On December 24, 2002, the Court granted Public Citizen's petition, and
ordered the Agency to proceed expeditiously with a Cr(VI) standard. See
Public Citizen Health Research Group v. Chao, 314 F.3d 143 (3rd Cir.
2002)). In a subsequent order, the Court established a compressed
schedule for completion of the rulemaking, with deadlines of October 4,
2004 for publication of a proposed standard and January 18, 2006 for
publication of a final standard (Ex. 35-304).
In 2003, as required by the Small Business Regulatory Enforcement
Act (SBREFA), OSHA initiated SBREFA proceedings, seeking the advice of
small business representatives on the proposed rule. The SBREFA panel,
including representatives from OSHA, the Small Business Administration
(SBA), and the Office of Management and Budget (OMB), was convened on
December 23, 2003. The panel conferred with representatives from small
entities in chemical, alloy, and pigment manufacturing, electroplating,
welding, aerospace, concrete, shipbuilding, masonry, and construction
on March 16-17, 2004, and delivered its final report to OSHA on April
20, 2004. The Panel's report, including comments from the small entity
representatives (SERS) and recommendations to OSHA for the proposed
rule, is available in the Cr(VI) rulemaking docket (Ex. 34). The SBREFA
Panel made recommendations on a variety of subjects. The most important
recommendations with respect to alternatives that OSHA should consider
included: A higher PEL than the PEL of 1; excluding cement from the
scope of the standard; the use of SECALs for some industries; different
PELS for different Hexavalent chromium compounds; a multi-year phase-in
to the standards; and further consideration to approaches suited to the
special conditions of the maritime and construction industries. OSHA
has adapted many of these recommendations: The PEL is now 5; cement has
been excluded from the scope of the standard; a compliance alternative,
similar to a SECAL, has been used in aerospace industry; the standard
allows four years to phase in engineering controls; and a new
performance based monitoring approach for all industries, among other
changes, all of which should make it easier for all
industries with changing work place conditions to meet the standard in
a cost effective way. A full discussion of all of the recommendations,
and OSHA's responses to them, is provided in Section VIII of this
Preamble.
In addition to undertaking SBREFA proceedings, in early 2004, OSHA
provided the Advisory Committee on Construction Safety and Health
(ACCSH) and the Maritime Advisory Committee on Occupational Safety and
Health (MACOSH) with copies of the draft proposed rule for review. OSHA
representatives met with ACCSH in February 2004 and May 2004 to discuss
the rulemaking and receive their comments and recommendations. On
February 13, 2004, ACCSH recommended that portland cement should be
included within the scope of the proposed standard (Ex. 35-307, pp.
288-293) and that identical PELs should be set for construction,
maritime, and general industry (Ex. 35-307, pp. 293-297). On May 18,
2004, ACCSH recommended that the construction industry should be
included in the current rulemaking, and affirmed its earlier
recommendation regarding portland cement. OSHA representatives met with
MACOSH in March 2004. On March 3, 2004, MACOSH collected and forwarded
additional exposure monitoring data to OSHA to help the Agency better
evaluate exposures to Cr(VI) in shipyards (Ex. 35-309, p. 208). MACOSH
also recommended a separate Cr(VI) standard for the maritime industry,
arguing that maritime involves different exposures and requires
different means of exposure control than general industry and
construction (Ex. 35-309, p. 227).
In accordance with the Court's rulemaking schedule, OSHA published
the proposed standard for hexavalent chromium on October 4, 2004 (69 FR
at 59306). The proposal included a notice of public hearing in
Washington, DC (69 FR at 59306, 59445-59446). The notice also invited
interested persons to submit comments on the proposal until January 3,
2005. In the proposal, OSHA solicited public input on 65 issues
regarding the human health risks of Cr(VI) exposure, the impact of the
proposed rule on Cr(VI) users, and other issues of particular interest
to the Agency (69 FR at 59306-59312).
OSHA convened the public hearing on February 1, 2005, with
Administrative Law Judges John M. Vittone and Thomas M. Burke
presiding. At the conclusion of the hearing on February 15, 2005, Judge
Burke set a deadline of March 21, 2005, for the submission of post
hearing comments, additional information and data relevant to the
rulemaking, and a deadline of April 20, 2005, for the submission of
additional written comments, arguments, summations, and briefs. A wide
range of employees, employers, union representatives, trade
associations, government agencies and other interested parties
participated in the public hearing or contributed written comments.
Issues raised in their comments and testimony are addressed in the
relevant sections of this preamble (e.g., comments on the risk
assessment are discussed in section VI; comments on the benefits
analysis in section VIII). On December 22, 2005, OSHA filed a motion
with the U.S. Court of Appeals for the Third Circuit requesting an
extension of the court-mandated deadline for the publication of the
final rule by six weeks, to February 28, 2006 (Ex. 48-13). The Court
granted the request on January 17, 2006 (Ex. 48-15).
As mandated by the Act, the final standard on occupational exposure
to hexavalent chromium is based on careful consideration of the entire
record of this proceeding, including materials discussed or relied upon
in the proposal, the record of the hearing, and all written comments
and exhibits received.
OSHA has developed separate final standards for general industry,
shipyards, and the construction industry. The Agency has concluded that
excess exposure to Cr(VI) in any form poses a significant risk of
material impairment to the health of workers, by causing or
contributing to adverse health effects including lung cancer, non-
cancer respiratory effects, and dermal effects. OSHA determined that
the TWA PEL should not be set above 5 [mu]g/m3 based on the
evidence in the record and its own quantitative risk assessment. The
TWA PEL of 5 [mu]g/m3 reduces the significant risk posed to
workers by occupational exposure to Cr(VI) to the maximum extent that
is technologically and economically feasible. (See discussion of the
PEL in Section XV below.)
IV. Chemical Properties and Industrial Uses
Chromium is a metal that exists in several oxidation or valence
states, ranging from chromium (-II) to chromium (+VI). The elemental
valence state, chromium (0), does not occur in nature. Chromium
compounds are very stable in the trivalent state and occur naturally in
this state in ores such as ferrochromite, or chromite ore
(FeCr2O4). The hexavalent, Cr(VI) or chromate, is
the second most stable state. It rarely occurs naturally; most Cr(VI)
compounds are man made.
Chromium compounds in higher valence states are able to undergo
"reduction" to lower valence states; chromium compounds in lower
valence states are able to undergo "oxidation" to higher valence
states. Thus, Cr(VI) compounds can be reduced to Cr(III) in the
presence of oxidizable organic matter. Chromium can also be reduced in
the presence of inorganic chemicals such as iron.
Chromium does exist in less stable oxidation (valence) states such
as Cr(II), Cr(IV), and Cr(V). Anhydrous Cr(II) salts are relatively
stable, but the divalent state (II, or chromous) is generally
relatively unstable and is readily oxidized to the trivalent (III or
chromic) state. Compounds in valence states such as (IV) and (V)
usually require special handling procedures as a result of their
instability. Cr(IV) oxide (CrO2) is used in magnetic
recording and storage devices, but very few other Cr(IV) compounds have
industrial use. Evidence exists that both Cr(IV) and Cr(V) are formed
as transient intermediates in the reduction of Cr(VI) to Cr(III) in the
body.
Chromium (III) is also an essential nutrient that plays a role in
glucose, fat, and protein metabolism by causing the action of insulin
to be more effective. Chromium picolinate, a trivalent form of chromium
combined with picolinic acid, is used as a dietary supplement, because
it is claimed to speed metabolism.
Elemental chromium and the chromium compounds in their different
valence states have various physical and chemical properties, including
differing solubilities. Most chromium species are solid. Elemental
chromium is a steel gray solid, with high melting and boiling points
(1857 [deg]C and 2672 [deg]C, respectively), and is insoluble in water
and common organic solvents. Chromium (III) chloride is a violet or
purple solid, with high melting and sublimation points (1150 [deg]C and
1300 [deg]C, respectively), and is slightly soluble in hot water and
insoluble in common organic solvents. Ferrochromite is a brown-black
solid; chromium (III) oxide is a green solid; and chromium (III)
sulfate is a violet or red solid, insoluble in water and slightly
soluble in ethanol. Chromium (III) picolinate is a ruby red crystal
soluble in water (1 part per million at 25 [deg]C). Chromium (IV) oxide
is a brown-black solid that decomposes at 300 [deg]C and is insoluble
in water.
Cr(VI) compounds have mostly lemon yellow to orange to dark red
hues. They are typically crystalline, granular, or powdery although one
compound (chromyl chloride) exists in liquid form. For example, chromyl
chloride is a dark red liquid that decomposes into chromate ion and
hydrochloric acid in water. Chromic acids are dark red crystals that are
very soluble in water. Other examples of soluble chromates are sodium
chromate (yellow crystals) and sodium dichromate (reddish to bright orange
crystals). Lead chromate oxide is typically a red crystalline powder. Zinc
chromate is typically seen as lemon yellow crystals which decompose in
hot water and are soluble in acids and liquid ammonia. Other chromates
such as barium, calcium, lead, strontium, and zinc chromates vary in
color from light yellow to greenish yellow to orange-yellow and exist
in solid form as crystals or powder.
The Color Pigments Manufacturers Association (CPMA) provided
additional information on lead chromate and some other chromates used
in their pigments (Ex. 38-205, pp. 12-13). CPMA describes two main lead
chromate color groups: the chrome yellow pigments and the orange to red
varieties known as molybdate orange pigments. The chrome yellow
pigments are solid solution crystal compositions of lead chromate and
lead sulfate. Molybdate orange pigments are solid solution crystal
compositions of lead chromate, lead sulfate, and lead molybdate (Ex.
38-205, p. 12). CPMA also describes a basic lead chromate called
"chrome orange," and a lead chromate precipitated "onto a core" of
silica (Ex. 38-205, p. 13).
OSHA re-examined available information on solubility values in
light of comments from the CPMA and Dominion Color Corporation (DCC) on
qualitative solubility designations and CPMA's claim of low
bioavailability of lead chromate due to its extremely low solubility
(Exs. 38-201-1, p. 4; 38-205, p. 95). There was not always agreement or
consistency with the qualitative assignments of solubilities.
Quantitative values for the same compound also differ depending on the
source of information.
The Table IV-1 is the result of OSHA's re-examination of
quantitative water solubility values and qualitative designations.
Qualitative designations as well as quantitative values are listed as
they were provided by the source. As can be seen by the Table IV-1,
qualitative descriptions vary by the descriptive terminology chosen by
the source.
BILLING CODE 4510-26-P

BILLING CODE 4510-26-C
OSHA has made some generalizations to describe the water
solubilities of chromates in subsequent sections of this Federal
Register notice. OSHA has divided Cr(VI) compounds and mixtures into
three categories based on solubility values. Compounds and mixtures
with water solubilities less than 0.01 g/l are referred to as water
insoluble. Compounds and mixtures between 0.01 g/l and 500 g/l are
referred to as slightly soluble. Compounds and mixtures with water
solubility values of 500 g/l or greater are referred to as highly
water soluble. It should be noted that these boundaries for insoluble,
slightly soluble, and highly soluble are arbitrary designations for the
sake of further description elsewhere in this document. Quantitative
values take precedence over qualitative designations. For example, zinc
chromates would be slightly soluble where their solubility values exceed 0.01 g/l.
Some major users of chromium are the metallurgical, refractory, and
chemical industries. Chromium is used by the metallurgical industry to
produce stainless steel, alloy steel, and nonferrous alloys. Chromium
is alloyed with other metals and plated on metal and plastic substrates
to improve corrosion resistance and provide protective coatings for
automotive and equipment accessories. Welders use stainless steel
welding rods when joining metal parts.
Cr(VI) compounds are widely used in the chemical industry in
pigments, metal plating, and chemical synthesis as ingredients and
catalysts. Chromates are used as high quality pigments for textile
dyes, paints, inks, glass, and plastics. Cr(VI) can be produced during
welding operations even if the chromium was originally present in
another valence state. While Cr(VI) is not intentionally added to
portland cement, it is often present as an impurity.
Occupational exposures to Cr(VI) can occur from inhalation of mists
(e.g., chrome plating, painting), dusts (e.g., inorganic pigments), or
fumes (e.g., stainless steel welding), and from dermal contact (e.g.,
cement workers).
There are about thirty major industries and processes where Cr(VI)
is used. These include producers of chromates and related chemicals
from chromite ore, electroplating, welding, painting, chromate pigment
production and use, steel mills, and iron and steel foundries. A
detailed discussion of the uses of Cr(VI) in industry is found in
Section VIII of this preamble.
V. Health Effects
This section summarizes key studies of adverse health effects
resulting from exposure to hexavalent chromium (Cr(VI)) in humans and
experimental animals, as well as information on the fate of Cr(VI) in
the body and laboratory research that relates to its toxic mode of
action. The primary health impairments from workplace exposure to
Cr(VI) are lung cancer, asthma, and damage to the nasal epithelia and
skin. While this chapter on health effects does not describe all of the
many studies that have been conducted on Cr(VI) toxicity, it includes a
selection of those that are relevant to the rulemaking and
representative of the scientific literature on Cr(VI) health effects.
A. Absorption, Distribution, Metabolic Reduction and Elimination
Although chromium can exist in a number of different valence
states, Cr(VI) is the form considered to be the greatest health risk.
Cr(VI) enters the body by inhalation, ingestion, or absorption through
the skin. For occupational exposure, the airways and skin are the
primary routes of uptake. The following discussion summarizes key
aspects of Cr(VI) uptake, distribution, metabolism, and elimination.
1. Deposition and Clearance of Inhaled Cr(VI) From the Respiratory
Tract
Various anatomical, physical and physiological factors determine
both the fractional and regional deposition of inhaled particulate
matter. Due to the airflow patterns in the lung, more particles tend to
deposit at certain preferred regions in the lung. It is therefore
possible to have a buildup of chromium at certain sites in the
bronchial tree that could create areas of very high chromium
concentration. A high degree of correspondence between the efficiency
of particle deposition and the frequency of bronchial tumors at sites
in the upper bronchial tree was reported in research by Schlesinger and
Lippman that compared the distribution of cancer sites in published
reports of primary bronchogenic tumors with experimentally determined
particle deposition patterns (Ex. 35-102).
Large inhaled particles (>5 [mu]m) are efficiently removed from the
air-stream in the extrathoracic region (Ex. 35-175). Particles greater
than 2.5 [mu]m are generally deposited in the tracheobronchial regions,
whereas particles less than 2.5 [mu]m are generally deposited in the
pulmonary region. Some larger particles (>2.5 [mu]m) can reach the
pulmonary region. The mucociliary escalator predominantly clears
particles that deposit in the extrathoracic and the tracheobronchial
region of the lung. Individuals exposed to high particulate levels of
Cr(VI) may also have altered respiratory mucociliary clearance.
Particulates that reach the alveoli can be absorbed into the
bloodstream or cleared by phagocytosis.
2. Absorption of Inhaled Cr(VI) Into the Bloodstream
The absorption of inhaled chromium compounds depends on a number of
factors, including physical and chemical properties of the particles
(oxidation state, size, solubility) and the activity of alveolar
macrophages (Ex. 35-41). The hexavalent chromate anions
(CrO4)2- enter cells via facilitated diffusion
through non-specific anion channels (similar to phosphate and sulfate
anions). As demonstrated in research by Suzuki et al., a portion of
water soluble Cr(VI) is rapidly transported to the bloodstream in rats
(Ex. 35-97). Rats were exposed to 7.3-15.9 mg Cr(VI)/m\3\ as potassium
dichromate for 2-6 hours. Following exposure to Cr(VI), the ratio of
blood chromium/lung chromium was 1.440.30 at 0.5 hours,
0.810.10 at 18 hours, 0.850.20 at 48 hours, and
0.960.22 at 168 hours after exposure.
Once the Cr(VI) particles reach the alveoli, absorption into the
bloodstream is greatly dependent on solubility. More soluble chromates
are absorbed faster than water insoluble chromates, while insoluble
chromates are poorly absorbed and therefore have longer resident time
in the lungs. This effect has been demonstrated in research by Bragt
and van Dura on the kinetics of three Cr(VI) compounds: highly soluble
sodium chromate, slightly soluble zinc chromate and water insoluble
lead chromate (Ex. 35-56). They instilled \51\chromium-labeled
compounds (0.38 mg Cr(VI)/kg as sodium chromate, 0.36 mg Cr(VI)/kg as
zinc chromate, or 0.21 mg Cr(VI)/kg as lead chromate) intratracheally
in rats. Peak blood levels of \51\chromium were reached after 30
minutes for sodium chromate (0.35 [mu]g chromium/ml), and after 24
hours for zinc chromate (0.60 [mu]g chromium/ml) and lead chromate
(0.007 [mu]g chromium/ml). At 30 minutes after administration, the
lungs contained 36, 25, and 81% of the respective dose of the sodium,
zinc, and lead chromate. On day six, >80% of the dose of all three
compounds had been cleared from the lungs, during which time the
disappearance from lungs followed linear first-order kinetics. The
residual amount left in the lungs on day 50 or 51 was 3.0, 3.9, and
13.9%, respectively. From these results authors concluded that zinc
chromate, which is less soluble than sodium chromate, is more slowly
absorbed from the lungs. Lead chromate was more poorly and slowly
absorbed, as indicated by very low levels in blood and greater
retention in the lungs. The authors also noted that the kinetics of
sodium and zinc chromates were very similar. Zinc chromate, which is
less soluble than sodium chromate, was slowly absorbed from the lung,
but the maximal blood levels were higher than those resulting from an
equivalent dose of sodium chromate. The authors believe that this was
probably the result of hemorrhages macroscopically visible in the lungs
of zinc chromate-treated rats 24 hours following intratracheal administration.
Boeing Corporation commented that this study does not show that the highly water
soluble sodium chromate is cleared more rapidly or retained in the lung for
shorter periods than the less soluble zinc chromate (Ex. 38-106-2, p.
18-19). This comment is addressed in the Carcinogenic Effects
Conclusion Section V.B.9 dealing with the carcinogenicity of slightly
soluble Cr(VI) compounds.
Studies by Langard et al. and Adachi et al. provide further
evidence of absorption of chromates from the lungs (Exs. 35-93; 189).
In Langard et al., rats exposed to 2.1 mg Cr(VI)/m\3\ as zinc chromate
for 6 hours/day achieved steady state concentrations in the blood after
4 days of exposure (Ex. 35-93). Adachi et al. studied rats that were
subject to a single inhalation exposure to chromic acid mist generated
from electroplating at a concentration of 3.18 mg Cr(VI)/m\3\ for 30
minutes which was then rapidly absorbed from the lungs (Ex. 189). The
amount of chromium in the lungs of these rats declined from 13.0 mg
immediately after exposure to 1.1 mg after 4 weeks, with an overall
half-life of five days.
Several other studies have reported absorption of chromium from the
lungs after intratracheal instillation (Exs. 7-9; 9-81; Visek et al.
1953 as cited in Ex. 35-41). These studies indicated that 53-85% of
Cr(VI) compounds (particle size < 5 [mu]m) were cleared from the lungs
by absorption into the bloodstream or by mucociliary clearance in the
pharynx; the rest remained in the lungs. Absorption of Cr(VI) from the
respiratory tract of workers has been shown in several studies that
identified chromium in the urine, serum and red blood cells following
occupational exposure (Exs. 5-12; 35-294; 35-84).
Evidence indicates that even chromates encapsulated in a paint
matrix may be released in the lungs (Ex. 31-15, p. 2). In a study of
chromates in aircraft spray paint, LaPuma et al. measured the mass of
Cr(VI) released from particles into water originating from three types
of paint particles: solvent-borne epoxy (25% strontium chromate
(SrCrO4)), water-borne epoxy (30% SrCrO4) and
polyurethane (20% SrCrO4) (Ex. 31-2-1). The mean fraction of
Cr(VI) released into the water after one and 24 hours for each primer
averaged: 70% and 85% (solvent epoxy), 74% and 84% (water epoxy), and
94% and 95% (polyurethane). Correlations between particle size and the
fraction of Cr(VI) released indicated that smaller particles (< 5 [mu]m)
release a larger fraction of Cr(VI) versus larger particles (>5 [mu]m).
This study demonstrates that the paint matrix only modestly hinders
Cr(VI) release into a fluid, especially with smaller particles. Larger
particles, which contain the majority of Cr(VI) due to their size,
appear to release proportionally less Cr(VI) (as a percent of total
Cr(VI)) than smaller particles. Some commenters suggested that the
above research shows that the slightly soluble Cr(VI) from aircraft
spray paint is less likely to reach and be absorbed in the
bronchoalveolar region of the lung than a highly soluble Cr(VI) form,
such as chromic acid aerosol (Exs. 38-106-2; 39-43, 44-33). This issue
is further discussed in the Carcinogenic Effects Conclusion Section
V.B.9.a and in the Quantitative Risk Assessment Section VI.G.4.a.
A number of questions remain unanswered regarding encapsulated
Cr(VI) and bioavailability from the lung. There is a lack of detailed
information on the efficiency of encapsulation and whether all of the
chromate molecules are encapsulated. The stability of the encapsulated
product in physiological and environmental conditions over time has not
been demonstrated. Finally, the fate of inhaled encapsulated Cr(VI) in
the respiratory tract and the extent of distribution in systemic
tissues has not been thoroughly studied.
3. Dermal Absorption of Cr(VI)
Both human and animal studies demonstrate that Cr(VI) compounds are
absorbed after dermal exposure. Dermal absorption depends on the
oxidation state of chromium, the vehicle and the integrity of the skin.
Cr(VI) readily traverses the epidermis to the dermis (Exs. 9-49; 309).
The histological distribution of Cr(VI) within intact human skin was
studied by Liden and Lundberg (Ex. 35-80). They applied test solutions
of potassium dichromate in petrolatum or in water as occluded circular
patches of filter paper to the skin. Results with potassium dichromate
in water revealed that Cr(VI) penetrated beyond the dermis and
penetration reached steady state with resorption by the lymph and blood
vessels by 5 hours. About 10 times more chromium penetrated when
potassium dichromate was applied in petrolatum than when applied in
water, indicating that organic solvents facilitate the absorption of
Cr(VI) from the skin. Research by Baranowska-Dutkiewicz also
demonstrated that the absorption rates of sodium chromate solutions
from the occluded forearm skin of volunteers increase with increasing
concentration (Ex. 35-75). The rates were 1.1 [mu]g Cr(VI)/cm\2\/hour
for a 0.01 molar solution, 6.4 [mu]g Cr(VI)/cm\2\/hour for a 0.1 molar
solution, and 10 [mu]g Cr(VI)/cm\2\/hour for a 0.2 molar solution.
Additional studies have demonstrated that the absorption of Cr(VI)
compounds can take place through the dermal route. Using volunteers,
Mali found that potassium dichromate penetrates the intact epidermis
(Exs. 9-49; 35-41). Wahlberg and Skog demonstrated the presence of
chromium in the blood, spleen, bone marrow, lymph glands, urine and
kidneys of guinea pigs dermally exposed to \51\chromium labeled Cr(VI)
compounds (Ex. 35-81).
4. Absorption of Cr(VI) by the Oral Route
Inhaled Cr(VI) can enter the digestive tract as a result of
mucocilliary clearance and swallowing. Studies indicate Cr(VI) is
absorbed from the gastrointestinal tract. For example, in a study by
Donaldson and Barreras, the six-day fecal and 24-hour urinary excretion
patterns of radioactivity in groups of six volunteers given Cr(VI) as
sodium chromate labeled with \51\chromium indicated that at least 2.1%
of the Cr(VI) was absorbed. After intraduodenal administration at least
10% of the Cr(VI) compound was absorbed. These studies also
demonstrated that Cr(VI) compounds are reduced to Cr(III) compounds in
the stomach, thereby accounting for the relatively poor
gastrointestinal absorption of orally administered Cr(VI) compounds
(Exs. 35-96; 35-41). In the gastrointestinal tract, Cr(VI) can be
reduced to Cr(III) by gastric juices, which is then poorly absorbed
(Underwood, 1971 as cited in Ex. 19-1; Ex. 35-85).
In a study conducted by Clapp et al., treatment of rats by gavage
with an unencapsulated lead chromate pigment or with a silica-
encapsulated lead chromate pigment resulted in no measurable blood
levels of chromium (measured as Cr(III), detection limit = 10 [mu]g/L)
after two or four weeks of treatment or after a two-week recovery
period. However, kidney levels of chromium (measured as Cr(III)) were
significantly higher in the rats that received the unencapsulated
pigment when compared to the rats that received the encapsulated
pigment, indicating that silica encapsulation may reduce the
gastrointestinal bioavailability of chromium from lead chromate
pigments (Ex. 11-5). This study does not address the bioavailability of
encapsulated chromate pigments from the lung where residence time could
be different.
5. Distribution of Cr(VI) in the Body
Once in the bloodstream, Cr(VI) is taken up into erythrocytes,
where it is reduced to lower oxidation states and forms chromium
protein complexes during reduction (Ex. 35-41). Once complexed with
protein, chromium cannot leave the cell and chromium ions are unable to
repenetrate the membrane and move back into the plasma (Exs. 7-6; 7-7;
19-1; 35-41; 35-52). Once inside the blood cell, the intracellular
Cr(VI) reduction to Cr(III) depletes Cr(VI) concentration in the red
blood cell (Ex. 35-89). This serves to enhance diffusion of Cr(VI) from
the plasma into the erythrocyte resulting in very low plasma levels of
Cr(VI). It is also believed that the rate of uptake of Cr(VI) by red
blood cells may not exceed the rate at which they reduce Cr(VI) to
Cr(III) (Ex. 35-99). The higher tissue levels of chromium after
administration of Cr(VI) than after administration of Cr(III) reflect
the greater tendency of Cr(VI) to traverse plasma membranes and bind to
intracellular proteins in the various tissues, which may explain the
greater degree of toxicity associated with Cr(VI) (MacKenzie et al.
1958 as cited in 35-52; Maruyama 1982 as cited in 35-41; Ex. 35-71).
Examination of autopsy tissues from chromate workers who were
occupationally exposed to Cr(VI) showed that the highest chromium
levels were in the lungs. The liver, bladder, and bone also had
chromium levels above background. Mancuso examined tissues from three
individuals with lung cancer who were exposed to chromium in the
workplace (Ex. 124). One was employed for 15 years as a welder, the
second and third worked for 10.2 years and 31.8 years, respectively, in
ore milling and preparations and boiler operations. The cumulative
chromium exposures for the three workers were estimated to be 3.45,
4.59, and 11.38 mg/m\3\-years, respectively. Tissues from the first
worker were analyzed 3.5 years after last exposure, the second worker
18 years after last exposure, and the third worker 0.6 years after last
exposure. All tissues from the three workers had elevated levels of
chromium, with the possible exception of neural tissues. Levels were
orders of magnitude higher in the lungs when compared to other tissues.
Similar results were also reported in autopsy studies of people who may
have been exposed to chromium in the workplace as well as chrome
platers and chromate refining workers (Exs. 35-92; 21-1; 35-74; 35-88).
Animal studies have shown similar distribution patterns after
inhalation exposure. For example, a study by Baetjer et al.
investigated the distribution of Cr(VI) in guinea pigs after
intratracheal instillation of slightly soluble potassium dichromate
(Ex. 7-8). At 24 hours after instillation, 11% of the original dose of
chromium from potassium dichromate remained in the lungs, 8% in the
erythrocytes, 1% in plasma, 3% in the kidney, and 4% in the liver. The
muscle, skin, and adrenal glands contained only a trace. All tissue
concentrations of chromium declined to low or nondetectable levels in
140 days, with the exception of the lungs and spleen.
6. Metabolic Reduction of Cr(VI)
Cr(VI) is reduced to Cr(III) in the lungs by a variety of reducing
agents. This serves to limit uptake into lung cells and absorption into
the bloodstream. Cr(V) and Cr(IV) are transient intermediates in this
process. The genotoxic effects produced by the Cr(VI) are related to
the reduction process and are further discussed in the section V.B.8 on
Mechanistic Considerations.
In vivo and in vitro experiments in rats indicated that, in the
lungs, Cr(VI) can be reduced to Cr(III) by ascorbate and glutathione. A
study by Suzuki and Fukuda showed that the reduction of Cr(VI) by
glutathione is slower than the reduction by ascorbate (Ex. 35-65).
Other studies have reported the reduction of Cr(VI) to Cr(III) by
epithelial lining fluid (ELF) obtained from the lungs of 15 individuals
by bronchial lavage. The average overall reduction capacity was 0.6
[mu]g Cr(VI)/mg of ELF protein. In addition, cell extracts made from
pulmonary alveolar macrophages derived from five healthy male
volunteers were able to reduce an average of 4.8 [mu]g Cr(VI)/10\6\
cells or 14.4 [mu]g Cr(VI)/mg protein (Ex. 35-83). Postmitochondrial
(S12) preparations of human lung cells (peripheral lung parenchyma and
bronchial preparations) were also able to reduce Cr(VI) to Cr(III) (De
Flora et al. 1984 as cited in Ex. 35-41).
7. Elimination of Cr(VI) From the Body
Excretion of chromium from Cr(VI) compounds is predominantly in the
urine, although there is some biliary excretion into the feces. In both
urine and feces, the chromium is present as low molecular weight
Cr(III) complexes. Absorbed chromium is excreted from the body in a
rapid phase representing clearance from the blood and at least two
slower phases representing clearance from tissues. Urinary excretion
accounts for over 50% of eliminated chromium (Ex. 35-41). Although
chromium is excreted in urine and feces, the intestine plays only a
minor part in chromium elimination, representing only about 5% of
elimination from the blood (Ex. 19-1). Normal urinary levels of
chromium in humans have been reported to range from 0.24-1.8 [mu]g/L
with a median level of 0.4 [mu]g/L (Ex. 35-79). Humans exposed to 0.01-
0.1 mg Cr(VI)/m\3\ as potassium dichromate (8-hour time-weighted
average) had urinary excretion levels from 0.0247 to 0.037 mg Cr(III)/
L. Workers exposed mainly to Cr(VI) compounds had higher urinary
chromium levels than workers exposed primarily to Cr(III) compounds. An
analysis of the urine did not detect Cr(VI), indicating that Cr(VI) was
rapidly reduced before excretion (Exs. 35-294; 5-48).
A half-life of 15-41 hours has been estimated for chromium in urine
for four welders using a linear one-compartment kinetic model (Exs. 35-
73; 5-52; 5-53). Limited work on modeling the absorption and deposition
of chromium indicates that adipose and muscle tissue retain chromium at
a moderate level for about two weeks, while the liver and spleen store
chromium for up to 12 months. The estimated half-life for whole body
chromium retention is 22 days for Cr(VI) (Ex. 19-1). The half-life of
chromium in the human lung is 616 days, which is similar to the half-
life in rats (Ex. 7-5).
Elimination of chromium was shown to be very slow in rats exposed
to 2.1 mg Cr(VI)/m\3\ as zinc chromate six hours/day for four days.
Urinary levels of chromium remained almost constant for four days after
exposure and then decreased (Ex. 35-93). After intratracheal
administration of sodium dichromate to rats, peak urinary chromium
concentrations were observed at six hours, after which the urinary
concentrations declined rapidly (Ex. 35-94). The more prolonged
elimination of the moderately soluble zinc chromate as compared to the
more soluble sodium dichromate is consistent with the influence of
Cr(VI) solubility on absorption from the respiratory tract discussed
earlier.
Information regarding the excretion of chromium in humans after
dermal exposure to chromium or its compounds is limited. Fourteen days
after application of a salve containing water soluble potassium
chromate, which resulted in skin necrosis and sloughing at the
application site, chromium was found at 8 mg/L in the urine and 0.61
mg/100 g in the feces of one individual (Brieger 1920 as cited in Ex.
19-1). A slight increase over background levels of urinary chromium was
observed in four subjects submersed in a tub of chlorinated water containing
22 mg Cr(VI)/L as potassium dichromate for three hours (Ex. 31-22-6). For
three of the four subjects, the increase in urinary chromium excretion
was less than 1 [mu]g/day over the five-day collection period. Chromium
was detected in the urine of guinea pigs after radiolabeled sodium
chromate solution was applied to the skin (Ex. 35-81).
8. Physiologically-Based Pharmacokinetic Modeling
Physiologically-based pharmacokinetic (PBPK) models have been
developed that simulate absorption, distribution, metabolism, and
excretion of Cr(VI) and Cr(III) compounds in humans (Ex. 35-95) and
rats (Exs. 35-86; 35-70). The original model (Ex. 35-86) evolved from a
similar model for lead, and contained compartments for the lung, GI
tract, skin, blood, liver, kidney, bone, well-perfused tissues, and
slowly perfused tissues. The model was refined to include two lung
subcompartments for chromium, one of which allowed inhaled chromium to
enter the blood and GI tract and the other only allowed chromium to
enter the GI tract (Ex. 35-70). Reduction of Cr(VI) to Cr(III) was
considered to occur in every tissue compartment except bone.
The model was developed from several data sets in which rats were
dosed with Cr(VI) or Cr(III) intravenously, orally or by intratracheal
instillation, because different distribution and excretion patterns
occur depending on the route of administration. In most cases, the
model parameters (e.g., tissue partitioning, absorption, reduction
rates) were estimated by fitting model simulations to experimental
data. The optimized rat model was validated against the 1978 Langard
inhalation study (Ex. 35-93). Chromium blood levels were overpredicted
during the four-day inhalation exposure period, but blood levels during
the post-exposure period were well predicted by the model. The model-
predicted levels of liver chromium were high, but other tissue levels
were closely estimated.
A human PBPK model recently developed by O'Flaherty et al. is able
to predict tissue levels from ingestion of Cr(VI) (Ex. 35-95). The
model incorporates differential oral absorption of Cr(VI) and Cr(III),
rapid reduction of Cr(VI) to Cr(III) in major body fluids and tissues,
and concentration-dependent urinary clearance. The model does not
include a physiologic lung compartment, but can be used to estimate an
upper limit on pulmonary absorption of inhaled chromium. The model was
calibrated against blood and urine chromium concentration data from a
group of controlled studies in which adult human volunteers drank
solutions of soluble Cr(III) or Cr(VI).
PBPK models are increasingly used in risk assessments, primarily to
predict the concentration of a potentially toxic chemical that will be
delivered to any given target tissue following various combinations of
route, dose level, and test species. Further development of the
respiratory tract portion of the model, specific Cr(VI) rate data on
extracellular reduction and uptake into lung cells, and more precise
understanding of critical pathways inside target cells would improve
the model value for risk assessment purposes.
9. Summary
Based on the studies presented above, evidence exists in the
literature that shows Cr(VI) can be systemically absorbed by the
respiratory tract. The absorption of inhaled chromium compounds depends
on a number of factors, including physical and chemical properties of
the particles (oxidation state, size, and solubility), the reduction
capacity of the ELF and alveolar macrophages and clearance by the
mucocliary escalator and phagocytosis. Highly water soluble Cr(VI)
compounds (e.g. sodium chromate) enter the bloodstream more readily
than highly insoluble Cr(VI) compounds (e.g. lead chromate). However,
insoluble compounds may have longer residence time in lung. Absorption
of Cr(VI) can also take place after oral and dermal exposure,
particularly if the exposures are high.
The chromate (CrO4) 2- enters cells via
facilitated diffusion through non-specific anion channels (similar to
phosphate and sulfate anions). Following absorption of Cr(VI) compounds
from various exposure routes, chromium is taken up by the blood cells
and is widely distributed in tissues as Cr(VI). Inside blood cells and
tissues, Cr(VI) is rapidly reduced to lower oxidation states and bound
to macromolecules which may result in genotoxic or cytotoxic effects.
However, in the blood a substantial proportion of Cr(VI) is taken up
into erythrocytes, where it is reduced to Cr(III) and becomes bound to
hemoglobin and other proteins.
Inhaled Cr(VI) is reduced to Cr(III) in vivo by a variety of
reducing agents. Ascorbate and glutathione in the ELF and macrophages
have been shown to reduce Cr(VI) to Cr(III) in the lungs. After oral
exposure, gastric juices are also responsible for reducing Cr(VI) to
Cr(III). This serves to limit the amount of Cr(VI) systemically
absorbed.
Absorbed chromium is excreted from the body in a rapid phase
representing clearance from the blood and at least two slower phases
representing clearance from tissues. Urinary excretion is the primary
route of elimination, accounting for over 50% of eliminated chromium.
Although chromium is excreted in urine and feces, the intestine plays
only a minor part in chromium elimination representing only about 5% of
elimination from the blood.
B. Carcinogenic Effects
There has been extensive study on the potential for Cr(VI) to cause
carcinogenic effects, particularly cancer of the lung. OSHA reviewed
epidemiologic data from several industry sectors including chromate
production, chromate pigment production, chromium plating, stainless
steel welding, and ferrochromium production. Supporting evidence from
animal studies and mechanistic considerations are also evaluated in
this section.
1. Evidence from Chromate Production Workers
The epidemiologic literature of workers in the chromate production
industry represents the earliest and best-documented relationship
between exposure to chromium and lung cancer. The earliest study of
chromate production workers in the United States was reported by Machle
and Gregorius in 1948 (Ex. 7-2). In the United States, two chromate
production plants, one in Baltimore, MD, and one in Painesville, OH,
have been the subject of multiple studies. Both plants were included in
the 1948 Machle and Gregorius study and again in the study conducted by
the Public Health Service and published in 1953 (Ex. 7-3). Both of
these studies reported the results in aggregate. The Baltimore chromate
production plant was studied by Hayes et al. (Ex. 7-14) and more
recently by Gibb et al. (Ex. 31-22-11). The chromate production plant
in Painesville, OH, has been followed since the 1950s by Mancuso with
his most recent follow-up published in 1997. The most recent study of
the Painesville plant was published by Luippold et al. (Ex. 31-18-4).
The studies by Gibb and Luippold present historical exposure data for
the time periods covered by their respective studies. The Gibb exposure
data are especially interesting since the industrial hygiene data were
collected on a routine basis and not for compliance purposes. These
routine air measurements may be more representative of those typically
encountered by the exposed workers. In Great Britain, three plants have been
studied repeatedly, with reports published between 1952 and 1991. Other
studies of cohorts in the United States, Germany, Italy and Japan are
also reported. The elevated lung cancer mortality reported in the great
majority of these cohorts and the significant upward trends with
duration of employment and cumulative exposure provide some of the
strongest evidence that Cr(VI) is carcinogenic to workers. A summary of
selected human epidemiologic studies in chromate production workers is
presented in Table V-1.
BILLING CODE 4510-26-P


BILLING CODE 4510-26-C
The basic hexavalent chromate production process involves milling
and mixing trivalent chromite ore with soda ash, sometimes in the
presence of lime (Exs. 7-103; 35-61). The mixture is 'roasted' at a
high temperature, which oxidizes much of the chromite to hexavalent
sodium chromate. Depending on the lime content used in the process, the
roast also contains other chromate species, especially calcium
chromate under high lime conditions. The highly water-soluble sodium
chromate is water-extracted from the water-insoluble trivalent chromite
and the less water-soluble chromates (e.g., calcium chromate) in the
'leaching' process. The sodium chromate leachate is reacted with
sulfuric acid and sodium bisulfate to form sodium dichromate. The
sodium dichromate is prepared and packaged as a crystalline powder to
be sold as final product or sometimes used as the starting material to
make other chromates such as chromic acid and potassium dichromate.
a. Cohort Studies of the Baltimore Facility. The Hayes et al. study of
the Baltimore, Maryland chromate production plant was designed to
determine whether changes in the industrial process at one chromium
chemical production facility were associated with a decreased risk of
cancer, particularly cancer of the respiratory system (Ex. 7-14). Four
thousand two hundred and seventeen (4,217) employees were identified as
newly employed between January 1, 1945 and December 31, 1974. Excluded
from this initial enumeration were employees who: (1) were working as
of 1945, but had been hired prior to 1945 and (2) had been hired since
1945 but who had previously been employed at the plant. Excluded from
the final cohort were those employed less than 90 days; women; those
with unknown length of employment; those with no work history; and
those of unknown age. The final cohort included 2,101 employees (1,803
hourly and 298 salaried).
Hayes divided the production process into three departments: (1)
The mill and roast or "dry end" department which consists of
grinding, roasting and leaching processes; (2) the bichromate
department which consists of the acidification and crystallization
processes; and (3) the special products department which produces
secondary products including chromic acid. The bichromate and special
products departments are referred to as the "wet end".
The construction of a new mill and roast and bichromate plant that
opened during 1950 and 1951 and a new chromic acid and special products
plant that opened in 1960 were cited by Hayes as "notable production
changes" (Ex. 7-14). The new facilities were designed to "obtain
improvements in process technique and in environmental control of
exposure to chromium bearing dusts * * *" (Ex. 7-14).
Plant-related work and health histories were abstracted for each
employee from plant records. Each job on the employee's work history
was characterized according to whether the job exposure occurred in (1)
a newly constructed facility, (2) an old facility, or (3) could not be
classified as having occurred in the new or the old facility. Those who
ever worked in an old facility or whose work location(s) could not be
distinguished based upon job title were considered as having a high or
questionable exposure. Only those who worked exclusively in the new
facility were defined for study purposes as "low exposure". Data on
cigarette smoking were abstracted from plant records, but were not
utilized in any analyses since the investigators thought them "not to
be of sufficient quality to allow analysis."
One thousand one hundred and sixty nine (1,169) cohort members were
identified as alive, 494 not individually identified as alive and 438
as deceased. Death certificates could not be located for 35 reported
decedents. Deaths were coded to the 8th revision of the International
Classification of Diseases.
Mortality analysis was limited to the 1,803 hourly employees
calculating the standardized mortality ratios (SMRs) for specific
causes of death. The SMR is a ratio of the number of deaths observed in
the study population to the number that would be expected if that study
population had the same specific mortality rate as a standard reference
population (e.g., age-, gender-, calendar year adjusted U.S.
population). The SMR is typically multiplied by 100, so a SMR greater
than 100 represents an elevated mortality in the study cohort relative
to the reference group. In the Hayes study, the expected number of
deaths was based upon Baltimore, Maryland male mortality rates
standardized for age, race and time period. For those where race was
unknown, the expected numbers were derived from mortality rates for
whites. Cancer of the trachea, bronchus and lung accounted for 69% of
the 86 cancer deaths identified and was statistically significantly
elevated (O=59; E=29.16; SMR=202; 95% CI: 155-263).
Analysis of lung cancer deaths among hourly workers by year of
initial employment (1945-1949; 1950-1959 and 1960-1974), exposure
category (low exposure or questionable/high exposure) and duration of
employment (short term defined as 90 days-2 years; long term defined as
3 years +) was also conducted. For those workers characterized as
having questionable/high exposure, the SMRs were significantly elevated
for the 1945-1949 and the 1950-1959 hire periods and for both short-
and long-term workers (not statistically significant for the short-term
workers initially hired 1945-1949). For those characterized as low
exposure, there was an elevated SMR for the long-term workers hired
between 1950 and 1959, but based only on three deaths (not
statistically significant). No lung cancer cases were observed for
workers hired 1960-1974.
Case-control analyses of (1) a history of ever having been employed
in selected jobs or combinations of jobs or (2) a history of specified
morbid conditions and combinations of conditions reported on plant
medical records were conducted. Cases were defined as decedents (both
hourly and salaried were included in the analyses) whose underlying or
contributing cause of death was lung cancer. Controls were defined as
deaths from causes other than malignant or benign tumors. Cases and
controls were matched on race (white/non-white), year of initial
employment (+/-3 years), age at time of initial employment (+/-5 years)
and total duration of employment (90 days-2 years; 3-4 years and 5
years +). An odds ratio (OR) was determined where the ratio is the odds
of employment in a job involving Cr(VI) exposure for the cases relative
to the controls.
Based upon matched pairs, analysis by job position showed
significantly elevated odds ratios for special products (OR=2.6) and
bichromate and special products (OR=3.3). The relative risk for
bichromate alone was also elevated (OR=2.1, not statistically
significant).
The possible association of lung cancer and three health conditions
(skin ulcers, nasal perforation and dermatitis) as recorded in the
plant medical records was also assessed. Of the three medical
conditions, only the odds ratio for dermatitis was statistically
significant (OR=3.0). When various combinations of the three conditions
were examined, the odds ratio for having all three conditions was
statistically significantly elevated (OR=6.0).
Braver et al. used data from the Hayes study discussed above and
the results of 555 air samples taken during the period 1945-1950 by the
Baltimore City Health Department, the U.S. Public Health Service, and
the companies that owned the plant, in an attempt to examine the
relationship between exposure to Cr(VI) and the occurrence of lung
cancer (Ex. 7-17). According to the authors, methods for determining
the air concentrations of Cr(VI) have changed since the industrial
hygiene data were collected at the Baltimore plant between 1945 and
1959. The authors asked the National Institute for Occupational Safety
and Health (NIOSH) and the Occupational Safety and Health
Administration (OSHA) to review the available documents on the methods
of collecting air samples, stability of Cr(VI) in the sampling media
after collection and the methods of analyzing Cr(VI) that were used to
collect the samples during that period.
Air samples were collected by both midget impingers and high volume
samplers. According to the NIOSH/OSHA review, high volume samplers
could have led to a "significant" loss of Cr(VI) due to the reduction
of Cr(VI) to Cr(III) by glass or cellulose ester filters, acid
extraction of the chromate from the filter, or improper storage of
samples. The midget impinger was "less subject" to loss of Cr(VI)
according to the panel since neither filters nor acid extraction from
filters was employed. However, if iron was present or if the samples
were stored for too long, conversion from Cr(VI) to Cr(III) may have
occurred. The midget impinger can only detect water soluble Cr(VI). The
authors noted that, according to a 1949 industrial hygiene survey by
the U.S. Public Health Service, very little water insoluble Cr(VI) was
found at the Baltimore plant. One NIOSH/OSHA panel member characterized
midget impinger results as "reproducible" and "accuracy * * * fairly
solid unless substantial reducing agents (e.g., iron) are present"
(Ex. 7-17, p. 370). Based upon the panel's recommendations, the authors
used the midget impinger results to develop their exposure estimates
even though the panel concluded that the midget impinger methods "tend
toward underestimation" of Cr(VI).
The authors also cite other factors related to the industrial
hygiene data that could have potentially influenced the accuracy of
their exposure estimates (either overestimating or underestimating the
exposure). These include: Measurements may have been taken primarily in
"problem" areas of the plant; the plants may have been cleaned or
certain processes shut down prior to industrial hygiene monitoring by
outside groups; respirator use; and periodic high exposures (due to
infrequent maintenance operations or failure of exposure control
equipment) which were not measured and therefore not reflected in the
available data.
The authors estimated exposure indices for cohorts rather than for
specific individuals using hire period (1945-1949 or 1950-1959) and
duration of exposure, defined as short (at least 90 days but less than
three years) and long (three years or more). The usual exposure to
Cr(VI) for both the short- and long-term workers hired 1945-1949 was
calculated as the average of the mean annual air concentration for
1945-1947 and 1949 (data were missing for 1948). This was estimated to
be 413 [mu]g/m3. The usual exposure to Cr(VI) was estimated
to be 218 [mu]g/m3 for the short and long employees hired
between 1950 and 1959 based on air measurements in the older facility
in the early 1950s.
Cumulative exposure was calculated as the usual exposure level
times average duration. Short-term workers, regardless of length of
employment, were assumed to have received 1.6 years of exposure
regardless of hire period. For long-term workers, the average length of
exposure was 12.3 years. Those hired 1945-1949 were assigned five years
at an exposure of 413 [mu]g/m3 and 7.3 years at an exposure
of 218 [mu]g/m3. For the long-term workers hired between
1950 and 1959, the average length of exposure was estimated to be 13.4
years. The authors estimated that the cumulative exposures at which
"significant increases in lung cancer mortality" were observed in the
Hayes study were 0.35, 0.67, 2.93 and 3.65 mg/m3--years. The
association seen by the authors appears more likely to be the result of
duration of employment rather than the magnitude of exposure since the
variation in the latter was small.
Gibb et al. relied upon the Hayes study to investigate mortality in
a second cohort of the Baltimore plant (Ex. 31-22-11). The Hayes cohort
was composed of 1,803 hourly and 298 salaried workers newly employed
between January 1, 1945 and December 31, 1974. Gibb excluded 734
workers who began work prior to August 1, 1950 and included 990 workers
employed after August 1, 1950 who worked less than 90 days, resulting
in a cohort of 2,357 males followed for the period August 1, 1950
through December 31, 1992. Fifty-one percent (1,205) of the cohort was
white; 36% (848) nonwhite. Race was unknown for 13% (304) of the
cohort. The plant closed in 1985.
Deaths were coded according to the 8th revision of the
International Classification of Diseases. Person years of observation
were calculated from the beginning of employment until death or
December 31, 1992, whichever came earlier. Smoking data (yes/no) were
available for 2,137 (93.3%) of the cohort from company records.
Between 1950 and 1985, approximately 70,000 measurements of
airborne Cr(VI) were collected utilizing several different sampling
methods. The program of routine air sampling for Cr(VI) was initiated
to "characterize 'typical/usual exposures' of workers" (Ex. 31-22-11,
p. 117). Area samples were collected during the earlier time periods,
while both area and personal samples were collected starting in 1977.
Exposure estimates were derived from the area sampling systems and were
adjusted to "an equivalent personal exposure estimate using job-
specific ratios of the mean area and personal sampling exposure
estimates for the period 1978-1985 * * *" (Ex. 31-22-11, p. 117).
According to the author, comparison of the area and personal samples
showed "no significant differences" for about two-thirds of the job
titles. For several job titles with a "significant point source of
contamination" the area sampling methods "significantly
underestimated" personal exposure estimates and were adjusted "by the
ratio of the two" (Ex. 31-22-11, p. 118).
A job exposure matrix (JEM) was constructed, where air sampling
data were available, containing annual average exposure for each job
title. Data could not be located for the periods 1950-1956 and 1960-
1961. Exposures were modeled for the missing data using the ratio of
the measured exposure for a job title to the average of all measured
job titles in the same department. For the time periods where
"extensive" data were missing, a simple straight line interpolation
between years with known exposures was employed.
To estimate airborne Cr(III) concentrations, 72 composite dust
samples were collected at or near the fixed site air monitoring
stations about three years after the facility closed. The dust samples
were analyzed for Cr(VI) content using ion chromatography. Cr(III)
content was determined through inductively coupled plasma spectroscopic
analysis of the residue. The Cr(III):Cr(VI) ratio was calculated for
each area corresponding to the air sampling zones and the measured
Cr(VI) air concentration adjusted based on this ratio. Worker exposures
were calculated for each job title and weighted by the fraction of time
spent in each air-monitoring zone. The Cr(III):Cr(VI) ratio was derived
in this manner for each job title based on the distribution of time
spent in exposure zones in 1978. Cr(VI) exposures in the JEM were
multiplied by this ratio to estimate Cr(III) exposures.
Information on smoking was collected at the time of hire for
approximately 90% of the cohort. Of the 122 lung cancer cases, 116 were
smokers and four were non smokers at the time of hire. Smoking status
was unknown for two lung cancer cases. As discussed below, these data
were used by the study authors to adjust for smoking in their
proportional hazards regression models used to determine whether lung
cancer mortality in the worker cohort increased with increasing cumulative
Cr(VI) exposure.
A total of 855 observed deaths (472 white; 323 nonwhite and 60 race
unknown) were reported. SMRs were calculated using U.S. rates for
overall mortality. Maryland rates (the state in which the plant was
located) were used to analyze lung cancer mortality in order to better
account for regional differences in disease fatality. SMRs were not
adjusted for smoking. In the public hearing, Dr. Gibb explained that it
was more appropriate to adjust for smoking in the proportional hazards
models than in the SMRs, because the analyst must make more assumptions
to adjust the SMRs for smoking than to adjust the regression model (Tr.
124).
A statistically significant lung cancer SMR, based on the national
rate, was found for whites (O=71; SMR=186; 95% CI: 145-234); nonwhites
(O=47; SMR=188; 95% CI: 138-251) and the total cohort (O=122; SMR=180;
95% CI: 149-214). The ratio of observed to expected lung cancer deaths
(O/E) for the entire cohort stratified by race and cumulative exposure
quartile were computed. Cumulative exposure was lagged five years (only
exposure occurring five years before a given age was counted). The cut
point for the quartiles divided the cohort into four equal groups based
upon their cumulative exposure at the end of their working history (0-
0.00149 mgCrO\3\/m3-yr; 0.0015-0.0089 mgCrO3/m\3\-yr; 0.009-
0.0769 mgCrO3/m\3\-yr; and 0.077-5.25 mgCrO3/
m\3\-yr). For whites, the relative risk of lung cancer was
significantly elevated for the second through fourth exposure quartiles
with O/E values of 0.8, 2.1, 2.1 and 1.7 for the four quartiles,
respectively. For nonwhites, the O/E values by exposure quartiles were
1.1, 0.9, 1.2 and 2.9, respectively. Only the highest exposure quartile
was significantly elevated. For the total cohort, a significant
exposure-response trend was observed such that lung cancer mortality
increased with increasing cumulative Cr(VI) exposure.
Proportional hazards models were used to assess the relationship
between chromium exposure and the risk of lung cancer. The lowest
exposure quartile was used as the reference group. The median exposure
in each quartile was used as the measure of cumulative Cr(VI) exposure.
When smoking status was included in the model, relative lung cancer
risks of 1.83, 2.48 and 3.32 for the second, third and fourth exposure
quartiles respectively were estimated. Smoking, Cr(III) exposure, and
work duration were also significant predictors of lung cancer risk in
the model.
The analysis attempted to separate the effects into two
multivariate proportionate hazards models (one model incorporated the
log of cumulative Cr(VI) exposure, the log of cumulative Cr(III)
exposure and smoking; the second incorporated the log of cumulative
Cr(VI), work duration and smoking). In either regression model, lung
cancer mortality remained significantly associated (p < .05) with
cumulative Cr(VI) exposure even after controlling for the combination
of smoking and Cr(III) exposure or the combination of smoking and work
duration. On the other hand, lung cancer mortality was not
significantly associated with cumulative Cr(III) or work duration in
the multivariate analysis indicating lung cancer risk was more strongly
correlated with cumulative Cr(VI) exposure than the other variables.
Exponent, as part of a larger submission from the Chrome Coalition,
submitted comments on the Gibb paper prior to the publication of the
proposed rule. These comments asked that OSHA review methodological
issues believed by Exponent to impact upon the usefulness of the Gibb
data in a risk assessment analysis. While Exponent states that the Gibb
study offers data that "are substantially better for cancer risk than
the Mancuso study * * * they believe that further scrutiny of some of
the methods and analytical procedures is necessary (Ex. 31-18-15-1, p.
5).
The issues raised by Exponent and the Chrome Coalition (Ex. 31-18-
14) concerning the Gibb paper are: selection of the appropriate
reference population for compilation of expected numbers for use in the
SMR analysis; inclusion of short term workers (< 1 year); expansion of
the number of exposure groupings to evaluate dose response trends;
analyzing dose response by peak JEM exposure levels; analyzing dose-
response at exposures above and below the current PEL and calculating
smoking-adjusted SMRs for use in dose-response assessments. Exponent
obtained the original data from the Gibb study. The data were
reanalyzed to address the issues cited above. Exponent's findings are
presented in Exhibit 31-18-15-1 and are discussed below.
Exponent suggested that Gibb's use of U.S. and Maryland mortality
rates for developing expectations for the SMR analysis was
inappropriate. It suggested that Baltimore city mortality rates would
have been the appropriate standard to select since those mortality
rates would more accurately reflect the mortality experience of those
who worked at the plant. Exponent reran the SMR analysis to compare the
SMR values reported by Gibb (U.S. mortality rates for SMR analysis)
with the results of an SMR analysis using Maryland mortality rates and
Baltimore mortality rates. Gibb reported a lung cancer SMR of 1.86 (95%
CI: 1.45-2.34) for white males based upon 71 lung cancer deaths using
U.S. mortality rates. Reanalysis of the data produced a lung cancer SMR
of 1.85 (95% CI: 1.44-2.33) for white males based on U.S. mortality
rates, roughly the same value obtained by Gibb. When Maryland and
Baltimore rates are used, the SMR drops to 1.70 and 1.25 respectively.
Exponent suggested conducting sensitivity analysis that excludes
short-term workers (defined as those with one year of employment) since
the epidemiologic literature suggests that the mortality of short-term
workers is different than long-term workers. Short-term workers in the
Gibb study comprise 65% of the cohort and 54% of the lung cancers. The
Coalition also suggested that data pertaining to short-term employees'
information are of "questionable usefulness for assessing the
increased cancer risk from chronic occupational exposure to Cr(VI)"
(Ex. 31-18-15-1, p. 5).
Lung cancer SMRs were calculated for those who worked for less than
one year and for those who worked one year or more. Exponent defined
short-term workers as those who worked less than one year "because it
is consistent with the inclusion criteria used by others studying
chromate chemical production worker cohorts" (Ex. 31-18-15-1, p. 12).
Exponent also suggested that Gibb's breakdown of exposure by quartile
was not the most "appropriate" way of assessing dose-response since
cumulative Cr(VI) exposures remained near zero until the 50th to 60th
percentile, "so there was no real distinction between the first two
quartiles * * * (Ex. 31-18-15-1, p. 24). They also suggested that
combining "all workers together at the 75th quartile * * * does not
properly account for the heterogeneity of exposure in this group" (Ex.
31-18-15-1, p. 24). The Exponent reanalysis used six cumulative
exposure levels of Cr(VI) compared with the four cumulative exposure
levels of Cr(VI) in the Gibb analysis. The lower levels of exposure
were combined and "more homogeneous" categories were developed for
the higher exposure levels.
Using these re-groupings and excluding workers with less than one
year of employment, Exponent reported that the highest SMRs are seen in
the highest exposure group (1.5-< 5.25 mg CrO3/m\3\-years) for both white
and nonwhite, based on either the Maryland or the Baltimore mortality rates.
The authors did not find "that the inclusion of short-term workers had a
significant impact on the results, especially if Baltimore rates are used
in the SMR calculations' (Ex. 31-18-15-1, p. 28).
Analysis of length of employment and "peak" (i.e., highest
recorded mean annual) exposure level to Cr(VI) was conducted. Exponent
reported that approximately 50% of the cohort had "only very low"
peak exposure levels (<7.2 [mu]g CrO3/m\3\ or approximately
3.6 [mu]g/m\3\ of Cr(VI)). The majority of the short-term workers had
peak exposures of <100 [mu]g CrO3/m\3\. There were five peak
Cr(VI) exposure levels (<7.2 [mu]g CrO3/m\3\; 7.2-<19.3
[mu]g CrO3/m\3\; 19.3-<48.0 [mu]g CrO3/m\3\;
48.0-<105 [mu]g CrO3/m\3\; 105-<182 [mu]g CrO3/
m\3\; and 182-<806 [mu]g CrO3/m\3\) included in the
analyses. Overall, the lung cancer SMRs for the entire cohort grouped
according to the six peak exposure categories were slightly higher
using Maryland reference rates compared to Baltimore reference rates.
The Exponent analysis of workers who were ever exposed above the
current PEL versus those never exposed above the current PEL produced
slightly higher SMRs for those ever exposed, with the SMRs higher using
the Maryland standard rather than the Baltimore standard. The only
statistically significant result was for all lung cancer deaths
combined.
Assessment was made of the potential impact of smoking on the lung
cancer SMRs since Gibb did not adjust the SMRs for smoking. Exponent
stated that the smoking-adjusted SMRs are more appropriate for use in
the risk assessment than the unadjusted SMRs. It should be noted that
smoking adjusted SMRs could not be calculated using Baltimore reference
rates. As noted by the authors, the smoking adjusted SMRs produced
using Maryland reference rates are, by exposure, "reasonably
consistent with the Baltimore-referenced SMRs" (Ex. 31-18-15-1, p.
41).
Gibb et al. included workers regardless of duration of employment,
and the cohort was heavily weighted by those individuals who worked
less than 90 days. In an attempt to clarify this issue, Exponent
produced analyses of short-term workers, particularly with respect to
exposures. Exponent redefined short-term workers as those who worked
less than one year, to be consistent with the definition used in other
studies of chromate producers. OSHA finds this reanalysis excluding
short-term workers to be useful. It suggests that including cohort
workers employed less than one year did not substantively alter the
conclusions of Gibb et al. with regard to the association between
Cr(VI) exposure and lung cancer mortality. It should be noted that in
the Hayes study of the Baltimore plant, the cohort is defined as anyone
who worked 90 days or more.
Hayes et al. used Baltimore mortality rates while Gibb et al. used
U.S. mortality rates to calculate expectations for overall SMRs. To
calculate expectations for the analysis of lung cancer mortality and
exposure, Gibb et al. used Maryland state mortality rates. The SMR
analyses provided by Exponent using both Maryland and Baltimore rates
are useful. The data showed that using Baltimore rates raised the
expected number of lung cancer deaths and, thus, lowered the SMRs.
However, there remained a statistically significant increase in lung
cancer risk among the exposed workers and a significant upward trend
with cumulative Cr(VI) exposure. The comparison group should be as
similar as possible with respect to all other factors that may be
related to the disease except the determinant under study. Since the
largest portion of the cohort (45%) died in the city of Baltimore, and
even those whose deaths occurred outside of Baltimore (16%) most likely
lived in proximity to the city, the use of Baltimore mortality rates as
an external reference population is preferable.
Gibb's selection of the cut points for the exposure quartiles was
accomplished by dividing the workers in the cohort into four equal
groups based on their cumulative exposure at the end of their working
history. Using the same method but excluding the short-term workers
would have resulted in slightly different cumulative exposure
quartiles. Exponent expressed a preference for a six-tiered exposure
grouping. The impact of using different exposure groupings is further
discussed in section VI.C of the quantitative risk assessment.
The exposure matrix of Gibb et al. utilizes an unusually high-
quality set of industrial hygiene data. Over 70,000 samples taken to
characterize the "typical/usual" working environment is more
extensive industrial hygiene data then is commonly available for most
exposure assessments. However, there are several unresolved issues
regarding the exposure assessment, including the impact of the
different industrial hygiene sampling techniques used over the sampling
time frame, how the use of different sampling techniques was taken into
account in developing the exposure assessment and the use of area vs.
personal samples.
Exponent and the Chrome Coalition also suggested that the SMRs
should have been adjusted for smoking. According to Exponent, smoking
adjusted SMRs based upon the Maryland mortality rates produced SMRs
similar to the SMRs obtained using Baltimore mortality rates (Ex. 31-
18-15-1). The accuracy of the smoking data is questionable since it
represents information obtained at the time of hire. Hayes abstracted
the smoking data from the plant medical records, but "found it not to
be of sufficient quality to allow analysis." One advantage to using
the Baltimore mortality data may be to better control for the potential
confounding of smoking.
The Gibb study is one of the better cohort mortality studies of
workers in the chromium production industry. The quality of the
available industrial hygiene data and its characterization as
"typical/usual" makes the Gibb study particularly useful for risk
assessment.
b. Cohort Studies of the Painesville Facility. The Ohio Department of
Health conducted epidemiological and environmental studies at a plant
in Painesville that manufactured sodium bichromate from chromite ore.
Mancuso and Hueper (Ex. 7-12) reported an excess of respiratory cancer
among chromate workers when compared to the county in which the plant
was located. Among the 33 deaths in males who had worked at the plant
for a minimum of one year, 18.2% were from respiratory cancer. In
contrast, the expected frequency of respiratory cancer among males in
the county in which the plant was located was 1.2%. Although the
authors did not include a formal statistical comparison, the lung
cancer mortality rate among the exposed workers would be significantly
greater than the county rate.
Mancuso (Ex. 7-11) updated his 1951 study of 332 chromate
production workers employed during the period 1931-1937. Age adjusted
mortality rates were calculated by the direct method using the
distribution of person years by age group for the total chromate
population as the standard. Vital status follow-up through 1974 found
173 deaths. Of the 66 cancer deaths, 41 (62.1%) were lung cancers. A
cluster of lung cancer deaths was observed in workers with 27-36 years
since first employment.
Mancuso used industrial hygiene data collected in 1949 to calculate
weighted average exposures to water-soluble (presumed to be Cr(VI)),
insoluble (presumed to be principally Cr(III)) and
total chromium (Ex. 7-98). The age-adjusted lung cancer death rate
increased from 144.6 (based upon two deaths) to 649.6 (based upon 14
deaths) per 100,000 in five exposure categories ranging from a low of
0.25-0.49 to a high of 4.0+ mg/m\3\-years for the insoluble Cr(III)
exposures. For exposure to soluble Cr(VI), the age adjusted lung cancer
rates ranged from 80.2 (based upon three deaths) to 998.7 (based upon
12 deaths) in five exposure categories ranging from < 0.25 to 2.0+ mg/
m\3\-years. For total chromium, the age-adjusted death rates ranged
from 225.7 (based upon three deaths) to 741.5 (based upon 16 deaths)
for exposures ranging from 0.50-0.99 mg/m\3\-years to 6.0+ mg/m\3\-
years.
Age-adjusted lung cancer death rates also were calculated by
classifying workers by the levels of insoluble Cr(III) and total
chromium exposure. From the data presented, it appears that for a fixed
level of insoluble Cr(III), the lung cancer risk appears to increase as
the total chromium increases (Ex. 7-11).
Mancuso (Ex. 23) updated the 1975 study. As of December 31, 1993,
283 (85%) cohort members had died and 49 could not be found. Of the 102
cancer deaths, 66 were lung cancers. The age-adjusted lung cancer death
rate per 100,000 ranged from 187.9 (based upon four deaths) to 1,254.1
(based upon 15 deaths) for insoluble Cr(III) exposure categories
ranging from 0.25-0.49 to 4.00-5.00 mg/m\3\ years. For the highest
exposure to insoluble Cr(III) (6.00+ mg/m\3\ years) the age-adjusted
lung cancer death rate per 100,000 fell slightly to 1,045.5 based upon
seven deaths.
The age-adjusted lung cancer death rate per 100,000 ranged from
99.7 (based upon five deaths) to 2,848.3 (based upon two deaths) for
soluble Cr(VI) exposure categories ranging from < 0.25 to 4.00+ mg/m\3\
years. For total chromium, the age-adjusted lung cancer death rate per
100,000 ranged from 64.7 (based upon two deaths) to 1,106.7 (based upon
21 deaths) for exposure categories ranging from < 0.50 to 6.00+ mg/m\3\
years.
To investigate whether the increase in the lung cancer death rate
was due to one form of chromium compound (presumed insoluble Cr(III) or
soluble Cr(VI)), age-adjusted lung cancer mortality rates were
calculated by classifying workers by the levels of exposure to
insoluble Cr(III) and total chromium. For a fixed level of insoluble
Cr(III), the lung cancer rate appears to increase as the total chromium
increases for each of the six total chromium exposure categories,
except for the 1.00-1.99 mg/m\3\-years category. For the fixed exposure
categories for total chromium, increasing exposures to levels of
insoluble Cr(III) showed an increased age-adjusted death rate from lung
cancer in three of the six total chromium exposure categories.
For a fixed level of soluble Cr(VI), the lung cancer death rate
increased as total chromium categories of exposure increased for three
of the six gradients of soluble Cr(VI). For the fixed exposure
categories of total chromium, the increasing exposure to specific
levels of soluble Cr(VI) led to an increase in two of the six total
chromium exposure categories. Mancuso concluded that the relationship
of lung cancer is not confined solely to either soluble or insoluble
chromium. Unfortunately, it is difficult to attribute these findings
specifically to Cr(III) [as insoluble chromium] and Cr(VI) [as soluble
chromium] since it is likely that some slightly soluble and insoluble
Cr(VI) as well as Cr(III) contributed to the insoluble chromium
measurement.
Luippold et al. conducted a retrospective cohort study of 493
former employees of the chromate production plant in Painesville, Ohio
(Ex. 31-18-4). This Painesville cohort does not overlap with the
Mancuso cohort and is defined as employees hired beginning in 1940 who
worked for a minimum of one year at Painesville and did not work at any
other facility owned by the same company that used or produced Cr(VI).
An exception to the last criterion was the inclusion of workers who
subsequently were employed at a company plant in North Carolina (number
not provided). Four cohort members were identified as female. The
cohort was followed for the period January 1, 1941 through December 31,
1997. Thirty-two percent of the cohort worked for 10 or more years.
Information on potential confounders was limited. Smoking status
(yes/no) was available for only 35% of the cohort from surveys
administered between 1960 and 1965 or from employee medical files. For
those employees where smoking data were available, 78% were smokers
(responded yes on at least one survey or were identified as smokers
from the medical file). Information on race also was limited, the death
certificate being the primary source of information.
Results of the vital status follow-up were: 303 deaths; 132
presumed alive and 47 vital status unknown. Deaths were coded to the
9th revision of the International Classification of Diseases. Cause of
death could not be located for two decedents. For five decedents the
cause of death was only available from data collected by Mancuso and
was recoded from the 7th to the 9th revision of the ICD. There were no
lung cancer deaths among the five recoded deaths.
SMRs were calculated based upon two reference populations: The U.S.
(white males) and the state of Ohio (white males). Lung cancer SMRs
stratified by year of hire, duration of exposure, time since first
employment and cumulative exposure group also were calculated.
Proctor et al. analyzed airborne Cr(VI) levels throughout the
facility for the years 1943 to 1971 (the plant closed April 1972) from
800 area air sampling measurements from 21 industrial hygiene surveys
(Ex. 35-61). A job exposure matrix (JEM) was constructed for 22
exposure areas for each month of plant operation. Gaps in the matrix
were completed by computing the arithmetic mean concentration from area
sampling data, averaged by exposure area over three time periods (1940-
1949; 1950-1959 and 1960-1971) which coincided with process changes at
the plant (Ex. 31-18-1)
The production of water-soluble sodium chromate was the primary
operation at the Painesville plant. It involved a high lime roasting
process that produced a water insoluble Cr(VI) residue (calcium
chromate) as byproduct that was transported in open conveyors and
likely contributed to worker exposure until the conveyors were covered
during plant renovations in 1949. The average airborne soluble Cr(VI)
from industrial hygiene surveys in 1943 and 1948 was 0.72 mg/m\3\ with
considerable variability among departments. During these surveys, the
authors believe the reported levels may have underestimated total
Cr(VI) exposure by 20 percent or less for some workers due to the
presence of insoluble Cr(VI) dust.
Reductions in Cr(VI) levels over time coincided with improvements
in the chromate production process. Industrial hygiene surveys over the
period from 1957 to 1964 revealed average Cr(VI) levels of 270 [mu]g/
m\3\. Another series of plant renovations in the early 1960s lowered
average Cr(VI) levels to 39 [mu]g/m\3\ over the period from 1965 to
1972. The highest Cr(VI) concentrations generally occurred in the
shipping, lime and ash, and filtering operations while the locker
rooms, laboratory, maintenance shop and outdoor raw liquor storage
areas had the lowest Cr(VI) levels.
The average cumulative Cr(VI) exposure (mg/m\3\-yrs) for the cohort
was 1.58 mg/m\3\-yrs and ranged from 0.006 to 27.8 mg/m\3\-yrs. For
those who died from lung cancer, the average Cr(VI) exposure was 3.28
mg/m\3\-yrs and ranged from 0.06 to 27.8 mg/m\3\-yrs.
According to the authors, 60% of the cohort accumulated an estimated
Cr(VI) exposure of 1.00 mg/m\3\-yrs or less.
Sixty-three per cent of the study cohort was reported as deceased
at the end of the follow-up period (December 31, 1997). There was a
statistically significant increase for the all causes of death category
based on both the national and Ohio state standard mortality rates
(national: O=303; E=225.6; SMR=134; 95% CI: 120-150; state: O=303;
E=235; SMR=129; 95% CI: 115-144). Fifty-three of the 90 cancer deaths
were cancers of the respiratory system with 51 coded as lung cancer.
The SMR for lung cancer is statistically significant using both
reference populations (national O= 51; E=19; SMR 268; 95% CI: 200-352;
state O=51; E=21.2; SMR 241; 95% CI: 180-317).
SMRs also were calculated by year of hire, duration of employment,
time since first employment and cumulative Cr(VI) exposure, mg/m\3\-
years. The highest lung cancer SMRs were for those hired during the
earliest time periods. For the period 1940-1949, the lung cancer SMR
was 326 (O=30; E=9.2; 95% CI: 220-465); for 1950-1959, the lung cancer
SMR was 275 (O=15; E=5.5; 95% CI: 154-454). For the period 1960-1971,
the lung cancer SMR was just under 100 based upon six deaths with 6.5
expected.
Lung cancer SMRs based upon duration of employment (years)
increased as duration of employment increased. For those with one to
four years of employment, the lung cancer SMR was 137 based upon nine
deaths (E=6.6; 95% CI: 62-260); for five to nine years of employment,
the lung cancer SMR was 160 (O=8; E=5.0; 95% CI: 69-314). For those
with 10-19 years of employment, the lung cancer SMR was 169 (O=7;
E=4.1; 95% CI: 68-349), and for those with 20 or more years of
employment, the lung cancer SMR was 497 (O=27; E=5.4; 95% CI: 328-723).
Analyses of cumulative Cr(VI) exposure found the lung cancer SMR
(based upon the Ohio standard) in the highest exposure group (2.70-
27.80 mg/m\3\-yrs) was 463 (O=20; E=4.3; 95% CI: 183-398). In the 1.05-
2.69 mg/m\3\-yrs cumulative exposure group, the lung cancer SMR was 365
based upon 16 deaths (E=4.4; 95% CI: 208-592). For the cumulative
exposure groups 0.49-1.04, 0.20-0.48 and 0.00-0.19, the lung cancer
SMRs were 91 (O=4; E=4.4; 95% CI: 25-234; 184 (O=8; E=4.4; 95% CI: 79-
362) and 67 (O=3; E=4.5; 95% CI: 14-196). A test for trend showed a
strong relationship between lung cancer mortality and cumulative Cr(VI)
exposure (p=0.00002). The authors claim that the SMRs are also
consistent with a threshold effect since there was no statistically
significant trend for excess lung cancer mortality with cumulative
Cr(VI) exposures less than about 1 mg/m\3\-yrs. The issue of whether
the cumulative Cr(VI) exposure-lung cancer response is best represented
by a threshold effect is discussed further in preamble section VI on
the quantitative risk assessment.
The Painesville cohort is small (482 employees). Excluded from the
cohort were six employees who worked at other chromate plants after
Painesville closed. However, exceptions were made for employees who
subsequently worked at the company's North Carolina plant (number not
provided) because exposure data were available from the North Carolina
plant. Subsequent exposure to Cr(VI) by other terminated employees is
unknown and not taken into account by the investigators. Therefore, the
extent of the bias introduced is unknown.
The 10% lost to follow-up (47 employees) in a cohort of this size
is striking. Four of the forty-seven had "substantial" follow-up that
ended in 1997 just before the end date of the study. For the remaining
43, most were lost in the 1950s and 1960s (most is not defined). Since
person-years are truncated at the time individuals are lost to follow
up, the potential implication of lost person years could impact the
width of the confidence intervals.
The authors used U.S. and Ohio mortality rates for the standards to
compute the expectations for the SMRs, stating that the use of Ohio
rates minimizes bias that could occur from regional differences in
mortality. It is unclear why county rates were not used to address the
differences in regional mortality.
c. Other Cohort Studies. The first study of cancer of the respiratory
system in the U.S. chromate producing industry was reported by Machle
and Gregorius (Ex. 7-2). The study involved a total of 11,000 person-
years of observation between 1933 and 1947. There were 193 deaths; 42
were due to cancer of the respiratory system. The proportion of
respiratory cancer deaths among chromate workers was compared with
proportions of respiratory cancer deaths among Metropolitan Life
Insurance industrial policyholders. A non-significant excess
respiratory cancer among chromate production workers was found. No
attempt was made to control for confounding factors (e.g., age). While
some exposure data are presented, the authors state that one cannot
associate tumor rates with tasks (and hence specific exposures) because
of "shifting of personnel" and the lack of work history records.
Baetjer reported the results of a case-control study based upon
records of two Baltimore hospitals (Ex. 7-7). A history of working with
chromates was determined from these hospital records and the proportion
of lung cancer cases determined to have been exposed to chromates was
compared with the proportion of controls exposed. Of the lung cancer
cases, 3.4% had worked in a chromate manufacturing plant, while none of
the controls had such a history recorded in the medical record. The
results were statistically significant and Baetjer concluded that the
data confirmed the conclusions reached by Machle and Gregorius that
"the number of deaths due to cancer of the lung and bronchi is greater
in the chromate-producing industry than would normally be expected"
(Ex. 7-7, p. 516).
As a part of a larger study carried out by the U.S. Public Health
Service, the morbidity and mortality of male workers in seven U.S.
chromate manufacturing plants during the period 1940-1950 was reported
(Exs. 7-1; 7-3). Nearly 29 times as many deaths from respiratory cancer
(excluding larynx) were found among workers in the chromate industry
when compared to mortality rates for the total U.S. for the period
1940-1948. The lung cancer risk was higher at the younger ages (a 40-
fold risk at ages 15-45; a 30-fold risk at ages 45-54 and a 20-fold
risk at ages 55-74). Analysis of respiratory cancer deaths (excluding
larynx) by race showed an observed to expected ratio of 14.29 for white
males and 80 for nonwhite males.
Taylor conducted a mortality study in a cohort of 1,212 chromate
workers followed over a 24 year (1937-1960) period (Ex. 7-5). The
workers were from three chromate plants that included approximately 70%
of the total population of U.S. chromate workers in 1937. In addition,
the plants had been in continuous operation for the study period
(January 1, 1937 to December 31, 1960). The cohort was followed
utilizing records of Old Age and Survivors Disability Insurance
(OASDI). Results were reported both in terms of SMRs and conditional
probabilities of survival to various ages comparing the mortality
experience of chromate workers to the U.S. civilian male population. No
measures of chromate exposure were reported although results are
provided in terms of duration of employment. Taylor concluded that not
only was there an excess in mortality from respiratory cancer, but from
other causes as well, especially as duration of employment increased.
In a reanalysis of Taylor's data, Enterline excluded those workers
born prior to 1889 and analyzed the data by follow-up period using U.S.
rates (Ex. 7-4). The SMR for respiratory cancer for all time periods
showed a nine-fold excess (O=69 deaths; E=7.3). Respiratory cancer
deaths comprised 28% of all deaths. Two of the respiratory cancer
deaths were malignant neoplasms of the maxillary sinuses, a number
according to Enterline, "greatly in excess of that expected based on
the experience of the U.S. male population." Also slightly elevated
were cancers of the digestive organs (O=16; E=10.4) and non-malignant
respiratory disease (O=13; E=8.9).
Pastides et al. conducted a cohort study of workers at a North
Carolina chromium chemical production facility (Ex. 7-93). Opened in
1971, this facility is the largest chromium chemical production
facility in the United States. A low-lime process was used since the
plant began operation. Three hundred and ninety eight workers employed
for a minimum of one year between September 4, 1971 and December 31,
1989 comprised the study cohort. A self-administered employee
questionnaire was used to collect data concerning medical history,
smoking, plant work history, previous employment and exposure to other
potential chemical hazards. Personal air monitoring results for Cr(VI)
were available from company records for the period February 1974
through April 1989 for 352 of the 398 cohort members. A job matrix
utilizing exposure area and calendar year was devised. The exposure
means from the matrix were linked to each employee's work history to
produce the individual exposure estimates by multiplying the mean
Cr(VI) value from the matrix by the duration (time) in a particular
exposure area (job). Annual values were summed to estimate total
cumulative exposure.
Personal air monitoring indicated that TWA Cr(VI) air
concentrations were generally very low. Roughly half the samples were
less than 1 [mu]g/m3, about 75 percent were below 3 [mu]g/
m3, and 96 percent were below 25 [mu]g/m3. The
average worker's age was 42 years and mean duration of employment was
9.5 years. Two thirds of the workers had accumulated less than 0.01
[mu]g/m3-yr cumulative Cr(VI) exposure. SMRs were computed
using National, State (not reported) and county mortality rates (eight
adjoining North Carolina counties, including the county in which the
plant is located). Two of the 17 recorded deaths in the cohort were
from lung cancers. The SMRs for lung cancer were 127 (95% CI: 22-398)
and 97 (95% CI: 17-306) based on U.S. and North Carolina county
mortality rates, respectively. The North Carolina cohort is still
relatively young and not enough time has elapsed to reach any
conclusions regarding lung cancer risk and Cr(VI) exposure.
In 2005, Luippold et al. published a study of mortality among two
cohorts of chromate production workers with low exposures (Ex. 47-24-
2). Luippold et al. studied a total of 617 workers with at least one
year of employment, including 430 at the North Carolina plant studied
by Pastides et al. (1994) ("Plant 1") and 187 hired after the 1980
institution of exposure-reducing process and work practice changes at a
second U.S. plant ("Plant 2"). A high-lime process was never used at
Plant 1, and workers drawn from Plant 2 were hired after the
institution of a low lime process, so that exposures to calcium
chromate in both cohorts were likely minimal. Personal air-monitoring
measures available from 1974 to 1988 for the first plant and from 1981
to 1998 for the second plant indicated that exposure levels at both
plants were low, with overall geometric mean concentrations below 1.5
[mu]g/m3 and area-specific average personal air sampling
values not exceeding 10 [mu]g/m3 for most years (Ex. 47-24-
2, p. 383).
Workers were followed through 1998. By the end of follow-up, which
lasted an average of 20.1 years for workers at Plant 1 and 10.1 years
at Plant 2, 27 cohort members (4%) were deceased. There was a 41%
deficit in all-cause mortality when compared to all-cause mortality
from age-specific state reference rates, suggesting a strong healthy
worker effect. Lung cancer was 16% lower than expected based on three
observed vs. 3.59 expected cases, also using age-specific state
reference rates (Ex. 47-24-2, p. 383). The authors stated that "[t]he
absence of an elevated lung cancer risk may be a favorable reflection
of the postchange environment", but cautioned that longer follow-up
allowing an appropriate latency for the entire cohort would be required
to confirm this conclusion (Ex. 47-24-2, p. 381). OSHA received several
written testimony regarding this cohort during the post-hearing comment
period. These are discussed in section VI.B.7 on the quantitative risk
assessment.
A study of four chromate producing facilities in New Jersey was
reported by Rosenman (Ex. 35-104). A total of 3,408 individuals were
identified from the four facilities over different time periods (plant
A from 1951-1954; plant B from 1951-1971; plant C from 1937-1964 and
plant D 1937-1954). No Cr(VI) exposure data was collected for this
study. Proportionate mortality ratios (PMRs) and proportionate cancer
mortality ratios (PCMRs), adjusted by race, age, and calendar year,
were calculated for the three companies (plants A and B are owned by
one company). Unlike SMRs, PMRs are not based on the expected mortality
rates in a standardized population but, instead, merely represent the
proportional distribution of deaths in the cohort relative to the
general U.S. population. Analyses were done evaluating duration of work
and latency from first employment.
Significantly elevated PMRs were seen for lung cancer among white
males (170 deaths, PMR=1.95; 95% CI: 1.67-2.27) and black males (54
deaths, PMR=1.88; 95% CI: 1.41-2.45). PMRs were also significantly
elevated (regardless of race) for those who worked 1-10, 11-20 and >20
years and consistently higher for white and black workers 11-20 years
and >20 years since first hire. The results were less consistent for
those with 10 or fewer years since first hire.
Bidstrup and Case reported the mortality experience of 723 workers
at three chromate producing factories in Great Britain (Ex. 7-20). Lung
cancer mortality was 3.6 times that expected (O=12; E=3.3) for England
and Wales. Alderson et al. conducted a follow-up of workers from the
three plants in the U.K. (Bolton, Rutherglen and Eaglescliffe)
originally studied by Bidstrup (Ex. 7-22). Until the late 1950s, all
three plants operated a "high-lime" process. This process potentially
produced significant quantities of calcium chromate as a by-product as
well as the intended sodium dichromate. Process changes occurred during
the 1940s and 1950s. The major change, according to the author, was the
introduction of the "no-lime" process, which eliminated unwanted
production of calcium chromate. The no-lime process was introduced at
Eaglescliffe 1957-1959 and by 1961 all production at the plant was by
this process. Rutherglen operated a low-lime process from 1957/1959
until it closed in 1967. Bolton never changed to the low lime process.
The plant closed in 1966. Subjects were eligible for entry into the
study if they had received an X-ray examination at work and had been
employed for a minimum of one year between 1948 and 1977. Of the 3,898
workers enumerated at the three plants, 2,715 met the cohort entrance
criteria, (alive: 1,999; deceased: 602; emigrated: 35; and lost to
follow-up: 79). Those lost to follow-up were not included in the
analyses. Eaglescliffe contributed the greatest number of subjects to
the study (1,418). Rutherglen contributed the largest number of total
deaths (369, or 61%). Lung cancer comprised the majority of cancer deaths
and was statistically significantly elevated for the entire cohort
(O=116; E=47.96; SMR= 240; p < 0.001). Two deaths from nasal cancer were
observed, both from Rutherglen.
SMRs were computed for Eaglescliffe by duration of employment,
which was defined based upon plant process updates (those who only
worked before the plant modification, those who worked both before and
after the modifications, or those who worked only after the
modifications were completed). Of the 179 deaths at the Eaglescliffe
plant, 40 are in the pre-change group; 129 in the pre-/post-change and
10 in the post-change. A total of 36 lung cancer deaths occurred at the
plant, in the pre-change group O=7; E=2.3; SMR=303; in the pre-/post-
change group O=27; E=13; SMR=2.03 and in the post-change group O=2;
E=1.07; SMR=187.
In an attempt to address several potential confounders, regression
analysis examined the contributions of various risk factors to lung
cancer. Duration of employment, duration of follow-up and working
before or after plant modification appear to be greater risk factors
for lung cancer, while age at entry or estimated degree of chromate
exposure had less influence.
Davies updated the work of Alderson, et al. concerning lung cancer
in the U.K. chromate producing industry (Ex. 7-99). The study cohort
included payroll employees who worked a minimum of one year during the
period January 1, 1950 and June 30, 1976 at any of the three facilities
(Bolton, Eaglescliffe or Rutherglen). Contract employees were excluded
unless they later joined the workforce, in which case their contract
work was taken into account.
Based upon the date of hire, the workers were assigned to one of
three groups. The first, or "early" group, consists of workers hired
prior to January 1945 who are considered long term workers, but do not
comprise a cohort since those who left or died prior to 1950 are
excluded. The second group, "pre-change" workers, were hired between
January 1, 1945 to December 31, 1958 at Rutherglen or to December 31,
1960 at Eaglescliffe. Bolton employees starting from 1945 are also
termed pre-change. The cohort of pre-change workers is considered
incomplete since those leaving 1946-1949 could not be included and
because of gaps in the later records. For those who started after 1953
and for all men staying 5+ years, this subcohort of pre-change workers
is considered complete. The third group, "post-change" workers,
started after the process changes at Eaglescliffe and Rutherglen became
fully effective and are considered a "complete" cohort. A "control"
group of workers from a nearby fertilizer facility, who never worked in
or near the chromate plant, was assembled.
A total of 2,607 employees met the cohort entrance criteria. As of
December 31, 1988, 1,477 were alive, 997 dead, 54 emigrated and 79
could not be traced (total lost to follow-up: 133). SMRs were
calculated using the mortality rates for England and Wales and the
mortality rates for Scotland. Causes of death were ascertained for all
but three decedents and deaths were coded to the revision of the
International Classification of Diseases in effect at the time of
death. Lung cancer in this study is defined as those deaths where the
underlying cause of death is coded as 162 (carcinoma of the lung) or
239.1 (lung neoplasms of unspecified nature) in the 9th revision of the
ICD. Two deaths fell into the latter category. The authors attempted to
adjust the national mortality rates to allow for differences based upon
area and social class.
There were 12 lung cancer deaths at Bolton, 117 at Rutherglen, 75
at Eaglescliffe and one among staff for a total of 205 lung cancer
deaths. A statistically significant excess of lung cancer deaths (175
deaths) among early and pre-change workers is seen at Rutherglen and
Eaglescliffe for both the adjusted and unadjusted SMRs. For Rutherglen,
for the early period based upon 68 observed deaths, the adjusted SMR
was 230 while the unadjusted SMR was 347 (for both SMRs p< 0.001). For
the 41 pre-change lung cancer deaths at Rutherglen, the adjusted SMR
was 160 while the unadjusted SMR was 242 (for both SMRs p< 0.001). At
Eaglescliffe, there were 14 lung cancer deaths in the early period
resulting in an adjusted SMR of 196 and an unadjusted SMR of 269 (for
both SMRs p< 0.05). For the pre-change period at Eaglescliffe, the
adjusted SMR was 195 and the unadjusted was 267 (p< 0.001 for both
SMRs). At Bolton there is a non-significant excess among pre-change
men. There are no apparent excesses in the post-change groups, the
staff groups or in the non-exposed fertilizer group.
There is a highly significant overall excess of nasal cancers with
two cases at Eaglescliffe and two cases at Rutherglen (O=4,
Eadjusted=0.26; SMR=1538). All four men with nasal cancer had more than
20 years of exposure to chromates.
Aw reported on two case-control studies conducted at the previously
studies Eaglescliffe plant (Ex. 245). In 1960, the plant, converted
from a "high-lime" to a "no-lime" process, reducing the likelihood
of calcium chromate formation. As of March 1996, 2,672 post-change
workers had been employed, including 891 office personnel. Of the post-
change plant personnel, 56% had been employed for more than one year.
Eighteen lung cancer cases were identified among white male post-change
workers (13 deceased; five alive). Duration of employment for the cases
ranged from 1.5 to 25 years with a mean of 14.4. Sixteen of the lung
cancer cases were smokers.
In the first case-control study reported, the 15 lung cancer cases
identified up to September 1991 were matched to controls by age and
hire date (five controls per case). Cases and controls were compared
based upon their job categories within the plant. The results showed
that cases were more likely to have worked in the kiln area than the
controls. Five of the 15 cases had five or more years in the kiln area
where Cr(VI) exposure occurred vs. six of the 75 controls. A second
case-control study utilized the 18 lung cancer cases identified in post
change workers up to March 1996. Five controls per case were matched by
age (+/-5 years), gender and hire date. Both cases and controls had a
minimum of one year of employment. A job exposure matrix was being
constructed that would allow the investigators to "estimate exposure
to hexavalent chromates for each worker in the study for all the jobs
done since the start of employment at the site until 1980." Starting
in 1970 industrial hygiene sampling was performed to determine exposure
for all jobs at the plant. Cr(VI) exposure levels for the period
between 1960 and 1969 were being estimated based on the recall of
employees regarding past working conditions relative to current
conditions from a questionnaire. The author stated that preliminary
analysis suggests that the maximum recorded or estimated level of
exposure to Cr(VI) for the cases was higher than that of the controls.
However, specific values for the estimated Cr(VI) exposures were not
reported.
Korallus et al. conducted a study of 1,140 active and retired
workers with a minimum of one year of employment between January 1,
1948 and March 31, 1979 at two German chromate production plants (Ex.
7-26). Workers employed prior to January 1, 1948 (either active or
retired) and still alive at that date were also included in the cohort.
The primary source for determining cause of death was medical
records. Death certificates were used only when medical records could
not be found. Expected deaths were calculated using the male population
of North Rhineland-Westphalia. Elevated SMRs for cancer of the
respiratory system (50 lung cancers and one laryngeal cancer) were seen
at both plants (O=21; E=10.9; SMR=192 and O=30; E=13.4; SMR=224).
Korallus et al. reported an update of the study. The cohort
definition was expanded to include workers with one year of employment
between January 1, 1948 and December 31, 1987 (Ex. 7-91). One thousand
four hundred and seventeen workers met the cohort entrance criteria and
were followed through December 31, 1988. While death certificates were
used, where possible, to obtain cause of death, a majority of the cause
of death data was obtained from hospital, surgical and general
practitioner reports and autopsies because of Germany's data protection
laws. Smoking data for the cohort were incomplete.
Process modifications at the two plants eliminated the high-lime
process by January 1, 1958 at one location and January 1, 1964 at the
second location. In addition, technical measures were introduced which
led to reductions in the workplace air concentrations of chromate
dusts. Cohort members were divided into pre- and post-change cohorts,
with subcohorts in the pre-change group. SMRs were computed with the
expected number of deaths derived from the regional mortality rates
(where the plants are located). One plant had 695 workers (279 in the
pre-change group and 416 in the post change group). The second plant
had 722 workers (460 in the pre-change group and 262 in the post-change
group). A total of 489 deaths were ascertained (225 and 264 deaths). Of
the cohort members, 6.4% were lost to follow-up.
Lung cancer is defined as deaths coded 162 in the 9th revision of
the International Classification of Diseases. There were 32 lung cancer
deaths at one plant and 43 lung cancer deaths at the second plant. Lung
cancer SMRs by date of entry (which differ slightly by plant) show
elevated but declining SMRs for each plant, possibly due to lower
Cr(VI) exposure as a result of improvements in production process. The
lung cancer SMR for those hired before 1948 at Plant 1 is statistically
significant (O=13; SMR=225; 95% CI: 122-382). The overall lung cancer
SMR for Plant 1 is also statistically significantly elevated based upon
32 deaths (SMR=175; 95% CI: 120-246). At Plant 2, the only lung cancer
SMR that is not statistically significant is for those hired after 1963
(based upon 1 death). Lung cancer SMRs for those hired before 1948
(O=23; SMR=344; 95% CI: 224-508) and for those hired between 1948 and
1963 (O=19; SMR=196; 95% CI: 1.24-2.98) are statistically significantly
elevated. The overall lung cancer SMR at Plant 2 based upon 43 deaths
is 239 (95% CI: 177-317). No nasal cavity neoplasms were found. A
statistically significant SMR for stomach cancer was observed at Plant
2 (O=12; SMR=192; 95% CI: 104-324).
Recently, the mortality experience of the post-change workers
identified by Korallus et al. was updated in a study by Birk et al.
(Ex. 48-4). The study cohort consisted of 901 post-change male workers
from two German chromate production plants (i.e. 472 workers and 262
workers, respectively) employed for at least one year. Review of
employment records led to the addition of employees to the previous
Korallus cohort. Mortality experience of the cohort was evaluated
through 1998. A total of 130 deaths were ascertained, of which 22 were
due to cancer of the lung. Four percent of the cohort was lost to
follow-up. Specific cause of death could not be determined for 14
decedents. The mean duration of Cr(VI) exposure was 10 years and the
mean time since first exposure was 17 years. The proportion of workers
who ever smoked was 65 percent.
The cohort lacked sufficient job history information and air
monitoring data to develop an adequate job-exposure matrix required to
estimate individual airborne exposures (Ex. 48-1-2). Instead, the
researchers used the over 12,000 measurements of urinary chromium from
routine biomonitoring of plant employees collected over the entire
study period to derive individual cumulative urinary chromium estimates
as an exposure surrogate. The approximate geometric average of all
urinary chromium measurements in the two German plants from 1960 to
1998 was 7-8 [mu]g/dl (Ex. 48-1-2, Table 5). There was a general plant-
wide decline in average urinary chromium over time from 30 to 50 [mu]g/
dl in the 1960s to less than 5 [mu]g/dl in the 1990s (Ex. 48-4, Figure
1). However, there was substantial variation in urinary chromium by
work location and job group.
The study reported a statistically significant deficit in all cause
mortality (SMR=80 95% CI: 67-95) and mortality due to heart disease
(SMR=66 95% CI: 45-93) based on the age- and calendar year-adjusted
German national population rates indicating a healthy worker
population. However, the SMR for lung cancer mortality was elevated
(SMR=148 95% CI: 93-225) against the same reference population (Ex. 48-
4, Table 2). There was a statistically significant two-fold excess lung
cancer mortality (SMR=209; 95% CI: 108-365; 12 observed lung cancer
deaths) among workers in the highest cumulative exposure grouping (i.e.
>200 [mu]g Cr/L-yr). There was no increase in lung cancer mortality in
the lower exposure groups, but the number of lung cancer deaths was
small (i.e. < =5 deaths) and the confidence intervals were wide.
There were no obvious trends in lung cancer mortality with
employment duration or time since first employed, but the results were,
again, limited by the small number of study subjects per group.
Logistic regression analysis showed that cumulative urinary chromium >=
200 [mu]g Cr/L-yr was associated with a significantly higher risk of
lung cancer death (OR=6.9; 95% CI: 2.6-18.2) when compared against
workers exposed to lower cumulative urinary chromium exposures. This
risk was unchanged after controlling for smoking status indicating that
the elevated risks were unlikely to be confounded by smoking. Including
a peak exposure score to the regression analysis did not result in
additional risk beyond that associated with cumulative exposure alone.
Some commenters felt this German post-change cohort provided evidence
for an exposure threshold below which there is no risk of lung cancer.
This issue is addressed in Section VI.B.7 of the quantitative risk
assessment.
DeMarco et al. conducted a cohort study of chromate production
workers in northern Italy to assess the existence of excess risk of
respiratory cancer, specifically lung cancer (Ex. 7-54). The cohort was
defined as males who worked for a minimum of one year from 1948 to 1985
and had at least 10 years of follow-up. Five hundred forty workers met
the cohort definition. Vital status follow-up, carried out through June
30, 1985, found 427 cohort members alive, 110 dead and three lost to
follow-up. Analysis utilizing SMRs based on Italian national rates was
conducted. Of the 110 deaths, 42 were cancer deaths. The statistically
significant SMR for lung cancer based upon 14 observed deaths with 6.46
expected was 217 (95% CI: 118-363).
Exposure estimates were based upon the duration of cumulative
exposure and upon a risk score (low, medium, high and not assessed)
assigned to the department in which the worker was primarily employed.
A committee assigned the scores, based upon knowledge of the production
process or on industrial hygiene surveys taken in 1974, 1982 and 1984.
The risk score is a surrogate for the workplace concentrations of Cr(VI)
in the different plant departments. Since no substantial changes had been
made since World War II, the assumption was made that exposures remained
relatively stable. Lung cancer SMRs based upon type of exposure increased
with level of exposure (Low: O=1; E=1.43; SMR=70; Medium: O=5; E=202; SMR=2.48;
High: O=6; E=1.4; SMR=420; Not Assessed: O=2; E=1.6; SMR=126). Only the SMR
for those classified as having worked in departments characterized as high
exposure was statistically significant at the p< 0.05 level.
A cohort study of workers at a chromium compounds manufacturing
plant in Tokyo, Japan by Satoh et al. included males employed between
1918 and 1975 for a minimum of one year and for whom the necessary data
were available (Ex. 7-27). Date and cause of death data were obtained
from the death certificate (85%) or from other "reliable" written
testimony (15%). Of the 1,061 workers identified, 165 were excluded
from the study because information was missing. A total of 896 workers
met the cohort inclusion criteria and were followed through 1978. The
causes of 120 deaths were ascertained. SMRs based on age-cause specific
mortality for Japanese males were calculated for four different time
periods (1918-1949; 1950-1959; 1960-1969 and 1970-1978) and for the
entire follow-up period (1918-1978). An elevated SMR for lung cancer is
seen for the entire follow-up period (O=26; E=2.746; SMR=950). A
majority of the lung cancer deaths (20) occurred during the 1970-1978
interval.
Results from the many studies of chromate production workers from
different countries indicate a relationship between exposure to
chromium and malignant respiratory disease. The epidemiologic studies
done between 1948 and 1952 by Machle and Gregorius (Ex. 7-2), Mancuso
and Hueper (Ex. 7-12) and Brinton, et al. (Ex. 7-1) suggest a risk for
respiratory cancer among chromate workers between 15 and 29 times
expectation. Despite the potential problems with the basis for the
calculations of the expectations or the particular statistical methods
employed, the magnitude of the difference between observed and expected
is powerful enough to overcome these potential biases.
It is worth noting that the magnitude of difference in the relative
risks reported in a mortality study among workers in three chromate
plants in the U.K. (Ex.7-20) were lower than the relative risks
reported for chromate workers in the U.S. during the 1950s and 1960s.
The observed difference could be the result of a variety of factors
including different working conditions in the two countries, a shorter
follow-up period in the British study, the larger lost-to-follow-up in
the British study or the different statistical methods employed. While
the earlier studies established that there was an excess risk for
respiratory cancer from exposure to chromium, they were unable to
specify either a specific chromium compound responsible or an exposure
level associated with the risk. Later studies were able to use superior
methodologies to estimate standardized lung cancer mortality ratios
between chromate production cohorts and appropriate reference
populations (Exs. 7-14; 7-22; 7-26; 7-99; 7-91). These studies
generally found statistically increased lung cancer risk of around two-
fold. The studies usually found trends with duration of employment,
year of hire, or some production process change that tended to
implicate chromium exposure as the causative agent.
Some of the most recent studies were able to use industrial hygiene
data to reconstruct historical Cr(VI) exposures and show statistically
significant associations between cumulative airborne Cr(VI) and lung
cancer mortality (Exs. 23; 31-22-11; Ex. 31-18-4). Gibb et al. found
the significant association between Cr(VI) and lung cancer was evident
in models that accounted for smoking. The exposure'response
relationship from these chromate production cohorts provide strong
evidence that occupational exposure to Cr(VI) dust can increase cancer
in the respiratory tract of workers.
The Davies, Korallus, (German cohort), Luippold (2003), and
Luippold (2005) studies examine mortality patterns at chromate
producing facilities where one production process modification involved
conversion from a high-lime to a low-lime or a lime-free process (Exs.
7-99; 7-91; 31-18-4). In addition to process modification, technical
improvements also were implemented that lowered Cr(VI) exposure. One of
the plants in the Davies study retained the high-lime process and is
not discussed. The lung cancer SMRs for one British plant and both of
the German plants decline from early, to pre-change to post change time
periods. In the remaining British plants, the lung cancer SMR is
basically identical for the early and pre-change period, but does
decline in the post-change time period. The lung cancer SMR in the
Luippold 2003 cohort also declined over time as the amount of lime was
reduced in the roasting process. Other modifications at the Painesville
plant that reduced airborne Cr(VI) exposure, such as installation of
covered conveyors and conversion from batch to continuous process,
occurred at the same time (Ex. 35-61). The workers in the Luippold
(2005) study were not exposed to Cr(VI) in facilities using a high-lime
process. This study did not show excess risk; however, this may be a
consequence of short follow-up time (< 20 years for most workers) or
the small size of the study (< 4 expected lung cancers), as discussed
further in Section VI.B.7. In general, it is not clear whether reduced
levels of the high-lime byproduct, calcium chromate, or the roasting/
leaching end product, sodium dichromate, that resulted from the various
process changes is the reason for the decrease in lung cancer SMRs in
these cohorts. It should be noted that increased lung cancer risk was
experienced by workers at the Baltimore plant (e.g., Hayes and Gibb
cohorts) even though early air monitoring studies suggest that a high
lime process was probably not used at this facility (Ex. 7-17).
2. Evidence From Chromate Pigment Production Workers
Chromium compounds are used in the manufacture of pigments to
produce a wide range of vivid colors. Lead and zinc chromates have
historically been the predominant hexavalent chromium pigments,
although others such as strontium and barium chromate have also been
produced. These chromates vary considerably in their water solubility
with lead and barium chromates being the most water insoluble. All of
the above chromates are less water-soluble than the highly water-
soluble sodium chromate and dichromate that usually serve as the
starting material for chromium pigment production. The reaction of
sodium chromate or dichromate with the appropriate zinc or lead
compound to form the corresponding lead or zinc chromate takes place in
solution. The chromate pigment is then precipitated, separated, dried,
milled, and packaged. Worker exposures to chromate pigments are
greatest during the milling and packaging stages.
There have been a number of cohort studies of chromate pigment
production workers from the United States, the United Kingdom, France,
Germany, the Netherlands, Norway and Japan. Most of the studies found
significantly elevated lung cancers in workers exposed to Cr(VI)
pigments over many years when compared against standardized reference
populations. In general, the studies of chromate pigment workers lack
the historical exposure data found in some of the chromate production
cohorts. The consistently higher lung cancers across several worker
cohorts exposed to the less water-soluble Cr(VI) compounds complements
the lung cancer findings from the studies of workers producing highly
water soluble chromates and adds to the further evidence that occupational
exposure to Cr(VI) compounds should be regarded as carcinogenic. A summary
of selected human epidemiologic studies in chromate production workers is
presented in Table V-2.
BILLING CODE 4510-26-P

BILLING CODE 4510-26-C
Langard and Vigander updated a cohort study of lung cancer
incidence in 133 workers employed by a chromium pigment production
company in Norway (Ex. 7-36). The cohort was originally studied by
Langard and Norseth (Ex. 7-33). Twenty four men had more than three
years of exposure to chromate dust. From 1948, when the company was
founded, until 1951, only lead chromate pigment was produced.
From 1951 to 1956, both lead chromate and zinc chromate pigments were
produced and from 1956 to the end of the study period in 1972 only zinc
chromate was produced. Workers were exposed to chromates both as the
pigment and its raw material, sodium dichromate.
The numbers of expected lung cancers in the workers were calculated
using the age-adjusted incidence rates for lung cancer in the Norwegian
male population for the period 1955-1976. Follow-up using the Norwegian
Cancer Registry through December 1980, found the twelve cancers of
which seven were lung cancers. Six of the seven lung cancers were
observed in the subcohort of 24 workers who had been employed for more
than three years before 1973. There was an increased lung cancer
incidence in the subcohort based on an observed to expected ratio of 44
(O=6; E=0.135). Except for one case, all lung cancer cases were exposed
to zinc chromates and only sporadically to other chromates. Five of the
six cases were known to be smokers or ex-smokers. Although the authors
did not report any formal statistical comparisons, the extremely high
age-adjusted standardized incidence ratio suggests that the results
would likely be statistically significant.
Davies reported on a cohort study of English chromate pigment
workers at three factories that produced chromate pigments since the
1920s or earlier (Ex. 7-41). Two of the factories produced both zinc
and lead chromate. Both products were made in the same sheds and all
workers had mixed exposure to both substances. The only product at the
third factory was lead chromate.
Cohort members are defined as males with a minimum of one year of
employment first hired between 1933 and 1967 at plant A; 1948 and 1967
at plant B and 1946-1961 at plant C. The analysis excludes men who
entered employment later than 1967 because of the short follow-up
period. Three hundred and ninety six (396) men from Factory A, 136 men
from Factory B and 114 men from Factory C were followed to mid-1977.
Ninety-four workers with 3-11 months employment during 1932-1945 at
Factory A were also included. Expectations were based upon calendar
time period-, gender- and age-specific national cancer death rates for
England and Wales. The author adjusted the death rates for each factory
for local differences, but the exact methods of adjustment were not
explicit.
Exposure to chromates was assigned as high for those in the dry
departments where pigments were ground, blended and packed; medium for
those in the wet departments where precipitates were washed, pressed
and stove dried and in maintenance or cleaning which required time in
various departments; or low for those jobs which the author states
involved "slight exposure to chromates such as most laboratory jobs,
boiler stoking, painting and bricklaying" (Ex. 7-41, p. 159). The high
and medium exposure categories were combined for analytical purposes.
For those entering employment from 1932 to 1954 at Factory A, there
were 18 lung cancer deaths in the high/medium exposure group, with 8.2
deaths expected. The difference is significant at p< .01. In the low
exposure group, the number of observed and expected lung cancer deaths
was equal (two deaths). There were no lung cancer deaths at Factory A
for those hired between 1955-1960 and 1961-1967.
For those entering employment between 1948 and 1967 at Factory B,
there were seven observed lung cancer deaths in the high/medium
exposure group with 1.4 expected which is statistically significant at
p< .001. At Factory C (which manufactured only lead chromate), there was
one death in the high/medium exposure group and one death in the low
exposure group for those beginning employment between 1946 and 1967.
The author points out that:
There has been no excess lung cancer mortality amongst workers
with chromate exposure rated as "low", nor among those exposed
only to lead chromate. High and medium exposure-rated workers who in
the past had mixed exposure to both lead and zinc chromate have
experienced a marked excess of lung cancer deaths, even if employed
for as little as one year (Ex. 7-41, p. 157).
It is the author's opinion that the results "suggest that the
manufacture of zinc chromate may involve a lung cancer hazard" (Ex. 7-
41, p. 157).
Davies updated the lung cancer mortality at the three British
chromate pigment production factories (Ex. 7-42). The follow-up was
through December 31, 1981. The cohort was expanded to include all male
workers completing one year of service by June 30, 1975 but excluded
office workers.
Among workers at Factory A with high and medium exposure, mortality
was statistically significantly elevated over the total follow-up
period among entrants hired from 1932 to 1945 (O/E=2.22). A similar,
but not statistically significant, excess was seen among entrants hired
from 1946 to 1954 (O/E=2.23). The results for Factory B showed
statistically significantly elevated lung cancer mortality among
workers classified with medium exposures entering service during the
period from 1948 to 1960 (O/E=3.73) and from 1961 to 1967 (O/E=5.62).
There were no lung cancer deaths in the high exposure group in either
time period. At Factory C, analysis by entry date (early entrant and
the period 1946-1960) produced no meaningful results since the number
of deaths was small. When the two periods are combined, the O/E was
near unity. The author concluded that in light of the apparent absence
of risk at Factory C, "it seems reasonable to suggest that the hazard
affecting workers with mixed exposures at factories A and B * * * is
attributable to zinc chromates" (Ex. 7-42, p. 166). OSHA disagrees
with this conclusion, as discussed in section V.9.
Davies also studied a subgroup of 57 chromate pigment workers,
mostly employed between 1930 and 1945, who suffered clinical lead
poisoning (Ex. 7-43). Followed through 1981, there was a statistically
significantly elevated SMR for lung cancer based upon four cases (O=4;
E=2.8; SMR=145).
Haguenoer studied 251 French zinc and lead chromate pigment workers
employed for six months or more between January 1, 1958 and December
31, 1977 (Ex. 7-44). As of December 31, 1977, 50 subjects were
identified as deceased. Cause of death was obtained for 30 of the 50
deaths (60%). Lung cancer mortality was significantly elevated based on
11 fatalities (SMR=461; 95% CI: 270-790). The mean time from first
employment until detection of cancer was 17 years. The mean duration of
employment among cases was 15 years.
The Haguenoer cohort was followed up in a study by Deschamps et al.
(Ex. 234). Both lead and zinc chromate pigments were produced at the
plant until zinc chromate production ceased in 1986. The cohort
consisted of 294 male workers employed for at least six months between
1958 and 1987. At the end of the follow-up, 182 cohort members were
alive, 16 were lost to follow-up and 96 were dead. Because of French
confidentiality rules, the cause of death could not be obtained from
the death certificate; instead physicians and hospital records were
utilized. Using cause of death data from sources other than death
certificates raises the potential for misclassification bias. Cause of
death could not be obtained for five decedents. Data on smoking habits
was not available for a number of workers and was not used in the
analysis.
Since individual work histories were not available, the authors
made the assumption that the exposure level was the same for all
workers during their employment at the plant. Duration of employment
was used as a surrogate for exposure. Industrial hygiene measurements
taken in 1981 provide some idea of the exposure levels at the plant.
In the filtration department, Cr(VI) levels were between 2 and 3 [mu]g/m\3\;
in the grinding department between 6 and 165 [mu]g/m\3\; in the drying and
sacking department between 6 and 178 [mu]g/m\3\; and in the sacks
marking department more than 2000 [mu]g/m\3\.
The expected number of deaths for the SMR analysis was computed
from age-adjusted death rates in the northern region of France where
the plant was located. There was a significant increase in lung cancer
deaths based on 18 fatalities with five expected (SMR=360; 95% CI: 213-
568). Using duration of employment as a surrogate for exposure,
statistically significant SMRs were seen for the 10-15 years of
exposure (O=6, SMR=720, 95% CI: 264-1568), 15-20 years (O=4, SMR=481,
95% CI: 131-1231), and 20+ years (O=6, SMR=377, 95% CI: 1.38-8.21) time
intervals. There was a significantly elevated SMR for brain cancer
based upon two deaths (SMR=844, 95% CI: 102-3049). There was a non-
statistically significant increase for digestive tract cancer (O=9,
SMR=130) consisting of three esophageal cancers, two stomach cancers
and four colon cancers.
Equitable Environmental Health, Inc., on behalf of the Dry Color
Manufacturers Association, undertook a historical prospective mortality
study of workers involved in the production of lead chromate (Exs. 2-D-
3; 2-D-1). The cohort was defined as male employees who had been
exposed to lead chromate for a minimum of six months prior to December
1974 at one of three facilities in West Virginia, Kentucky or New
Jersey. The New Jersey facility had a unit where zinc chromate was
produced dating back to 1947 (Ex. 2-D-3). Most workers rotated through
this unit and were exposed to both lead and zinc chromates. Two men
were identified at the New Jersey facility with exposure solely to lead
chromate; no one with exposure only to zinc chromate was identified.
Subsequent review of the data found that the Kentucky plant also
produced zinc chromates from the late 1930s to early 1964. During the
period 1961-1962, zinc chromates accounted for approximately 12% of
chromate production at the plant. In addition, strontium chromate and
barium chromate also were produced at the plant.
The cohort consisted of 574 male employees from all three plants
(Ex. 2-D-1). Eighty-five deaths were identified with follow up through
December 1979. Six death certificates were not obtained. SMRs were
reported based on U.S. white male death rates. There were 53 deaths
from the New Jersey plant including a statistically significant SMR for
cancer of the trachea, bronchus and lung based upon nine deaths (E=3.9;
SMR=231; 95% CI: 106-438). One lung cancer decedent worked solely in
the production of lead chromates. Three of the lung cancer deaths were
black males. In addition, there were six deaths from digestive system
cancers, five of which were stomach cancers reported at the New Jersey
plant. The SMR for stomach cancer was statistically significantly
elevated (O=5; E=0.63; SMR=792; 99% CI: 171-2243). There were 21 deaths
from the West Virginia plant, three of which were cancer of the
trachea, bronchus and lung (E=2.3; SMR=130; 95% CI: 27-381). There were
11 deaths at the Kentucky plant, two of which were cancer of the
trachea, bronchus and lung (E=0.9; SMR=216; 95% CI: 26-780).
Sheffet et al. examined the lung cancer mortality among 1,946 male
employees in a chromate pigment factory in Newark, NJ, who were exposed
to both lead chromate and zinc chromate pigments (Ex. 7-48). The men
worked for a minimum of one month between January 1, 1940 and December
31, 1969. As of March 31, 1979, a total of 321 cohort members were
identified as deceased (211 white males and 110 non-white males). Cause
of death could not be ascertained for 37 white males and 12 non-white
males. The proportion of the cohort lost to follow up was high (15% of
white males and 20% of non-white males).
Positions at the plant were classified into three categories
according to intensity of exposure: high (continuous exposure to
chemical dust), moderate (occasional exposure to chemical dust or to
dry or wet pigments) and low (infrequent exposure by janitors or office
workers). Positions were also classified by type of chemical exposure:
chromates, other inorganic substances, and organics. The authors state
that in almost all positions individuals "who were exposed to any
chemicals were also exposed to hexavalent chromium in the form of
airborne lead and zinc chromates (Ex. 7-48, p. 46)." The proportion of
lead chromate to zinc chromate was approximately nine to one.
Calculations, based upon air samples during later years, give an
estimate for the study period of more than 2000 [mu]g airborne
chromium/m\3\ for the high exposure category, between 500 and 2000
[mu]g airborne chromium/m\3\ and less than 100 [mu]g airborne chromium/
m\3\ for the low exposure category. Other suspected carcinogens present
in the workplace air at much lower levels were nickel sulfate and
nickel carbonate.
Because of the large proportion of workers lost to follow-up (15%
of white males and 20% of non-white males) and the large numbers of
unknown cause of death (21% of white males and 12% of non-white males),
the authors calculated three separate mortality expectations based upon
race-, gender-, age-, and time-specific U.S. mortality ratios. The
first expectation was calculated upon the assumption that those lost to
follow-up were alive at the end of the study follow-up period. The
second expectation was calculated on the assumption that those whose
vital status was unknown were lost to follow-up as of their employment
termination date. The third expectation was calculated excluding those
of unknown vital status from the cohort. Deaths with unknown cause were
distributed in the appropriate proportions among known causes of death
which served as an adjustment to the observed deaths. The adjusted
deaths were used in all of the analyses.
A statistically significant ratio for lung cancer deaths among
white males (O/E=1.6) was observed when using the assumption that
either the lost to follow-up were assumed lost as of their termination
date or were excluded from the cohort (assumptions two and three
above). The ratio for lung cancer deaths for non-white males results in
an identical O/E of 1.6 for all three of the above scenarios, none of
which was statistically significant.
In addition, the authors also conducted Proportionate Mortality
Ratio (PMR) and Proportionate Cancer Mortality Ratio (PCMR) analyses.
For white males, the lung cancer PMR was 200 and the lung cancer PCMR
was 160 based upon 25.5 adjusted observed deaths (21 actual deaths).
Both were statistically significantly elevated at the p< .05 level. For
non-white males, the lung cancer PMR was 200 and the lung cancer PCMR
was 150 based upon 11.2 adjusted observed deaths (10 actual deaths).
The lung cancer PMR for non-white males was statistically significantly
elevated at the p< .05 level. Statistically significantly elevated PMRs
and PCMRs for stomach cancer in white males were reported (PMR=280;
PCMR=230) based upon 6.1 adjusted observed deaths (five actual).
The Sheffet cohort was updated in a study by Hayes et al. (Ex. 7-
46). The follow up was through December 31, 1982. Workers employed as
process operators or in other jobs which involved direct exposure to
chromium dusts were classified as having exposure to chromates. Airborne
chromium concentrations taken in "later years" were estimated to be
>500 [mu]g g/m\3\ for "exposed" jobs and >2000 [mu]g/m\3\ for
"highly exposed" jobs.
The cohort included 1,181 white and 698 non-white males. Of the 453
deaths identified by the end of the follow-up period, 41 were lung
cancers. For the entire study group, no statistically significant
excess was observed for lung cancer (SMR=116) or for cancer at any
other site. Analysis by duration of employment found a statistically
significant trend (p=.04) for lung cancer SMRs (67 for those employed
< 1 year; 122 for those employed 1-9 years and 151 for those employed
10+ years).
Analysis of lung cancer deaths by duration of employment in
chromate dust associated jobs found no elevation in risk for subjects
who never worked in these jobs (SMR=92) or for subjects employed less
than one year in these jobs (SMR=93). For those with cumulative
employment of 1-9 and 10+ years in jobs with chromate dust exposure,
the SMRs were 176 (nine deaths) and 194 (eight deaths) respectively.
Frentzel-Beyme studied the mortality experience of 1,396 men
employed for more than six months in one of five factories producing
lead and zinc chromate pigments located in Germany and the Netherlands
(Ex. 7-45). The observed deaths from the five factories were compared
with the expected deaths calculated on the basis of mortality figures
for the region in which the plant was located. Additional analysis was
conducted on relevant cohorts which included workers with a minimum of
10 years exposure, complete records for the entire staff, and exclusion
of foreign nationals. Jobs were assigned into one of three exposure
categories: High (drying and milling of the filtered pigment paste),
medium (wet processes including precipitation of the pigment, filtering
and maintenance, craftsmen and cleaning) and low or trivial exposure
(storage, dispatch, laboratory personnel and supervisors).
There were 117 deaths in the entire cohort of which 19 were lung
cancer deaths (E=9.3). The lung cancer SMRs in the relevant cohort
analyses were elevated at every plant; however, in only one instance
was the increased lung cancer SMR statistically significant, based upon
three deaths (SMR=386, p< 0.05). Analysis by type of exposure is not
meaningful due to the small number of lung cancer deaths per plant per
exposure classification.
Kano et al. conducted a study of five Japanese manufacturers who
produced lead chromates, zinc chromate, and/or strontium chromate to
assess if there was an excess risk of lung cancer (Ex. 7-118). The
cohort consisted of 666 workers employed for a minimum of one year
between 1950 and 1975. At the end of 1989, 604 subjects were alive,
five lost to follow-up and 57 dead. Three lung cancer deaths were
observed in the cohort with 2.95 expected (SMR=102; 95% CI: 0.21-2.98).
Eight stomach cancer deaths were reported with a non-statistically
significant SMR of 120.
Following the publication of the proposed rule, the Color Pigment
Manufacturers Association requested that OSHA reconsider its
preliminary conclusions with respect to the health effects of lead
chromate color pigments (Ex. 38-205). They relied on the Davies (Ex. 7-
43), Cooper [Equitable Environmental Health, Inc] (Ex. 2-D-1) and Kano
(Ex. 14-1-B) epidemiologic studies as the only available data on worker
cohorts exposed to lead chromate in the absence of other chromates
commonly found in pigment production (e.g., zinc chromate). The CPMA's
comments regarding the Davies, Cooper and Kano studies and OSHA's
response to them are discussed in section V.B.9.a.
3. Evidence from Workers in Chromium Plating
Chrome plating is the process of depositing chromium metal onto the
surface of an item using a solution of chromic acid. The items to be
plated are suspended in a diluted chromic acid bath. A fine chromic
acid mist is produced when gaseous bubbles, released by the
dissociation of water, rise to the surface of the plating bath and
burst. There are two types of chromium electroplating. Decorative or
"bright" involves depositing a thin (0.5-1 [mu]m) layer of chromium
over nickel or nickel-type coatings to provide protective, durable,
non-tarnishable surface finishes. Decorative chrome plating is used for
automobile and bicycle parts. Hard chromium plating produces a thicker
(exceeding 5 [mu]m) coating which makes it resistant and solid where
friction is usually greater, such as in crusher propellers and in
camshafts for ship engines. Limited air monitoring indicates that
Cr(VI) levels are five to ten times higher during hard plating than
decorative plating (Ex. 35-116).
There are fewer studies that have examined the lung cancer
mortality of chrome platers than of soluble chromate production and
chromate pigment production workers. The largest and best described
cohort studies investigated chrome plating cohorts in the United
Kingdom (Exs. 7-49; 7-57; 271; 35-62). They generally found elevated
lung cancer mortality among the chrome platers, especially those
engaged in chrome bath work, when compared to various reference
populations. The studies of British chrome platers are summarized in
Table V-3.
BILLING CODE 4510-26-P
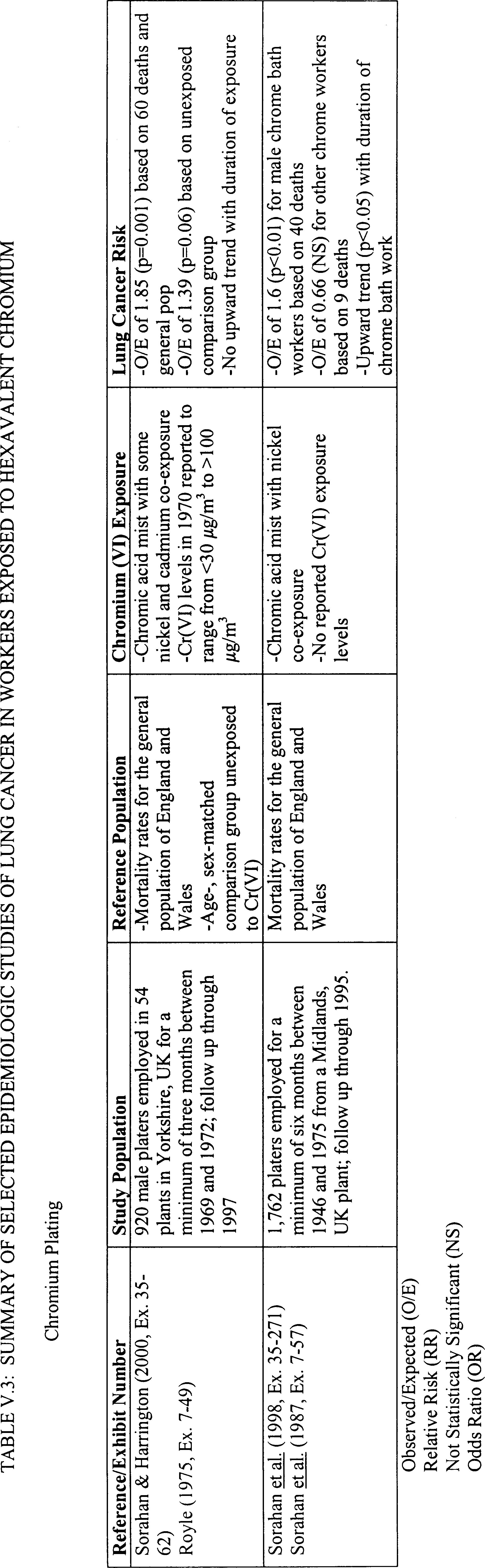
BILLING CODE 4510-26-C
Cohort studies of chrome platers in Italy, the United States, and
Japan are also discussed in this subsection. Co-exposure to nickel,
another suspected carcinogen, during plating operations can complicate
evaluation of an association between Cr(VI) and an increased risk of
lung cancer in chrome platers. Despite this, the International Agency
for Research on Cancer concluded that the epidemiological
studies provide sufficient evidence for carcinogenicity of Cr(VI) as
encountered in the chromium plating industry; the same conclusion
reached for chromate production and chromate pigment production (Exs.
18-1; 35-43). The findings implicate the highly water-soluble chromic
acid as an occupational carcinogen. This adds to the weight of evidence
that water-soluble (e.g., sodium chromates, chromic acid) and water-
insoluble forms (e.g., lead and zinc chromates) of Cr(VI) are able to
cause cancer of the lower respiratory tract.
Royle reported on a cohort mortality study of 1,238 chromium
platers employed for a minimum of three consecutive months between
February 20, 1969 and May 31, 1972 in 54 plating plants in West Riding,
Yorkshire, England (Ex. 7-49). A control population was enumerated from
other departments of the larger companies where chromium plating was
only a portion of the companies' activities and from the former and
current employees of two industrial companies in York where information
on past workers was available. Controls were matched for gender, age
(within two years) and date last known alive. In addition, 229 current
workers were matched for smoking habits.
As of May 1974, there were 142 deaths among the platers (130 males
and 12 females) and 104 deaths among the controls (96 males and 8
females). Among the male platers, there were 24 deaths from cancer of
the lung and pleura compared to 13 deaths in the control group. The
difference was not statistically significant. There were eight deaths
from gastrointestinal cancer among male platers versus four deaths in
the control group. The finding was not statistically significant.
The Royle cohort was updated by Sorahan and Harrington (Ex. 35-62).
Chrome plating was the primary activity at all 54 plants, however 49 of
the plants used nickel and 18 used cadmium. Also used, but in smaller
quantities according to the authors, were zinc, tin, copper, silver,
gold, brass or rhodium. Lead was not used at any of the plants. Four
plants, including one of the largest, only used chromium. Thirty-six
chrome platers reported asbestos exposure versus 93 comparison workers.
Industrial hygiene surveys were carried out at 42 plants during
1969-1970. Area air samples were done at breathing zone height. With
the exception of two plants, the chromic acid air levels were less than
30 [mu]g/m\3\. The two exceptions were large plants, and in both the
chromic acid levels exceeded 100 [mu]g/m\3\.
The redefined cohort consisted of 1087 platers (920 men and 167
women) from 54 plants employed for a minimum of three months between
February 1969 and May 31, 1972 who were alive on May 31, 1972.
Mortality data were also available for a comparison group of 1,163
workers (989 men and 174 women) with no chromium exposure. Both groups
were followed for vital status through 1997.
The lung cancer SMR for male platers was statistically significant
(O=60; E=32.5; SMR=185; 95% CI: 141-238). The lung cancer SMR for the
comparison group, while elevated, was not statistically significant
(O=47; E=36.9; SMR=127; 95% CI: 94-169). The only statistically
significant SMR in the comparison group was for cancer of the pleura
(O=7; E=0.57; SMR=1235; 95% CI: 497-2545).
Internal regression analyses were conducted comparing the mortality
rates of platers directly with those of the comparison workers. For
these analyses, lung cancers mentioned anywhere on the death
certificate were considered cases. The redefinition resulted in four
additional lung cancer cases in the internal analyses. There was a
statistically significant relative risk of 1.44 (p< 0.05) for lung
cancer mortality among chrome platers that was slightly reduced to 1.39
after adjustment for smoking habits and employment status. There was no
clear trend between lung cancer mortality and duration of Cr(VI)
exposure. However, any positive trend may have been obscured by the
lack of information on worker employment post-1972 and the large
variation in chromic acid levels among the different plants.
Sorahan reported the experience of a cohort of 2,689 nickel/
chromium platers from the Midlands, U.K. employed for a minimum of six
months between 1946 and 1975 and followed through December 1983 (Ex. 7-
57). There was a statistically significant lung cancer SMR for males
(O=63; E=40; SMR=158; p< 0.001). The lung cancer SMR for women, while
elevated (O=9; E=8.1; SMR=111), was not statistically significant.
Other statistically significant cancer SMRs for males included: stomach
(O=21; E=11.3; SMR=186; p< 0.05); liver (O=4; E=0.6; SMR=667; p< 0.01);
and nasal cavities (O=2; E=0.2; SMR=1000; p< 0.05). While there were
several elevated SMRs for women, none were statistically significant.
There were nine lung cancers and one nasal cancer among the women.
Analysis by type of first employment (i.e., chrome bath workers vs.
other chrome work) resulted in a statistically significant SMR for lung
cancer of 199 (O=46; E=23.1; p< 0.001) for chrome bath workers and a SMR
of 101 for other chrome work. The SMR for cancer of the stomach for
male chrome bath workers was also statistically significantly elevated
(O=13; E=6.3; SMR=206; p< 0.05); for stomach cancer in males doing other
chrome work, the SMR was 160 with 8 observed and 5 expected. Both of
the nasal cancers in males and the one nasal cancer in women were
chrome bath workers. The nasal cancer SMR for males was statistically
significantly elevated (O=2; E=0.1; SMR=2000; p< 0.05).
Regression analysis was used to examine evidence of association of
several types of cancers and Cr(VI) exposure duration among the cohort.
There was a significant positive association between lung cancer
mortality and exposure duration as a chrome bath worker controlling for
gender as well as year and age at the start of employment. There was no
evidence of an association between other cancer types and duration of
Cr(VI) exposure. There was no positive association between duration of
exposure to nickel bath work and cancer of the lung. The two largest
reported SMRs were for chrome bath workers 10-14 years (O=13; E=3.8;
SMR=342; p< 0.001) and 15-19 years (O=12; E=4.9; SMR=245; p< 0.01) after
starting employment. The positive associations between lung cancer
mortality and duration of chrome bath work suggests Cr(VI) exposure may
be responsible for the excess cancer risk.
Sorahan et al. reported the results of a follow-up to the nickel/
chromium platers study discussed above (Ex. 271). The cohort was
redefined and excluded employees whose personnel records could not be
located (650); those who started chrome work prior to 1946 (31) and
those having no chrome exposure (236). The vital status experience of
1,762 workers (812 men and 950 women) was followed through 1995. The
expected number of deaths was based upon the mortality of the general
population of England and Wales.
There were 421 deaths among the men and 269 deaths among the women,
including 52 lung cancers among the men and 17 among the women. SMRs
were calculated for different categories of chrome work: Period from
first chrome work; year of starting chrome work, and cumulative
duration of chrome work categories. Poison regression modeling was
employed to investigate lung cancer in relation to type of chrome work
and cumulative duration of work.
A significantly elevated lung cancer SMR was seen for male workers
with some period of chrome bath work (O=40; E=25.4; SMR=157; 95% CI: 113-
214, p< 0.01). Lung cancer was not elevated among male workers engaged
in other chrome work away from the chromic acid bath (O=9; E=13.7;
SMR=66; 95% CI: 30-125). Similar lung cancer mortality results were
found for female chrome bath workers (O=15; E=8.6; SMR=175; 95% CI: 98-
285; p< 0.06). After adjusting for sex, age, calendar year, year
starting chrome work, period from first chrome work, and employment
status, regression modeling showed a statistically significant positive
trend (p< 0.05) between duration of chrome bath work and lung cancer
mortality risk. The relative lung cancer risk for chrome bath workers
with more than five years of Cr(VI) exposure (i.e., relative to the
risk of those without any chrome bath work) was 4.25 (95% CI: 1.83-
9.37).
Since the Sorahan cohort consists of nickel/chromium workers, the
question arises of the potential confounding of nickel. In the earlier
study, 144 of the 564 employees with some period of chrome bath work
had either separate or simultaneous periods of nickel bath employment.
According to the authors, there was no clear association between cancer
deaths from stomach, liver, respiratory system, nose and larynx, and
lung and bronchus and the duration of nickel bath employment. In the
follow-up report, the authors re-iterate this result stating,
"findings for lung cancer in a cohort of nickel platers (without any
exposure to chrome plating) from the same factory are unexceptional"
(Ex. 35-271, p. 241).
Silverstein et al. reported the results of a cohort study of hourly
employees and retirees with at least 10 years of credited pension
service in a Midwestern plant manufacturing hardware and trim
components for use primarily in the automobile industry (Ex. 7-55). Two
hundred thirty eight deaths occurred between January 1, 1974 and
December 31, 1978. Proportional Mortality Ratio (PMR) analysis adjusted
for race, gender, age and year of death was conducted. For white males,
the PMR for cancer of the lung and pleura was 1.91 (p< 0.001) based upon
28 deaths. For white females, the PMR for cancer of the lung and pleura
was 3.70 (p< 0.001) based upon 10 deaths.
White males who worked at the plant for less than 15 years had a
lung cancer PMR of 1.65. Those with 15 or more years at the plant had a
lung cancer PMR of 2.09 (p< 0.001). For white males with less than 22.5
years between hire and death (latency) the lung cancer PMR was 1.78
(p< 0.05) and for those with 22.5 or more years, the PMR was 2.11
(p< 0.01).
A case-control analysis was conducted on the Silverstein cohort to
examine the association of lung cancer risk with work experience.
Controls were drawn from cardiovascular disease deaths (ICD 390-458,
8th revision). The 38 lung cancer deaths were matched to controls for
race and gender. Odds ratios (ORs) were calculated by department
depending upon the amount of time spent in the department (ever/never;
more vs. less than one year; and more vs. less than five years). Three
departments showed increasing odds ratios with duration of work;
however, the only statistically significant result was for those who
worked more than five years in department 5 (OR=9.17, p=0.04, Fisher's
exact test). Department 5 was one of the major die-casting and plating
areas of the plant prior to 1971.
Franchini et al. conducted a mortality study of employees and
retirees from nine chrome plating plants in Parma, Italy (Ex. 7-56).
Three plants produced hard chrome plating. The remaining six plants
produced decorative chromium plates. A limited number of airborne
chromium measurements were available. Out of a total of 10 measurements
at the hard chrome plating plants, the air concentrations of chromium
averaged 7 [mu]g/m\3\ (range of 1-50 [mu]g/m\3\) as chromic acid near
the baths and 3 [mu]g/m\3\ (range of 0-12 [mu]g/m\3\) in the middle of
the room.
The cohort consisted of 178 males (116 from the hard chromium
plating plants and 62 from the bright chromium plating plants) who had
worked for at least one year between January 1, 1951 and December 31,
1981. In order to allow for a 10-year latency period, only those
employed before January 1972 were included in further analysis. There
were three observed lung cancer deaths among workers in the hard chrome
plating plants, which was significantly greater than expected (O=3;
E=0.6; p< 0.05). There were no lung cancer deaths among decorative
chrome platers.
Okubo and Tsuchiya conducted a study of plating firms with five or
more employees in Tokyo (Exs. 7-51; 7-52). Five hundred and eighty nine
firms were sent questionnaires to ascertain information regarding
chromium plating experience. The response rate was 70.5%. Five thousand
one hundred seventy platers (3,395 males and 1,775 females) met the
cohort entrance criteria and were followed from April 1, 1970 to
September 30, 1976. There were 186 deaths among the cohort; 230 people
were lost to follow-up after retirement. The cohort was divided into
two groups: Chromium platers who worked six months or more and a
control group with no exposure to chromium (clerical, unskilled
workers). There were no deaths from lung cancer among the chromium
platers.
The Okubo cohort was updated by Takahashi and Okubo (Ex. 265). The
cohort was redefined to consist of 1,193 male platers employed for a
minimum of six months between April 1970 and September 1976 in one of
415 Tokyo chrome plating plants and who were alive and over 35 years of
age on September 30, 1976. The only statistically significant SMR was
for lung cancer for all platers combined (O=16; E=8.9; SMR=179; 95% CI:
102-290). The lung cancer SMR for the chromium plater subcohort was 187
based upon eight deaths and 172 for the nonchromium plater subcohort,
also based upon eight deaths. The cohort was followed through 1987.
Itoh et al. updated the Okubo metal plating cohort through December
1992 (Ex. 35-163). They reported a lung cancer SMR of 118 (95% CI: 99-
304).
4. Evidence From Stainless Steel Welders
Welding is a term used to describe the process for joining any
materials by fusion. The fumes and gases associated with the welding
process can cause a wide range of respiratory exposures which may lead
to an increased risk of lung cancer. The major classes of metals most
often welded include mild steel, stainless and high alloy steels and
aluminum. The fumes from stainless steel, unlike fumes from mild steel,
contain nickel and Cr(VI). There are several cohort and case-control
studies as well as two meta analyses of welders potentially exposed to
Cr(VI). In general, the studies found an excess number of lung cancer
deaths among stainless steel welders. However, few of the studies found
clear trends with Cr(VI) exposure duration or cumulative Cr(VI). In
most studies, the reported excess lung cancer mortality among stainless
steel welders was no greater than mild steel welders, even though
Cr(VI) exposure is much greater during stainless steel welding. This
weak association between lung cancer and indices of exposure limits the
evidence provided by these studies. Other limitations include the co-
exposures to other potential lung carcinogens, such as nickel,
asbestos, and cigarette smoke, as well as possible healthy worker
effects and exposure misclassification in some studies, which may
obscure a relationship betweeen Cr(VI) and lung cancer risk. These
limitations are discussed further in sections VI.B.5, VI.E.3, and
VI.G.4.
Nevertheless, these studies add some further support to the much
stronger link between Cr(VI) and lung cancer found in soluble chromate
production workers, chromate pigment production workers, and chrome
platers. The key studies are summarized in Table V-4.
BILLING CODE 4510-26-P


BILLING CODE 4510-26-C
Sjogren et al. reported on the mortality experience in two cohorts
of welders (Ex. 7-95). The cohort characterized as "high exposure"
consisted of 234 male stainless steel welders with a minimum of 5 years
of employment between 1950 and 1965. An additional criterion for
inclusion in the study was assurance from the employer that asbestos
had not been used or had been used only occasionally and never in a
dust-generating way. The cohort characterized as "low exposure"
consisted of 208 male railway track welders working at the Swedish
State Railways for at least 5 years between 1950 and 1965. In 1975, air
pollution in stainless steel welding was surveyed in Sweden. The median
time weighted average (TWA) value for Cr(VI) was 110 [mu]g
CrO3/m\3\ (57 [mu]g/m\3\ measured as CrVI). The highest
concentration was 750 [mu]g CrO3/m\3\ (390 [mu]g/m\3\
measured as CrVI) found in welding involving coated electrodes. For
gas-shielded welding, the median Cr(VI) concentration was 10 [mu]g
CrO3/m\3\ (5.2 [mu]g/m\3\ measured as CrVI) with the highest
concentration measured at 440 [mu]g CrO3/m\3\ (229 [mu]g/
m\3\ measured as CrVI). Follow-up for both cohorts was through December
1984. The expected number of deaths was based upon Swedish male death
rates. Of the 32 deaths in the "high exposure" group, five were
cancers of the trachea, bronchus and lung (E=2.0; SMR=249; 95% CI:
0.80-5.81). In the low exposure group, 47 deaths occurred, one from
cancer of the trachea, bronchus and lung.
Polednak compiled a cohort of 1,340 white male welders who worked
at the Oak Ridge nuclear facilities from 1943 to 1977 (Ex. 277). One
thousand fifty-nine cohort members were followed through 1974. The
cohort was divided into two groups. The first group included 536
welders at a facility where nickel-alloy pipes were welded; the second
group included 523 welders of mild steel, stainless steel and aluminum
materials. Smoking data were available for 33.6% of the total cohort.
Expectations were calculated based upon U.S. mortality rates for white
males. There were 17 lung cancer deaths in the total cohort (E=11.37;
SMR=150; 95% CI: 87-240). Seven of the lung cancer deaths occurred in
the group which routinely welded nickel-alloy materials (E=5.65;
SMR=124; 95% CI: 50-255) versus 10 lung cancer deaths in the "other"
welders (E=6.12; SMR=163; 95% CI: 78-300).
Becker et al. compiled a cohort of 1,213 stainless steel welders
and 1,688 turners from 25 German metal processing factories who had a
minimum of 6 months employment during the period 1950-1970 (Exs. 227;
250; 251). The data collected included the primary type of welding
(e.g., arc welding, gas-shielded welding, etc.) used by each person,
working conditions, average daily welding time and smoking status. The
most recent follow-up of the cohort was through 1995. Expected numbers
were developed using German mortality data. There were 268 deaths among
the welders and 446 deaths among the turners. An elevated, but non-
statistically significant, lung cancer SMR (O=28; E=23; SMR=121.5; 95%
CI: 80.7-175.6) was observed among the welders. There were 38 lung
cancer deaths among the turners with 38.6 expected, resulting in a SMR
slightly below unity. Seven deaths from cancer of the pleura (all
mesotheliomas) occurred among the welders with only 0.6 expected
(SMR=1,179.9; 95% CI: 473.1-2,430.5), compared to only one death from
cancer of the pleura among the turners, suggesting that the welders had
exposure to asbestos. Epidemiological studies have shown that asbestos
exposure is a primary cause of pleural mesotheliomas.
The International Agency for Research on Cancer (IARC) and the
World Health Organization (WHO) cosponsored a study on welders. IARC
and WHO compiled a cohort of 11,092 male welders from 135 companies in
nine European countries to investigate the relationship between the
different types of exposure occurring in stainless steel, mild steel
and shipyard welding and various cancer sites, especially lung cancer
(Ex. 7-114). Cohort entrance criteria varied by country. The expected
number of deaths was compiled using national mortality rates from the
WHO mortality data bank.
Results indicated the lung cancer deaths were statistically
significant in the total cohort (116 cases; E=86.81; SMR=134; 95% CI:
110-160). Cohort members were assigned to one of four subcohorts based
upon type of welding activity. While the lung cancer SMRs were elevated
for all of the subcohorts, the only statistically significant SMR was
for the mild steel-only welders (O=40; E=22.42; SMR=178; 95% CI: 127-
243). Results for the other subgroups were: shipyard welders (O=36;
E=28.62; SMR=126; 95% CI: 88-174); ever stainless steel welders (O=39;
E=30.52; SMR=128; 95% CI: 91-175); and predominantly stainless steel
welders (O=20; E=16.25; SMR=123; 95% CI: 75-190). When analyzed by
subcohort and time since first exposure, the SMRs increased over time
for every group except shipyard welders. For the predominantly
stainless steel welder subcohort, the trend to increase with time was
statistically significant (p < .05).
An analysis was conducted of lung cancer mortality in two stainless
steel welder subgroups (predominantly and ever) with a minimum of 5
years of employment. Cumulative Cr(VI) was computed from start of
exposure until 20 years prior to death. A lung cancer SMR of 170, based
upon 14 cases, was observed in the stainless steel ever subgroup for
those welders with >=0.5 mg-years/m\3\ Cr(VI) exposure; the lung cancer
SMR for those in the < 0.5 mg-years/m\3\ Cr(VI) exposure group was 123
(based upon seven cases). Neither SMR was statistically significant.
For the predominantly stainless steel welders, which is a subset of the
stainless steel ever subgroup, the corresponding SMRs were 167 (>=0.5
mg-years/m\3\ Cr(VI) exposure) based upon nine cases and 191 (< 0.5 mg-
years/m\3\ Cr(VI) exposure) based upon three cases. Neither SMR was
statistically significant.
In conjunction with the IARC/WHO welders study, Gerin et al.
reported the development of a welding process exposure matrix relating
13 combinations of welding processes and base metals used to average
exposure levels for total welding fumes, total chromium, Cr(VI) and
nickel (Ex. 7-120). Quantitative estimates were derived from the
literature supplemented by limited monitoring data taken in the 1970s
from only 8 of the 135 companies in the IARC/WHO mortality study. An
exposure history was constructed which included hire and termination
dates, the base metal welded (stainless steel or mild steel), the
welding process used and changes in exposure over time. When a detailed
welding history was not available for an individual, the average
company welding practice profile was used. In addition, descriptions of
activities, work force, welding processes and parameters, base metals
welded, types of electrodes or rods, types of confinement and presence
of local exhaust ventilation were obtained from the companies.
Cumulative dose estimates in mg/m\3\ years were generated for each
welder's profile (number of years and proportion of time in each
welding situation) by applying a welding process exposure matrix
associating average concentrations of welding fumes (mg/m\3\) to each
welding situation. The corresponding exposure level was multiplied by
length of employment and summed over the various employment periods
involving different welding situations. No dose response relationship
was seen for exposure to Cr(VI) for either those who were "ever
stainless steel welders" or those who were "predominantly stainless
steel welders". The authors note that if their exposure estimates are
correct, the study had the power to detect a significant result in the
high exposure group for Cr(VI). However, OSHA believes that there is
likely to be substantial exposure misclassification in this study, as
discussed further in section VI.G.4.
The IARC/WHO multicenter study is the sole attempt to undertake
even a semi-quantified exposure analysis of stainless steel welders'
potential exposure to nickel and Cr(VI) for <5 and >=0.5 mg-years/
m3 Cr(VI) exposures. The IARC/WHO investigators noted that
there was more than a twofold increase in SMRs between the long (>=20
years since first exposure) and short (< 20 years since first exposure)
observation groups for the predominantly stainless steel welders
"suggesting a relation of lung cancer mortality with the occupational
environment for this group" (Ex. 7-114, p. 152). The authors conclude
that the increase in lung cancer mortality does not appear to be
related to either duration of exposure or cumulative exposure to total
fume, chromium, Cr(VI) or nickel.
Moulin compiled a cohort of 2,721 French male welders and an
internal comparison group of 6,683 manual workers employed in 13
factories (including three shipyards) with a minimum of one year of
employment from 1975 to 1988 (Ex. 7-92). Three controls were selected
at random for each welder. Smoking data were abstracted from medical
records for 86.6% of welders and 86.5% of the controls. Smoking data
were incorporated in the lung cancer mortality analysis using methods
suggested by Axelson. Two hundred and three deaths were observed in the
welders and 527 in the comparison group. A non-statistically
significant increase was observed in the lung cancer SMR (O=19;
E=15.33; SMR=124; 95% CI: 0.75-1.94) for the welders. In the control
group, the lung cancer SMR was in deficit (O=44; E=46.72; SMR=94; 95%
CI: 0.68-1.26). The resulting relative risk was a non-significant 1.3.
There were three deaths from pleural cancer in the comparison group and
none in the welders, suggesting asbestos exposure in the comparison
group. The welders were divided into four subgroups (shipyard welders,
mild steel only welders, ever stainless steel welders and stainless
steel predominantly Cr(VI) welders). The highest lung cancer SMR was
for the mild steel welders O=9; SMR of 159). The lowest lung cancer
SMRs were for ever stainless steel welders (O=3; SMR= 92) and for
stainless steel predominantly Cr(VI) welders (O=2; SMR= 103). None of
the SMRs are statistically significant.
Hansen conducted a study of cancer incidence among 10,059 male
welders, stainless steel grinders and other metal workers from 79
Danish companies (Ex. 9-129). Cohort entrance criteria included: alive
on April 1, 1968; born before January 1, 1965; and employed for at
least 12 months between April 1, 1964 and December 31, 1984. Vital
status follow-up found 9,114 subjects alive, 812 dead and 133
emigrated. A questionnaire was sent to subjects and proxies for
decedents/emigrants in an attempt to obtain information about lifetime
occupational exposure, smoking and drinking habits. The overall
response rate was 83%. The authors stated that no major differences in
smoking habits were found between exposure groups with or without a
significant excess of lung cancer.
The expected number of cancers was based on age-adjusted national
cancer incidence rates from the Danish Cancer Registry. There were
statistically significantly elevated Standardized Incidence Ratios
(SIRs) for lung cancer in the welding (any kind) group (O=51; E=36.84;
SIR=138; 95% CI: 103-181) and in the mild steel only welders (O=28;
E=17.42; SIR=161; 95% CI: 107-233). The lung cancer SIR for mild steel
ever welders was 132 (O=46; E=34.75; 95% CI: 97-176); for stainless
steel ever welders 119 (O=23; E=19.39; 95% CI: 75-179) and for
stainless steel only welders 238 (O=5; E=2.10; 95% CI: 77-555).
Laurtitsen reported the results of a nested case-control conducted
in conjunction with the Hansen cancer incidence study discussed above
(Exs. 35-291; 9-129). Cases were defined as the 94 lung cancer deaths.
Controls were defined as anyone who was not a case, but excluded deaths
from respiratory diseases other than lung cancer (either as an
underlying or a contributing cause of death), deaths from "unknown
malignancies" and decedents who were younger than the youngest case.
There were 439 decedents eligible for use as controls.
The crude odds ratio (OR) for welding ever (yes/no) was 1.7 (95%
CI: 1.0-2.8). The crude OR for mild steel welding only was 1.3 (95% CI:
0.8-2.3) and for stainless steel welding only the crude OR was 1.3 (95%
CI: 0.3-4.3). When analyzed by number of years exposed, "ever"
stainless steel welding showed no relationship with increasing number
of years exposed. The highest odds ratio (2.9) was in the lowest
category (1-5 years) based upon seven deaths; the lowest odds ratio was
in the highest category (21+ years) based upon three deaths.
Kjuus et al. conducted a hospital-based case-control study of 176
male incident lung cancer cases and 186 controls (matched for age, +/-5
years) admitted to two county hospitals in southeast Norway during
1979-1983 (Ex. 7-72). Subjects were classified according to exposure
status of main occupation and number of years in each exposure category
and assigned into one of three exposure groups according to potential
exposure to respiratory carcinogens and other contaminants. A
statistically significantly elevated risk ratio for lung cancer
(adjusted for smoking) for the exposure factor "welding, stainless,
acid proof" of 3.3 (p< 0.05) was observed based upon 16 lung cancer
deaths. The unadjusted odds ratio is not statistically significant
(OR=2.8). However, the appropriateness of the analysis is questionable
since the exposure factors are not discrete (a case or a control may
appear in multiple exposure factors and therefore is being compared to
himself). In addition, the authors note that several exposure factors
were highly correlated and point out specifically that one-half of the
cases "exposed to either stainless steel welding fumes or fertilizers
also reported moderate to heavy asbestos exposure." When put into a
stepwise logistic regression model, exposure to stainless steel fumes,
which was initially statistically significant, loses its significance
when smoking and asbestos are first entered into the model.
Hull et al. conducted a case-control study of lung cancer in white
male welders aged 20-65 identified through the Los Angeles County tumor
registry (Southern California Cancer Surveillance Program) for the
period 1972 to 1987 (Ex. 35-243). Controls were welders 40 years of age
or older with non-pulmonary malignancies. Interviews were conducted to
obtain information about sociodemographic data, smoking history,
employment history and occupational exposures to specific welding
processes, metals welded, asbestos and confined space welding.
Interviews were completed for 90 (70%) of the 128 lung cancer cases and
116 (66%) of the controls. Analysis was conducted using 85 deceased
cases and 74 deceased controls after determining that the subject's
vital status influenced responses to questions concerning occupational
exposures. The crude odds ratio (ever vs. never exposed) for stainless
steel welding, based upon 34 cases, was 0.9 (95% CI: 0.3-1.4). For
manual metal arc welding on stainless steel, the crude odds ratio
was 1.3 (95% CI: 0.6-2.3) based upon 61 cases.
While the relative risk estimates in both cohort and case-control
of stainless steel welders are elevated, none are statistically
significant. However, when combined in two meta-analyses, a small but
statistically significant increase in lung cancer risk was reported.
Two meta-analyses of welders have been published. Moulin carried out a
meta-analysis of epidemiologic studies of lung cancer risk among
welders, taking into account the role of asbestos and smoking (Ex. 35-
285). Studies published between 1954 and 1994 were reviewed. The
inclusion criteria were clearly defined: only the most recent updates
of cohort studies were used and only the mortality data from mortality/
morbidity studies were included. Studies that did not provide the
information required by the meta-analysis were excluded.
Five welding categories were defined (shipyard welding, non-
shipyard welding, mild steel welding, stainless steel welding and all
or unspecified welding). The studies were assigned to a welding
category (or categories) based upon the descriptions provided in the
paper's study design section. The combined relative risks (odds ratios,
standardized mortality ratios, proportionate mortality ratios and
standardized incidence ratios) were calculated separately for the
population-based studies, case-control studies, and cohort studies, and
for all the studies combined.
Three case-control studies (Exs. 35-243; 7-120; 7-72) and two
cohort studies (Exs. 7-114; 35-277) were included in the stainless
steel welding portion of the meta-analysis. The combined relative risk
was 2.00 (O=87; 95% CI: 1.22-3.28) for the case-control studies and
1.23 (O=27; 95% CI: 0.82-1.85) for the cohort studies. When all five
studies were combined, the relative risk was 1.50 (O=114; 95% CI: 1.10-
2.05).
By contrast, the combined risk ratio for the case-control studies
of mild steel welders was 1.56 (O=58; 95% CI: 0.82-2.99) (Exs. 7-120;
35-243). For the cohort studies, the risk ratio was 1.49 (O=79; 95% CI:
1.15-1.93) (Exs. 35-270; 7-114). For the four studies combined, the
risk ratio was 1.50 (O=137; 95% CI: 1.18-191). The results for the
stainless steel welders and the mild steel welders are basically the
same.
The meta-analysis by Sjogren of exposure to stainless steel welding
fumes and lung cancer included studies published between 1984 and 1993,
which took smoking and potential asbestos exposure into account (Ex. 7-
113). Five studies met the author's inclusion criteria and were
included in the meta-analysis: two cohort studies, Moulin et al. (Ex.
35-283) and Sjogren et al. (Ex. 7-95); and three case-control studies,
Gerin, et al. (Ex. 7-120, Hansen et al. (Ex. 9-129) and Kjuus et al.
(Ex. 7-72). The calculated pooled relative risk for welders exposed to
stainless steel welding fumes was 1.94 (95% CI: 1.28-2.93).
5. Evidence from Ferrochromium Workers
Ferrochromium is produced by the electrothermal reduction of
chromite ore with coke in the presence of iron in electric furnaces.
Some of the chromite ore is oxidized into Cr(VI) during the process.
However, most of the ore is reduced to chrome metal. The manufacture of
ferroalloys results in a complex mixture of particles, fumes and
chemicals including nickel, Cr(III) and Cr(VI). Polycyclic aromatic
hydrocarbons (PAH) are released during the manufacturing process. The
co-exposure to other potential lung carcinogens combined with the lack
of a statistically significant elevation in lung cancer mortality among
ferrochromium workers were limitations in the key studies.
Nevertheless, the observed increase in the relative risks of lung
cancer add some further support to the much stronger link between
Cr(VI) and lung cancer found in soluble chromate production workers,
chromate pigment production workers, and chrome platers. The key
studies are summarized in Table V-5.
BILLING CODE 4510-26-P

BILLING CODE 4510-26-C
Langard et al. conducted a cohort study of male workers producing
ferrosilicon and ferrochromium for more than one year between 1928 and
1977 at a plant located on the west coast of Norway (Exs. 7-34; 7-37). The
cohort and study findings are summarized in Table V.5. Excluded from
the study were workers who died before January 1, 1953 or had an
unknown date of birth. The cohort was defined in the 1980 study as 976
male employees who worked for a minimum of one year prior to January 1,
1960. In the 1990 study, the cohort definition was expanded to include
those hired up to 1965.
Production of ferrosilicon at the plant began in 1928 and
ferrochromium production began in 1932. Job characterizations were
compiled by combining information from company personnel lists and
occupational histories contained in medical records and supplemented
with information obtained via interview with long-term employees. Ten
occupational categories were defined. Workers were assigned to an
occupational category based upon the longest time in a given category.
Industrial hygiene studies of the plant from 1975 indicated that
both Cr(III) and Cr(VI) were present in the working environment. The
ferrochromium furnance operators were exposed to measurements of 0.04-
0.29 mg/m3 of total chromium. At the charge floor the mean
concentration of total chromium was 0.05 mg/m3, 11-33% of
which was water soluble. The water soluble chromium was considered to
be in the hexavalent state.
Both observed and expected cases of cancer were obtained via the
Norwegian Cancer Registry. The observation period for cancer incidence
was January 1, 1953 to December 31, 1985. Seventeen incident lung
cancers were reported in the 1990 study (E=19.4; SIR=88). A deficit of
lung cancer incidence was observed in the ferrosilicon group (O=2;
E=5.8; SIR=35). In the ferrochromium group there were a significant
excess of lung cancer; 10 observed lung cancers with 6.5 expected
(SIR=154).
Axelsson et al. conducted a study of 1,932 ferrochromium workers to
examine whether exposure in the ferrochromium industry could be
associated with an increased risk of developing tumors, especially lung
cancer (Ex. 7-62). The study cohort and findings are summarized in
Table V.5. The study cohort was defined as males employed at a
ferrochromium plant in Sweden for at least one year during the period
January 1, 1930 to December 31, 1975.
The different working sites within the industry were classified
into four groups with respect to exposure to Cr(VI) and Cr(III).
Exposure was primarily to metallic and trivalent chromium with
estimated levels ranging from 0-2.5 mg/m3. Cr(VI) was also
present in certain operations with estimated levels ranging from 0-0.25
mg/m3. The highest exposure to Cr(VI) was in the arc-furnace
operations. Cr(VI) exposure also occurred in a chromate reduction
process during chromium alum production from 1950-1956. Asbestos-
containing materials had been used in the plant. Cohort members were
classified according to length and place of work in the plant.
Death certificates were obtained and coded to the revision of the
International Classification of Diseases in effect at the time of
death. Data on cancer incidence were obtained from the Swedish National
Cancer Registry. Causes of death in the cohort for the period 1951-1975
were compared with causes of death for the age-adjusted male population
in the county in which the plant was located.
There were seven cases of cancers of the trachea, bronchus and lung
and the pleura with 5.9 expected (SIR=119) for the period 1958-1975.
Four of the seven cases in the lung cancer group were maintenance
workers and two of the four cases were pleural mesotheliomas. In the
arc furnace group, which was thought to have the highest potential
exposure to both Cr(III) and Cr(VI), there were two cancers of the
trachea, bronchus and lung and the pleura. One of the cases was a
mesothelioma. Of the 380 deaths that occurred during the period 1951-
1975, five were from cancer of the trachea, bronchus and lung and the
pleura (E=7.2; SMR=70). For the "highly" exposed furnace workers,
there was one death from cancer of the trachea, bronchus and lung and
the pleura.
Moulin et al. conducted a cohort mortality study in a French
ferrochromium/stainless steel plant to determine if exposure to
chromium compounds, nickel compounds and polycyclic aromatic
hydrocarbons (PAHs) results in an increased risk of lung cancer (Ex.
282). The cohort was defined as men employed for at least one year
between January 1, 1952 and December 31, 1982; 2,269 men met the cohort
entrance criteria. No quantitative exposure data were available and no
information on the relative amounts of Cr(VI) and Cr(III) was provided.
In addition, some workers were also exposed to other carcinogens, such
as silica and asbestos. The authors estimated that 75.7% of the cohort
had been exposed to combinations of PAH, nickel and chromium compounds.
Of the 137 deaths identified, the authors determined 12 were due to
cancer of the trachea, bronchus and lung (E=8.56; SMR=140; 95% CI:
0.72-2.45). Eleven of the 12 lung cancers were in workers employed for
at least one year in the ferrochromium or stainless steel production
workshops (E=5.4; SMR=204; 95% CI: 1.02-3.64).
Pokrovskaya and Shabynina conducted a cohort mortality study of
male and female workers employed "some time" between 1955 and 1969 at
a chromium ferroalloy production plant in the U.S.S.R (Ex. 7-61).
Workers were exposed to both Cr(III) and Cr(VI) as well as to benzo [a]
pyrene. Neither the number of workers nor the number of cancer deaths
by site were provided. Death certificates were obtained and the deaths
were compared with municipal mortality rates by gender and 10 year age
groups. The investigators state that they were able to exclude those in
the comparison group who had chromium exposures in other industries.
The lung cancer SMR for male chromium ferroalloy workers was 440 in the
30-39 year old age group and 660 in the 50-59 year old age group
(p=0.001). There were no lung cancer deaths in the 40-49 and the 60-69
year old age groups. The data suggest that these ferrochromium workers
may have been had an excess risk of lung cancer.
The association between Cr(VI) exposure in ferrochromium workers
and the incidence of respiratory tract cancer these studies is
difficult to assess because of co-exposures to other potential
carcinogens (e.g., asbestos, PAHs, nickel, etc.), absence of a clear
exposure-response relationship and lack of information on smoking.
There is suggestive evidence of excess lung cancer mortality among
Cr(VI)-exposed ferrochromium workers in the Norwegian (Langard) cohort
when compared to a similar unexposed cohort of ferrosilicon workers.
However, there is little consistency for this finding in the Swedish
(Axelsson) or French (Moulin) cohorts.
6. Evidence From Workers in Other Industry Sectors
There are several other epidemiological studies that do not fit
into the five industry sectors previously reviewed. These include
worker cohorts in the aerospace industry, paint manufacture, and
leather tanning operations, among others. The two cohorts of aircraft
manufacturing workers are summarized in Table V-6. All of the cohorts
had some Cr(VI) exposure, but certain cohorts may have included a
sizable number of workers with little or no exposure to Cr(VI). This
creates an additional complexity in assessing whether the study
findings support a Cr(VI) etiology for cancer of the respiratory system.
BILLING CODE 4510-26-P

BILLING CODE 4510-26-C
Alexander et al. conducted a cohort study of 2,429 aerospace
workers with a minimum of six months of cumulative employment in jobs
involving chromate exposure during the period 1974 through 1994 (Ex. 31-16-3).
Exposure estimates were based on industrial hygiene measurements and work
history records. Jobs were classified into categories of "high"
(spray painters, decorative painters), "moderate" (sanders/maskers,
maintenance painters) and "low" (chrome platers, surface processors,
tank tenders, polishers, paint mixers) exposure. Each exposure category
was assigned a summary TWA exposure based upon the weighted TWAs and
information from industrial hygienists. The use of respiratory
protection was accounted for in setting up the job exposure matrix. The
index of cumulative total chromium exposure (reported as [mu]g/m\3\
chromate TWA-years) was computed by multiplying the years in each job
by the summary TWAs for each exposure category.
In addition to cumulative chromate exposure, chromate exposure jobs
were classified according to the species of chromate. According to the
authors, in painting operations the exposure is to chromate pigments
with moderate and low solubility such as zinc chromate, strontium
chromate and lead chromate; in sanding and polishing operations the
same chromate pigments exist as dust; while platers and tank tenders
are exposed to chromium trioxide, which is highly soluble.
Approximately 26% of the cohort was lost to follow-up. Follow-up on
the cohort was short (average 8.9 years per cohort member). Cases were
identified through the Cancer Surveillance System (CSS) at the Fred
Hutchinson Cancer Research Center in Seattle, Washington. CSS records
primary cancer diagnoses in 13 counties in western Washington. Expected
numbers were calculated using race-, gender-, age- and calendar-
specific rates from the Puget Sound reference population for 1974
through 1994. Fifteen lung cancer cases were identified with an overall
standardized incidence ratio (SIR) of 80 (95% CI: 0.4-1.3). The SIRs
for lung cancer by cumulative years of employment in the "high
exposure" painting job category were based upon only three deaths in
each of the cumulative years categories (<5 and >=5); years of
employment was inversely related to the risk of lung cancer. For those
in the "low exposure" category, the SIRs were 130 for those who
worked less than five years in that category (95% CI: 0.2-4.8) and 190
for those who worked five years or more (95% CI: 0.2-6.9). However,
there were only two deaths in each category. The SIR for those who
worked >=5 years was 270 (95% CI: 0.5-7.8), but based only on three
deaths.
Boice et al. conducted a cohort mortality study of 77,965 workers
employed for a minimum of one year on or after January 1960 in aircraft
manufacturing (Ex. 31-16-4). Routine exposures to Cr(VI) compounds
occurred primarily while operating plating and coating process
equipment or when using chromate based primers or paints. According to
the authors, 3,634 workers, or 8% of the cohort, had the potential for
routine exposure to chromate and 3,809 workers, or 8.4%, had the
potential for intermittent exposure to chromate. Limited chromate air
sampling was conducted between 1978 and 1991. The mean full shift air
measurement was 1.5 [mu]g CrO3/m\3\ (0.78 [mu]g Cr(VI)/m\3\)
indicating fairly low airborne Cr(VI) in the plant (Ex. 47-19-5).
Follow up of the cohort was through 1996. Expectations were
calculated based on the general population of California for white
workers, while general population rates for the U.S. were used for non-
white workers. For the 3,634 cohort members who had potential for
routine exposure to chromates, the lung cancer SMR (race and gender
combined) was 102 based upon 87 deaths (95% CI: 82-126). There was a
slight non-significant positive trend (p value >2.0) for lung cancer
with duration of potential exposure. The SMR was 108 (95% CI: 75-157)
for workers exposed to chromate for >=5 years. Among the painters,
there were 41 deaths from lung cancer yielding a SMR of 111 (95% CI:
80-151). For those who worked as a process operator or plater the SMR
for lung cancer was 103 based upon 38 deaths (95% CI: 73-141).
OSHA believes the Alexander (Ex. 31-16-3) and the Boice et al. (Ex.
31-16-4) studies have several limitations. The Alexander cohort has few
lung cancers (due in part to the young age of the population) and lacks
smoking data. The authors note that these factors "[limit] the overall
power of the study and the stability of the risk estimates, especially
in exposure-related subanalyses" (Ex. 31-16-3, p. 1256). Another
limitation of the study is the 26.3% of cohort members lost to follow-
up. Boice et al. is a large study of workers in the aircraft
manufacturing industry, but was limited by a lack of Cr(VI) exposure
measurement during the 1960s and most of the 1970s. I was also limited
by a substantial healthy worker survivor effect that may have masked
evidence of excess lung cancer mortality in Cr(VI) exposed workers (Ex.
31-16-4). These studies are discussed further in section VI, including
section VI.B.6 (Alexander cohort) and section VI.G.4.a (Alexander and
Boice cohorts).
Dalager et al. conducted a proportionate mortality study of 977
white male spray painters potentially exposed to zinc chromate in the
aircraft maintenance industry who worked at least three months and
terminated employment within ten years prior to July 31, 1959 (Ex. 7-
64). Follow-up was through 1977. The expected numbers of deaths were
obtained by applying the cause-specific proportionate mortality of U.S.
white males to the total numbers of deaths in the study group by five
year age groups and five year time intervals. Two hundred and two
deaths were observed. There were 21 deaths from cancer of the
respiratory system (PMR=184), which was statistically significant. The
Proportionate Cancer Mortality Ratio for cancer of the respiratory
system was not statistically significant (PCMR= 146). Duration of
employment as a painter with the military as indicated on the service
record was used as an estimate of exposure to zinc chromate pigments,
which were used as a metal primer. The PMRs increased as duration of
employment increased (< 5 years, O=9, E=6.4, PMR=141; 5-9 years, O=6,
E=3, PMR=200; and 10+ years, O=6, E=2, PMR=300) and were statistically
significant for those who worked 10 or more years.
Bertazzi et al. studied the mortality experience of 427 workers
employed for a minimum of six months between 1946 and 1977 in a plant
manufacturing paint and coatings (Ex. 7-65). According to the author,
chromate pigments represented the "major exposure" in the plant. The
mortality follow-up period was 1954-1978. There were eight deaths from
lung cancer resulting in a SMR of 227 on the local standard (95% CI:
156-633) and a SMR of 334 on the national standard (95% CI: 106-434).
The authors were unable to differentiate between exposures to different
paints and coatings. In addition, asbestos was used in the plant and
may be a potential confounding exposure.
Morgan conducted a cohort study of 16,243 men employed after
January 1, 1946 for at least one year in the manufacture of paint or
varnish (Ex. 8-4). Analysis was also conducted for seven subcohorts,
one of which was for work with pigments. Expectations were calculated
based upon the mortality experience of U.S. white males. The SMR for
cancer of the trachea, bronchus and lung was below unity based upon 150
deaths. For the pigment subcohort, the SMR for cancer of the trachea,
bronchus and lung was 117 based upon 43 deaths. In a follow-up study of
the subcohorts, case-control analyses were conducted for several causes
of death including lung cancer (Ex. 286). The details of matching were not
provided. The authors state that no significant excesses of lung cancer
risk by job were found. No odds ratios were presented.
Pippard et al. conducted a cohort mortality study of 833 British
male tannery workers employed in 1939 and followed through December 31,
1982 (Ex. 278). Five hundred and seventy three men worked in tanneries
making vegetable tanned leathers and 260 men worked in tanneries that
made chrome tanned leathers. The expected number of deaths was
calculated using the mortality rates of England and Wales as a whole.
The lung cancer SMR for the vegetable tanned leather workers was in
deficit (O=31; E=32.6; 95% CI: 65-135), while the lung cancer SMR for
the chrome tanned leather workers was slightly elevated but not
statistically significant (O=13; E=12; SMR=108; 95% CI: 58-185).
In a different study of two U.S. tanneries, Stern et al.
investigated mortality in a cohort of all production workers employed
from January 1, 1940 to June 11, 1979 at tannery A (N=2,807) and from
January 1, 1940 to May 1, 1980 at tannery B (N=6,558) (Ex. 7-68). Vital
status was followed through December 31, 1982. There were 1,582 deaths
among workers from the two tanneries. Analyses were conducted employing
both U.S. mortality rates and the mortality rates for the state in
which the plant is located. There were 18 lung/pleura cancer deaths at
tannery A and 42 lung/pleura cancer deaths at tannery B. The lung
cancer/pleura SMRs were in deficit on both the national standard and
the state standard for both tanneries. The authors noted that since the
1940s most chrome tanneries have switched to the one-bath tanning
method in which Cr(VI) is reduced to Cr(III).
Blot et al. reported the results of a cohort study of 51,899 male
workers of the Pacific Gas & Electric Company alive in January 1971 and
employed for at least six months before the end of 1986 (Ex. 239). A
subset of the workers were involved in gas generator plant operations
where Cr(VI) compounds were used in open and closed systems from the
1950s to early 1980s. One percent of the workers (513 men) had worked
in gas generator jobs, with 372 identified from post-1971 listing at
the company's three gas generator plants and 141 from gas generator job
codes. Six percent of the cohort members (3,283) had trained at one of
the gas generator plants (Kettleman).
SMRs based on national and California rates were computed. Results
in the paper are based on the California rates, since the overall
results reportedly did not differ substantially from those using the
national rates. SMRs were calculated for the entire cohort and for
subsets defined by potential for gas generator plant exposure. No
significant cancer excesses were observed and all but one cancer SMR
was in deficit. There were eight lung cancer deaths in the gas
generator workers (SMR=81; 95% CI: 0.35-1.60) and three lung cancer
deaths among the Kettleman trainees (SMR=57; 95% CI: 0.12-1.67). There
were no deaths from nasal cancer among either the gas generator workers
or the Kettleman trainees. The risk of lung cancer did not increase
with length of employment or time since hire.
Rafnsson and Johannesdottir conducted a study of 450 licensed
masons (cement finishers) in Iceland born between 1905 and 1945,
followed from 1951 through 1982 (Ex. 7-73). Stonecutters were excluded.
Expectations were based on the male population of Iceland. The SMR for
lung cancer was 314 and is statistically significant based upon nine
deaths (E=2.87; 95% CI: 1.43-5.95). When a 20 year latency was factored
into the analysis, the lung cancer SMR remained statistically
significant (O=8; E=2.19; SMR=365; 95% CI: 1.58-7.20).
Svensson et al. conducted a cohort mortality study of 1,164 male
grinding stainless steel workers employed for three months or more
during the period 1927-1981 (Ex.266). Workers at the facility were
reportedly exposed to chromium and nickel in the stainless steel
grinding process. Records provided by the company were used to assign
each worker to one of three occupational categories: those considered
to have high exposure to chromium, nickel as well as total dust, those
with intermediate exposure, and those with low exposure. Mortality
rates for males in Blekinge County, Sweden were used as the reference
population. Vital status follow-up was through December 31, 1983. A
total of 194 deaths were observed (SMR=91). No increased risk of lung
cancer was observed (SMR=92). The SMR for colon/rectum cancer was 2.47,
but was not statistically significant.
Cornell and Landis studied the mortality experience of 851 men who
worked in 26 U.S. nickel/chromium alloy foundries between 1968 and 1979
(Ex. 7-66). Standardized Proportionate Mortality Ratio (SPMR) analyses
were done using both an internal comparison group (foundry workers not
exposed to nickel/chromium) and the mortality experience of U.S. males.
The SPMR for lung cancer was 105 (O=60; E=56.9). No nasal cancer deaths
were observed.
Brinton et al. conducted a case-control study of 160 patients
diagnosed with primary malignancies of the nasal cavity and sinuses at
one of four hospitals in North Carolina and Virginia between January 1,
1970 and December 31, 1980 (Ex. 8-8). For each case determined to be
alive at the time of interview, two hospital controls were selected
matched on vital status, hospital, year of admission (2
years), age (5 years), race and state economic area or
county or usual residence. Excluded from control selection were
malignant neoplasms of the buccal cavity and pharynx, esophagus, nasal
cavity, middle ear and accessory sinuses, larynx, and secondary
neoplasms. Also excluded were benign neoplasms of the respiratory
system, mental disorders, acute sinusitis, chronic pharyngitis and
nasopharyngitis, chronic sinusitis, deflected nasal septum or nasal
polyps. For those cases who were deceased at the time of interview, two
different controls were selected. One control series consisted of
hospital controls as described previously. The second series consisted
of decedents identified through state vital statistics offices matched
for age (5 years), sex, race, county of usual residence and
year of death. A total of 193 cases were identified and 160 case
interviews completed. For those exposed to chromates, the relative risk
was not significantly elevated (OR=5.1) based upon five cases.
According to the authors, chromate exposure was due to the use of
chromate products in the building industry and in painting, rather than
the manufacture of chromates.
Hernberg et al. reported the results of a case-control study of 167
living cases of nasal or paranasal sinus cancer diagnosed in Denmark,
Finland and Sweden between July 1, 1977 and December 31, 1980 (Exs. 8-
7; 7-71). Controls were living patients diagnosed with malignant tumors
of the colon and rectum matched for country, gender and age at
diagnosis (3 years) with the cases. Both cases and controls
were interviewed by telephone to obtain occupational histories.
Patients with work-related exposures during the ten years prior to
their illness were excluded. Sixteen cases reported exposure to
chromium, primarily in the "stainless steel welding" and "nickel"
categories, versus six controls (OR=2.7l; 95% CI: 1.1-6.6).
7. Evidence From Experimental Animal Studies
Most of the key animal cancer bioassays for chromium compounds were
conducted before 1988. These studies have been critically reviewed by the
IARC in the Monograph Chromium, Nickel, and Welding (Ex. 35-43). OSHA
reviewed the key animal cancer bioassays in the NPRM (69 FR at 59341-59347)
and requested any additional data in experimental animals that were considered
important to evaluating the carcinogenicity of Cr(VI). The discussion below
describes these studies along with any new study information received
during the public hearing and comment periods.
In the experimental studies, Cr(VI) compounds were administered by
various routes including inhalation, intratracheal instillation,
intrabronchial implantation, and intrapleural injection, as well as
intramuscular and subcutaneous injection. For assessing human health
effects from occupational exposure, the most relevant route is
inhalation. However, as a whole, there were very few inhalation
studies. In addition to inhalation studies, OSHA is also relying on
intrabronchial implantation and intratracheal instillation studies for
hazard identification because these studies examine effects directly
administered to the respiratory tract, the primary target organ of
concern, and they give insight into the relative potency of different
Cr(VI) compounds. In comparison to studies examining inhalation,
intrabronchial implantation, and intratracheal instillation, studies
using subcutaneous injection and intramuscular administration of Cr(VI)
compounds were of lesser significance but were still considered for
hazard identification.
In its evaluation, OSHA took into consideration the exposure
regimen and experimental conditions under which the experiments were
performed, including the exposure level and duration; route of
administration; number, species, strain, gender, and age of the
experimental animals; the inclusion of appropriate control groups; and
consistency in test results. Some studies were not included if they did
not contribute to the weight of evidence, lacked adequate
documentation, were of poor quality, or were less relevant to
occupational exposure conditions (e.g., some intramuscular injection
studies).
The summarized animal studies are organized by Cr(VI) compound in
order of water solubility as defined in section IV on Chemical
Properties (i.e., Cr(VI) compounds that are highly soluble in water;
Cr(VI) compounds that are slightly soluble in water, and Cr(VI)
compounds that insoluble in water). Solubility is an important factor
in determining the carcinogenicity of Cr(VI) compounds (Ex. 35-47).
a. Highly Water Soluble Cr(VI) Compounds
Multiple animal carcinogenicity studies have been conducted on
highly water soluble sodium dichromate and chromic acid. The key
studies are summarized in Table V-7.
BILLING CODE 4510-26-P

BILLING CODE 4510-26-C
Chromic acid (Chromium trioxide). In a study by Adachi et al., ICR/JcI
mice were exposed by inhalation to 3.63 mg/m\3\ for 30 minutes per day,
two days per week for up to 12 months (Ex. 35-26-1). The mice were
observed for an additional six months. The authors used a miniaturized
chromium electroplating system to generate chromic acid for the study.
The authors found there were elevations in lung adenomas at 10-14
months (3/14 vs. 0/10) and lung adenocarcinomas at 15-18 months (2/19
vs. 0/10), but the results were not statistically significant. The
small number of animals (e.g. 10-20 per group) used in this study
limited its power to detect all but a relatively high tumor incidence
(e.g. >20%) with statistical precision. Statistically significant
increases in nasal papillomas were observed in another study by Adachi
et al., in which C57B1 mice were exposed by inhalation to 1.81 mg/m\3\
chromic acid for 120 min per day, two days per week for up to 12 months
(Ex. 35-26). At 18 months, the tumor incidence was 6/20 in exposed
animals vs. 0/20 in the control animals (p< 0.05).
In separate but similar studies, Levy et al. and Levy and Venitt,
using similar exposure protocol, conducted bronchial implantation
experiments in which 100 male and female Porton-Wistar rats were dosed
with single intrabronchial implantations of 2 mg chromic acid (1.04 mg
Cr(VI)) mixed 50:50 with cholesterol in stainless steel mesh pellets
(Exs. 11-2; 11-12). The authors found no statistically significant
increases in lung tumors, although Levy et al. found a bronchial
carcinoma incidence of 2/100 in exposed rats compared with 0/100 in
control rats. Levy and Venitt found a bronchial carcinoma incidence of
1/100 accompanied by a statistically significant increase in squamous
metaplasia, a lesion believed capable of progressing to carcinoma.
There was no statistically significant increase in the incidence of
squamous metaplasia in control rats or rats treated with Cr(III)
compounds in the same study. This finding suggests that squamous
metaplasia is specific to Cr(VI) and is not evoked by a non-specific
stimuli, the implantation procedure itself, or treatment with Cr(III)
containing materials.
Similar to Levy et al. and Levy and Venitt studies, Laskin et al.
gave a single intrabronchial implantation of 3-5 mg chromic acid mixed
50:50 with cholesterol in stainless steel mesh pellets to 100 male and
female Porton-Wistar rats (Ex. 10-1). The rats were observed for 2
years. No tumors were identified in the treated or control animals (0/
100 vs. 0/24).
Sodium dichromate. Glaser et al. exposed male Wistar rats to
aerosolized sodium dichromate by inhalation for 22-23 hours per day,
seven days per week for 18 months (Exs. 10-10; 10-11). The rats were
held for an additional 12 months at which point the study was
terminated. Lung tumor incidences among groups exposed to 25, 50, and
100 [mu]g Cr(VI)/m\3\ were 0/18, 0/18, and 3/19, respectively, vs. 0/37
for the control animals. Histopathology revealed one adenocarcinoma and
two adenomas in the highest group. The slightly elevated tumor
incidence at the highest dose was not statistically significant. A
small number of animals (20 per group) were used in this study limiting
its power to detect all but a relatively high tumor incidence (e.g.
>20%) with statistical precision. In addition, the administered doses
used in this study were fairly low, such that the maximum tolerated
dose (i.e., the maximum dose level that does not lead to moderate
reduction in body weight gain) may not have been achieved. Together,
these factors limit the interpretation of the study.
In an analysis prepared by Exponent and submitted by the Chrome
Coalition, Exponent stated that "inhalation studies of Glaser et al.
support a position that exposures to soluble Cr(VI) at concentrations
at least as high as the current PEL (i.e., 52 [mu]g/m\3\) do not cause
lung cancer" (Ex. 31-18-1, page 2). However, it should be noted that
the Glaser et al. studies found that 15% (\3/19\) of the rats exposed
to an air concentration just above the current PEL developed lung
tumors, and that the elevated tumor incidence was not statistically
significant in the highest dose group because the study used a small
number of animals. OSHA believes the Glaser study lacks the statistical
power to state with sufficient confidence that Cr(VI) exposure does not
cause lung cancer at the current PEL, especially when given the
elevated incidence of lung tumors at the next highest dose level.
Steinhoff et al. studied the carcinogenicity of sodium dichromate
in Sprague-Dawley rats (Ex. 11-7). Forty male and 40 female Sprague-
Dawley rats were divided into two sets of treatment groups. In the
first set, doses of 0.01, 0.05 or 0.25 mg/kg body weight in 0.9% saline
were instilled intratracheally five times per week. In the second set
of treatment groups, 0.05, 0.25 or 1.25 mg/kg body weight in 0.9%
saline doses were instilled intratracheally once per week. Duration of
exposure in both treatment groups was 30 months. The total cumulative
dose for the lowest treatment group of animals treated once per week
was the same as the lowest treatment group treated five times per week.
Similarly, the medium and high dose groups treated once per week had
total doses equivalent to the medium and high dose animals treated five
times per week, respectively. No increased incidence of lung tumors was
observed in the animals dosed five times weekly. However, in the
animals dosed once per week, tumor incidences were 0/80 in control
animals, 0/80 in the 0.05 mg/kg exposure group, 1/80 in the 0.25 mg/kg
exposure group and 14/80 in the 1.25 mg/kg exposure group (p < 0.01).
The tumors were malignant in 12 of the 14 animals in the 1.25 mg/kg
exposure group. Tracheal instillation at the highest dose level (i.e.
1.25 mg/kg) caused emphysematous lesions and pulmonary fibrosis in the
lungs of Cr(VI)-treated rats. A similar degree of lung damage did not
occur at the lower dose levels. Exponent commented that the Steinhoff
and Glaser results are evidence that the risk of lung cancer from
occupational exposure does not exist below a threshold Cr(VI) air
concentration of approximately 20 [mu]g/m\3\ (Ex. 38-233-4). This
comment is addressed in Section VI.G.2.c.
In separate but similar studies, Levy et al. and Levy and Venitt
implanted stainless steel mesh pellets filled with a single dose of 2
mg sodium dichromate (0.80 mg Cr(VI)) mixed 50:50 with cholesterol in
the bronchi of male and female Porton-Wistar rats (Exs. 11-2; 11-12).
Control groups (males and females) received blank pellets or pellets
loaded with cholesterol. The rats were observed for two years. Levy et
al. and Levy and Venitt reported a bronchial tumor incidence of 1/100
and 0/89, respectively, for exposed rats. However, the latter study
reported a statistically significant increase in squamous metaplasia, a
lesion believed capable of progressing to carcinoma, among exposed rats
when compared to unexposed rats. There were no bronchial tumors or
squamous metaplasia in any of the control animals and no significant
increases in lung tumors were observed in the two studies.
b. Slightly Water Soluble Cr(VI) Compounds
Animal carcinogenicity studies have been conducted on slightly
water soluble calcium chromate, strontium chromate, and zinc chromates.
The key studies are summarized in Table V-8.
BILLING CODE 4510-26-P

BILLING CODE 4510-26-C
Calcium chromate. Nettesheim et al. conducted the only available
inhalation carcinogenicity study with calcium chromate showing
borderline statistical significance for increased lung adenomas in
C57B1/6 mice exposed to 13 mg/m\3\ for 5 hours per day, 5 days per week
over the life of the mice. The tumor incidences were 6/136 in exposed
male mice vs. 3/136 in control male mice and 8/136 in exposed female
mice vs. 2/136 in control female mice (Ex. 10-8).
Steinhoff et al. observed a statistically significant increase in
lung tumors in Sprague-Dawley rats exposed by intratracheal
instillation to 0.25 mg/kg body weight calcium chromate in 0.9% saline
five times weekly for 30 months (Ex. 11-7). Tumors were found in 6/80
exposed animals vs. 0/80 in unexposed controls (p< 0.01). Increased
incidence of lung tumors was also observed in those rats exposed to
1.25 mg/kg calcium chromate once per week (14/80 vs. 0/80 in controls)
for 30 months. At the highest dose, the authors observed 11 adenomas,
one adenocarcinoma, and two squamous carcinomas. The total administered
doses for both groups of dosed animals (1 x 1.25 mg/kg and 5 x 0.25 mg/
kg) were equal, but the tumor incidence in the rats exposed once per
week was approximately double the incidence in rats exposed to the same
weekly dose divided into five smaller doses. The authors suggested that
the dose-rate for calcium chromate compounds may be important in
determining carcinogenic potency and that limiting higher single
exposures may offer greater protection against carcinogenicity than
reducing the average exposure alone.
Snyder et al. administered Cr(VI)-contaminated soil of defined
aerodynamic diameter (2.9 to 3.64 micron) intratracheally to male
Sprague-Dawley rats (Ex. 31-18-12). For the first six weeks of
treatment, the rats were instilled with weekly suspensions of 1.25 mg
of material per kg body weight, followed by 2.5 mg/kg every other week,
until treatments were terminated after 44 weeks. The investigation
included four exposure groups: control animals (50 rats), rats
administered Cr(VI)-contaminated soil (50 rats), rats administered
Cr(VI)-contaminated soil supplemented with calcium chromate (100 rats),
and rats administered calcium chromate alone (100 rats). The total
Cr(VI) dose for each group was: control group (0.000002 mg Cr(VI)/kg),
soil alone group (0.324 mg Cr(VI)/kg), soil plus calcium chromate group
(7.97 mg Cr(VI)/kg), and calcium chromate alone group (8.70 mg Cr(VI)/
kg). No primary tumors were observed in the control group or the
chromium contaminated soil group. Four primary tumors of the lung were
found in the soil plus calcium chromate group and one primary lung
tumor was observed in the group treated with calcium chromate alone;
however, these incidences did not reach statistical significance.
Statistically significant increases in the incidence of bronchial
carcinoma in rats exposed to calcium chromate through intrabronchial
instillation were reported by Levy et al. (Ex. 11-2) and Levy and
Venitt (Ex. 11-12). These studies, using a similar protocol, implanted
a single dose of 2 mg calcium chromate (0.67 mg Cr(VI)) mixed 50:50
with cholesterol in stainless steel pellets into the bronchi of Porton-
Wistar rats. Levy et al. and Levy and Venitt found bronchial carcinoma
incidences of 25/100 and 8/84, respectively, following a 24-month
observation. The increased incidences were statistically significant
when compared to the control group. Levy and Venitt also reported
statistically significant increases in squamous metaplasia in the
calcium chromate-treated rats (Ex. 11-12).
Laskin et al. observed 8/100 tumors in rats exposed to a single
dose of 3-5 mg calcium chromate mixed with cholesterol in stainless
steel mesh pellets implanted in the bronchi (Ex. 10-1). Animals were
observed for a total of 136 weeks. The sex, strain, and species of the
rats were not specified in the study. Tumor incidence in control
animals was 0/24. Although tumor incidence did not reach statistical
significance in this study, OSHA agrees with the IARC evaluation that
the incidences are due to calcium chromate itself rather than
background variation.
Strontium chromate. Strontium chromate was tested by intrabronchial
implantation and intrapleural injection. In a study by Levy et al., two
strontium chromate compounds mixed 50:50 with cholesterol in stainless
steel mesh pellets were administered by intrabronchial instillation of
a 2 mg (0.48 mg Cr(VI)) dose into 100 male and female Porton-Wistar
rats (Ex. 11-2). Animals were observed for up to 136 weeks. The
strontium chromate compounds induced bronchial carcinomas in 43/99 (Sr,
42.2%; CrO4, 54.1%) and 62/99 rats (Sr, 43.0%; Cr, 24.3%)],
respectively, compared to 0/100 in the control group. These results
were statistically significant. The strontium chromates produced the
strongest carcinogenic response out of the 20 Cr(VI) compounds tested
by the intrabronchial implantation protocol. Boeing Corporation
commented that the intrabronchial implantation results with strontium
chromate should not be relied upon in an evaluation of carcinogenicity
and that the data is inconsistent with other Cr(VI) studies (Ex. 38-
106-2, p. 26). This comment is discussed in the Carcinogenic Effects
Conclusion Section V.B.9 dealing with the carcinogenicity of slightly
soluble Cr(VI) compounds.
In the study by Hueper, strontium chromate was administered by
intrapleural injection (doses unspecified) lasting 27 months (Ex. 10-
4). Local tumors were observed in 17/28 treated rats vs. 0/34 for the
untreated rats. Although the authors did not examine the statistical
significance of tumors, the results clearly indicate a statistical
significance.
Zinc chromate compounds. Animal studies have been conducted to
examine several zinc chromates of varying water solubilities and
composition. In separate, but similarly conducted studies, Levy et al.
and Levy and Venitt studied two zinc chromate powders, zinc potassium
chromate, and zinc tetroxychromate (Exs. 11-2; 11-12). Two milligrams
of the compounds were administered by intrabronchial implantation to
100 male and female Porton-Wistar rats. Zinc potassium chromate (0.52
mg Cr(VI)) produced a bronchial tumor incidence of 3/61 which was
statistically significant (p< 0.05) when compared to a control group
(Ex. 11-12). There was also an increased incidence of bronchial tumors
(5/100, p=0.04; 3/100, p=0.068) in rats receiving the zinc chromate
powders (0.44 mg Cr(VI)). Zinc tetroxychromate (0.18 mg Cr(VI)) did not
produce a statistically significant increase in tumor incidence (1/100)
when compared to a control group. These studies show that most slightly
water soluble zinc chromate compounds elevated incidences of tumors in
rats.
Basic potassium zinc chromate was administered to mice, guinea pigs
and rabbits via intratracheal instillation (Ex. 35-46). Sixty-two
Strain A mice were given six injections of 0.03 ml of a 0.2% saline
suspension of the zinc chromate at six week intervals and observed
until death. A statistically significant increase in tumor incidence
was observed in exposed animals when compared to controls (31/62 vs. 7/
18). Statistically significant effects were not observed among guinea
pigs or rabbits. Twenty-one guinea pigs (sex and strain not given)
received six injections of 0.3 ml of a 1% suspension of zinc chromate
at three monthly intervals and observed until death. Results showed
pulmonary adenomas in only 1/21 exposed animals vs. 0/18 in controls.
Seven rabbits (sex and strain not given) showed no increase in lung
tumors when given 3-5 injections of 1 ml of a saline suspension of 10
mg zinc chromate at 3-month intervals. However, as noted by IARC, the
small numbers of animals used in the guinea pig and rabbit experiments
(as few as 13 guinea pigs and 7 rabbits per group) limit the power of
the study to detect increases in cancer incidence.
Hueper found that intrapleural injection of slightly water soluble
zinc yellow (doses were unspecified) resulted in statistically significant
increases in local tumors in rats (sex, strain, and age of rat
unspecified; dose was unspecified). The incidence of tumors in exposed
rats was 22/33 vs. 0/34 in controls (Ex. 10-4).
Maltoni et al. observed increases in the incidence of local tumors
after subcutaneous injection of slightly water soluble zinc yellow in
20 male and 20 female Sprague-Dawley rats (statistical significance was
not evaluated) (Ex. 8-37). Tumor incidences were 6/40 in 20%
CrO3 dosed animals at 110 weeks and 17/40 in 40%
CrO3 dosed animals at 137 weeks compared to 0/40 in control
animals.
c. Water Insoluble Cr(VI) Compounds
There have been a number of animal carcinogenicity studies
involving implantation or injection of principally water insoluble
zinc, lead, and barium chromates. The key studies are summarized in
Table V-9.
BILLING CODE 4510-26-P

BILLING CODE 4510-26-C
Lead chromate and lead chromate pigments. Levy et al. examined the
carcinogenicity of lead chromate and several lead chromate-derived
pigments in 100 male and female Porton-Wistar rats after a single
intrabronchial implantation followed by a two year observation period
(Ex. 11-12). The rats were dosed with two mg of a lead chromate
compound and lead chromate pigments, which were mixed 50:50 with
cholesterol in stainless steel mesh pellets and implanted in the
bronchi of experimental animals. The lead chromate and lead chromate
pigment compositions consisted of the following: lead chromate (35.8%
CrO4; 0.32 mg Cr(VI)), primrose chrome yellow (12.6% Cr;
0.25 mg Cr(VI)), molybdate chrome orange (12.9% Cr; 0.26 mg Cr(VI)),
light chrome yellow (12.5% Cr; 0.25 mg Cr(VI)), supra LD chrome yellow
(26.9% CrO3; 0.28 mg Cr(VI)), medium chrome yellow (16.3%
Cr; 0.33 mg Cr(VI)) and silica encapsulated medium chrome yellow (10.5%
Cr; 0.21 mg Cr(VI)). No statistically significant tumors were observed
in the lead chromate group compared to controls (1/98 vs. 0/100),
primrose chrome yellow group (1/100 vs. 0/100), and supra LD chrome
yellow group (1/100 vs. 0/100). The authors also noted no tumors in the
molybdate chrome orange group, light chrome yellow group, and silica
encapsulated medium chrome yellow group.
Maltoni (Ex. 8-25), Maltoni (Ex. 5-2), and Maltoni et al. (Ex. 8-
37) examined the carcinogenicity of lead chromate, basic lead chromate
(chromium orange) and molybdenum orange in 20 male and 20 female
Sprague-Dawley rats by a single subcutaneous administration of the lead
chromate compound in water. Animals were observed for 117 to 150 weeks.
After injection of 30 mg lead chromate, local injection site sarcomas
were observed in 26/40 exposed animals vs. 0/60 and 1/80 in controls.
Although the authors did not examine the statistical significance of
sarcomas, the results clearly indicate a statistical significance.
Animals injected with 30 mg basic lead chromate (chromium orange) were
found to have an increased incidence of local injection site sarcomas
(27/40 vs. 0/60 and 1/80 in controls). Animals receiving 30 mg
molybdenum orange in 1 ml saline were also found to have an increased
incidence of local injection site sarcomas (36/40 vs. 0/60 controls).
Carcinogenesis was observed after intramuscular injection in a
study by Furst et al. (Ex. 10-2). Fifty male and female Fischer 344
rats were given intramuscular injections of 8 mg lead chromate in
trioctanoin every month for nine months and observed up to 24 months.
An increase in local tumors at the injection site (fibrosarcomas and
rhabdomyosarcomas) was observed (31/47 in treated animals vs. 0/22 in
controls). These rats also had an increased incidence of renal
carcinomas (3/23 vs. 0/22 in controls), but IARC noted that the renal
tumors may be related to the lead content of the compound. In the same
study, 3 mg lead chromate was administered to 25 female NISH Swiss
weanling mice via intramuscular injection every 4 months for up to 24
months. In the exposed group, the authors observed three lung
alveologenic carcinomas after 24 months of observation and two
lymphomas after 16 months of observation. Two control groups were used:
an untreated control group (22 rats) and a vehicle injected control
group (22 rats). The authors noted that one alveologenic carcinoma and
one lymphoma were observed in each control group. The Color Pigment
Manufacturers Association (CPMA) commented that the lack of elevated
tumor incidence in the intrabronchial implantation studies confirmed
that lead chromate was not carcinogenic and that the positive injection
studies by the subcutaneous, intrapleural, and intramuscular routes
were of questionable relevance (Ex. 38-205, p. 93). This comment is
further discussed in the Carcinogenic Effects Conclusion Section V.B.9
dealing with the carcinogenicity of lead chromate.
Barium chromate. Barium chromate was tested in rats via
intrabronchial, intrapleural and intramuscular administration. No
excess lung or local tumors were observed (Ex. 11-2; Ex. 10-4; Ex. 10-
6).
d. Summary. Several Cr(VI) compounds produced tumors in laboratory
animals under a variety of experimental conditions using different
routes of administration. The animals were generally given the test
material(s) by routes other than inhalation (e.g., intratracheal
administration, intramuscular injection, intrabronchial implantation,
and subcutaneous injection). Although the route of administration may
have differed from that found in an occupational setting, these studies
have value in the identification of potential health hazards associated
with Cr(VI) and in assessing the relative potencies of various Cr(VI)
compounds.
OSHA believes that the results from Adachi et al. (Ex. 35-26-1),
Adachi et al. (Ex. 35-26), Glaser et al. (Ex. 10-4), Glaser et al. (Ex.
10-10), Levy et al. (Ex. 11-2), and Steinhoff et al. (Ex. 11-7) studies
provide valuable insight on the carcinogenic potency of Cr(VI)
compounds in laboratory animals. Total dose administered, dose rate,
amount of dosage, dose per administration, number of times
administered, exposure duration and the type of Cr(VI) compound are
major influences on the observed tumor incidence in animals. It was
found that slightly water soluble calcium, strontium, and zinc
chromates showed the highest incidence of lung tumors, as indicated in
the results of the Steinhoff and Levy studies, even when compared to
similar doses of the more water soluble sodium chromates and chromic
acid compounds. The highly insoluble lead chromates did not produce
lung tumors by the intrabronchial implantation procedure but did
produce tumors by subcutaneous injection and intramuscular injection.
8. Mechanistic Considerations
Mechanistic information can provide insight into the biologically
active form(s) of chromium, its interaction with critical molecular
targets, and the resulting cellular responses that trigger neoplastic
transformation. There has been considerable scientific study in recent
years of Cr(VI)-initiated cellular and molecular events believed to
impact development of respiratory carcinogenesis. Much of the research
has been generated using in vitro techniques, cell culture systems, and
animal administrations. The early mechanistic data were reviewed by
IARC in 1990 (Ex. 35-43). Recent experimental research has identified
several biological steps critical to the mode of action by which Cr(VI)
transforms normal lung cells into a neoplastic phenotype. These are:
(a) Cellular uptake of Cr(VI) and its extracellular reduction, (b)
intracellular Cr(VI) reduction to produce biologically active products,
(c) damage to DNA, and (d) activation of signaling pathways in response
to cellular stress. Each step will be described in detail below.
a. Cellular Uptake and Extracellular Reduction. The ability of
different Cr(VI) particulate forms to be taken up by the
bronchoalveolar cells of the lung is an essential early step in the
carcinogenic process. Particle size and solubility are key physical
factors that influence uptake into these cells. Large particulates (>10
[mu]m) are generally deposited in the upper nasopharygeal region of the
respiratory tract and do not reach the bronchoalveolar region of the
lungs. Smaller Cr(VI) particulates will increasingly reach these lower
regions and come into contact with target cells.
Once deposited in the lower respiratory tract, solubility of Cr(VI)
particulates becomes a major influence on disposition. Highly water
soluble Cr(VI), such as sodium chromate and chromic acid, rapidly
dissolves in the fluids lining the lung epithelia and can be taken up
by lung cells via facilitated diffusion mediated by sulfate/phosphate
anion transport channels (Ex. 35-148). This is because Cr(VI) exists in
a tetrahedral configuration as a chromate oxyanion similar to the
physiological anions, sulfate and phosphate (Ex. 35-231). Using cultured
human epithelial cells, Liu et al. showed that soluble Cr(VI) uptake was
time- and dose-dependant over a range of 1 to 300 [mu]m in the medium with
30 percent of the Cr(VI) transported into the cells within two hours and
67 percent at 16 hours at the lowest concentration (Ex. 31-22-18).
Water insoluble Cr(VI) particulates do not readily dissolve into
epithelial lining fluids of the bronchoalveolar region. This has led to
claims that insoluble chromates, such as lead chromate pigments, are
not bioavailable and, therefore, are unable to cause carcinogenesis
(Ex. 31-15). However, several scientific studies indicate that
insoluble Cr(VI) particulates can come in close contact with the
bronchoalveolar epithelial cell surface, allowing enhanced uptake into
cells. Wise et al. showed that respirable lead chromate particles
adhere to the surface of rodent cells in culture causing cell-enhanced
dissolution of the chromate ion as well as phagocytosis of lead
chromate particles (Exs. 35-68; 35-67). The intracellular accumulation
was both time- and dose-dependant. Cellular uptake resulted in damage
to DNA, apoptosis (i.e., form of programmed cell death), and neoplastic
transformation (Ex. 35-119). Singh et al. showed that treatment of
normal human lung epithelial cells with insoluble lead chromate
particulates (0.4 to 2.0 [mu]g/cm\2\) or soluble sodium chromate (10
[mu]M) for 24 hours caused Cr(VI) uptake, Cr-DNA adduct formation, and
apoptosis (Ex. 35-66). The proximate genotoxic agent in these cell
systems was determined to be the chromate rather than the lead ions
(Ex. 35-327). Elias et al. reported that cell-enhanced particle
dissolution and uptake was also responsible for the cytotoxicity and
neoplastic transformation in Syrian hamster embryo cells caused by
Cr(VI) pigments, including several complex industrial chrome yellow and
molybdate orange pigments (Ex. 125). These studies are key experimental
evidence in the determination that water-insoluble Cr(VI) compounds, as
well as water soluble Cr(VI) compounds, are to be regarded as
carcinogenic agents. This determination is further discussed in the
next section (see V.B.9).
Reduction to the poorly permeable Cr(III) in the epithelial lining
fluid limits cellular uptake of Cr(VI). Ascorbic acid and glutathione
(GSH) are believed to be the key molecules responsible for the
extracellular reduction. Cantin et al. reported high levels of GSH in
human alveolar epithelial lining fluid and Susuki et al. reported
significant levels of ascorbic acid in rat lung lavage fluids (Exs. 35-
147; 35-143). Susuki and Fukuda studied the kinetics of soluble Cr(VI)
reduction with ascorbic acid and GSH in vitro and following
intratracheal instillation (Ex. 35-90). They reported that the rate of
reduction was proportional to Cr(VI) concentration with a half-life of
just under one minute to several hours. They found the greatest
reduction rates with higher levels of reductants. Ascorbic acid was
more active than GSH. Cr(VI) reduction was slower in vivo than
predicted from in vitro and principally involved ascorbic acid, not
GSH. This research indicates that extracellular Cr(VI) reduction to
Cr(III) is variable depending on the concentration and nature of the
reductant in the epithelial fluid lining regions of the respiratory
tract. De Flora et al. determined the amount of soluble Cr(VI) reduced
in vitro by human bronchiolar alveolar fluid and pulmonary alveolar
macrophage fractions over a short period and used these specific
activities to estimate an "overall reducing capacity" of 0.9-1.8 mg
Cr(VI) and 136 mg Cr(VI) per day per individual, respectively (Ex. 35-
140).
De Flora, Jones, and others have interpreted the extracellular
reduction data to mean that very high levels of Cr(VI) are required to
"overwhelm" the reductive defense mechanism before target cell uptake
can occur and, as such, impart a "threshold" character to the
exposure-response (Exs. 35-139; 31-22-7). However, the threshold
capacity concept does not consider that facilitated lung cell uptake
and extracellular reduction are dynamic and parallel processes that
happen concurrently. If their rates are comparable then some cellular
uptake of Cr(VI) would be expected, even at levels that do not
"overwhelm" the reductive capacity. Based on the in vitro kinetic
data, it would appear that such situations are plausible, especially
when concentrations of ascorbic acid are low. Unfortunately, there has
been little systematic study of the dose-dependence of Cr(VI) uptake in
the presence of physiological levels of ascorbate and GSH using
experimental systems that possess active anion transport capability.
The implications of extracellular reduction on the shape of Cr(VI)
dose--lung cancer response curve is further discussed in Section
VI.G.2.c.
Wise et al. did study uptake of a single concentration of insoluble
lead chromate particles (0.8 [mu]g/cm2) and soluble sodium
chromate (1.3 [mu]M) in Chinese hamster ovary cells co-treated with a
physiological concentration (1mM) of ascorbate (Ex. 35-68). They found
that the ascorbate substantially reduced, but did not eliminate,
chromate ion uptake over a 24 hour period. Interestingly, ascorbate did
not affect phagocytic uptake of lead chromate particles, although it
eliminated the Cr(VI)-induced clastogenesis (e.g., DNA strand breakage
and chromatid exchange) as measured under their experimental
conditions.
Singh et al. suggested that cell surface interactions with
insoluble lead chromate particulates created a concentrated
microenvironment of chromate ions resulting in higher intracellular
levels of chromium than would occur from soluble Cr(VI) (Ex. 35-149).
Cell membrane-enhanced uptake of Cr(VI) is consistent with the
intratracheal and intrabronchial instillation studies in rodents that
show greater carcinogenicity with slightly soluble (e.g., calcium
chromate and strontium chromate) than with the highly water-soluble
chromates (e.g., sodium chromate and chromic acid) (Ex. 11-2).
Finally, Cr(VI) deposited in the tracheobronchial and alveolar
regions of the respiratory tract is cleared by the mucocilliary
escalator (soluble and particulate Cr(VI)) and macrophage phagocytosis
(particulate Cr(VI) only). In most instances, these clearance processes
take hours to days to completely clear Cr(VI) from the lung, but it can
take considerably longer for particulates deposited at certain sites.
For example, Ishikawa et al. showed that some workers had substantial
amounts of chromium particulates at the bifurcations of the large
bronchii for more than two decades after cessation of exposure (Ex. 35-
81). Mancuso reported chromium in the lungs of six chromate production
workers who died from lung cancer (as cited in Ex. 35-47). The interval
between last exposure to Cr(VI) until autopsy ranged from 15 months to
16 years. Using hollow casts of the human tracheobronchial tree and
comparing particle deposition with reported occurrence of bronchogenic
tumors, Schlesinger and Lippman were able to show good correlations
between sites of greatest deposition and increased incidence of
bronchial tumors (Ex. 35-102).
b. Intracellular Reduction of Cr(VI). Once inside the cell, the
hexavalent chromate ion is rapidly reduced to intermediate oxidation
states, Cr(V) and Cr(IV), and the more chemically stable Cr(III).
Unlike Cr(VI), these other chromium forms are able to react with DNA
and protein to generate a variety of adducts and complexes. In
addition, reactive oxygen species (ROS) are produced during the
intracellular reduction of Cr(VI) that are also capable of damaging
DNA. These reactive intermediates, and not Cr(VI) itself, are considered
to be the ultimate genotoxic agents that initiate the carcinogenic process.
After crossing the cell membrane, Cr(VI) compounds can be non-
enzymatically converted to Cr(III) by several intracellular reducing
factors (Ex. 35-184). The most plentiful electron donors in the cell
are GSH, and other thiols, such as cysteine, and ascorbate. Connett and
Wetterhahn showed that a Cr(VI)-thioester initially forms in the
presence of GSH (Ex. 35-206). A two-phase reduction then occurs with
rapid conversion to Cr(V) and glutathionyl radical followed by
relatively slower reduction to Cr(III) that requires additional
molecules of GSH. Depletion of cellular GSH and other thiols is
believed to retard complete reduction of Cr(VI) to Cr(III), allowing
buildup of intermediates Cr(V) and Cr(IV). The molecular kinetics of
the Cr(VI) to Cr(III) reduction with ascorbate is less well understood
but can also involve intermediate formation of Cr(V) and free radicals
(Ex. 35-184).
Another important class of intracellular Cr(VI) reductions are
catalyzed by flavoenzymes, such as GSH reductase, lipoyl dehydrogenase,
and ferredoxin-NADP oxidoreductase. The most prominent among these is
GSH reductase that uses NADPH as a cofactor in the presence of
molecular oxygen (O2) to form Cr(V)-NADPH complexes. During
the reaction, O2 undergoes one electron reduction to the
superoxide radical (O2-) which produces hydrogen
peroxide (H2O2) through the action of the enzyme
superoxide dismutase. The Cr(V)-NADPH can then react with
H2O2 to regenerate Cr(VI) giving off hydroxyl
radicals, a highly reactive oxygen species, by a Fenton-like reaction.
It is, therefore, possible for a single molecule of Cr(VI) to produce
many molecules of potentially DNA damaging ROS through a repeated
reduction/oxidation cycling process. Shi and Dalal used electron spin
resonance (ESR) to establish formation of Cr(V)-NADPH and hydroxyl
radical in an in vitro system (Ex. 35-169; 35-171). Sugiyama et al.
reported Cr(V) formation in cultured Chinese hamster cells treated with
soluble Cr(VI) (Ex.35-133). Using a low frequency ESR, Liu et al.
provided evidence of Cr(V) formation in vivo in mice injected with
soluble Cr(VI) (Ex. 35-141-28).
Several studies have documented that Cr(VI) can generate Cr(V) and
ROS in cultured human lung epithelial cells and that this reduction/
oxidation pathway leads to DNA damage, activation of the p53 tumor
suppressor gene and stress-induced transcription factor NF-[kappa]B,
cell growth arrest, and apptosis (Exs. 35-125; 35-142; 31-22-18; 35-
135). Leonard et al. used ESR spin trapping, catalase, metal chelators,
free radical scavengers, and O2-free atmospheres to show
that hydroxyl radical generation involves a Fenton-like reaction with
soluble potassium dichromate (Ex. 31-22-17) and insoluble lead chromate
(Ex.35-137) in vitro. Liu et al. showed that the Cr(IV)/Cr(V) compounds
are also able to generate ROS with H2O2 in a
Fenton reduction/oxidation cycle in vitro (Ex. 35-183).
Although most intracellular reduction of Cr(VI) is believed to
occur in the cytoplasm, Cr(VI) reduction can also occur in mitochondria
and the endoplasmic reticulum. Cr(VI) reduction can occur in the
mitochondria through the action of the electron transport complex (Ex.
35-230). The microsomal cytochrome P-450 system in the endoplasmic
reticulum also enzymatically reduces Cr(VI) to Cr(V), producing ROS
through reduction/oxidation cycling as described above (Ex. 35-171).
c. Genotoxicity and Damage to DNA. A large number of studies have
examined multiple types of genotoxicity in a wide range of experimental
test systems. Many of the specific investigations have been previously
reviewed by IARC (Ex. 35-43), Klein (Ex. 35-134), ATSDR (Ex. 35-41),
and the K.S. Crump Group (Ex. 35-47) and will only be briefly
summarized here. The body of evidence establishes that both soluble and
insoluble forms of Cr(VI) cause structural DNA damage that can lead to
genotoxic events such as mutagenisis, clastogenisis, inhibition of DNA
replication and transcription, and altered gene expression, all of
which probably play a role in neoplastic transformation. The reactive
intermediates and products that occur from intracellular reduction of
Cr(VI) cause a wide variety of DNA lesions. The type(s) of DNA damage
that are most critical to the carcinogenic process is an area of active
investigation.
Many Cr(VI) compounds are mutagenic in bacterial and mammalian test
systems (Ex. 35-118). In the bacterial Salmonella typhimurium strains,
soluble Cr(VI) caused base pair substitutions at A-T sites as well as
frame shift mutations (Ex. 35-161). Nestmann et al. also reported
forward and frame shift mutations in Salmonella typhimurium with pre-
solubilized lead chromate (Ex. 35-162). Several Cr(VI) compounds have
produced mutagenic responses at various genetic loci in mammalian cells
(Ex. 12-7). Clastogenic damage, such as sister chromatid exchange and
chromosomal aberrations, have also been reported for insoluble Cr(VI)
and soluble Cr(VI) (Exs. 35-132; 35-115). Mammalian cells undergo
neoplastic transformation following treatment with soluble Cr(VI) or
insoluble Cr(VI), including a number of slightly soluble zinc and
insoluble lead chromate pigments (Exs. 12-5; 35-186).
Genotoxicity has been reported from Cr(VI) administration to
animals in vivo. Soluble Cr(VI) induced micronucleated erythrocytes in
mice following intraperitoneal (IP) administration (Ex. 35-150). It
also increased the mutation frequency in liver and bone marrow
following IP administration to lacZ transgenic mice (Exs. 35-168; 35-
163). Izzotti et al. reported DNA damage in the lungs of rats exposed
to soluble Cr(VI) by intratracheal instillation (Ex. 35-170).
Intratracheal instillation of soluble Cr(VI) produced a time- and dose-
dependant elevation in mutant frequency in the lung of Big Blue
transgenic mice (Ex. 35-174). Oral administration of soluble Cr(VI) in
animals did not produce genotoxicity in several studies probably due to
route-specific differences in absorption. OSHA is not aware of
genotoxicity studies from in vivo administration of insoluble Cr(VI).
Studies of chromosomal and DNA damage in workers exposed to Cr(VI) vary
in their findings. Some studies reported higher levels of chromosomal
aberrations, sister chromatid exchanges, or DNA strand breaks in
peripheral lymphocytes of stainless steel welders (Exs. 35-265; 35-160)
and electroplaters (Ex. 35-164). Other studies were not able to find
excess damage in DNA from the blood lymphocytes of workers exposed to
Cr(VI) (Exs. 35-185; 35-167). These reports are difficult to interpret
since co-exposure to other genotoxic agents (e.g., other metals,
cigarette smoke) likely existed and the extent of Cr(VI) exposures were
not known.
Because of the consistent positive response across multiple assays
in a wide range of experimental systems from prokaryotic organisms
(e.g., bacteria) to human cells in vitro and animals in vivo, OSHA
regards Cr(VI) as an agent able to induce carcinogenesis through a
genotoxic mode of action. Both soluble and insoluble forms of Cr(VI)
are reported to cause genotoxicity and neoplastic transformation. On
the other hand, Cr(III) compounds do not easily cause genotoxicity in
intact cellular systems, presumably due to the inability of Cr(III) to
penetrate cell membranes (Exs. 12-7; 35-186).
There has been a great deal of research to identify the types of
damage to DNA caused by Cr(VI), the reactive intermediates that are
responsible for the damage, and the specific genetic lesions critical to
carcinogenesis. It was shown that Cr(VI) was inactive in DNA binding assays
with isolated nuclei or purified DNA (Ex. 35-47). However, Cr(III) was able
to produce DNA protein cross-links, sister chromatid exchanges, and chromosomal
aberrations in an acellular system. Zhitkovich et al. showed that incubation of
Chinese hamster ovary cells with soluble Cr(VI) produced ternary complexes of
Cr(III) cross-linked to cysteine, other amino acids, or glutathione and
the DNA phosphate backbone (Ex. 312). Utilizing the pSP189 shuttle
vector plasmid, they showed these DNA-Cr(III)-amino acid cross-links
were mutagenic when introduced in human fibroblasts (Ex. 35-131).
Another research group showed that plasmid DNA treated with Cr(III)
produced intrastrand crosslinks and the production of these lesions
correlated with DNA polymerase arrest (Ex. 35-126). The same
intrastrand crosslinks and DNA polymerase arrest could also be induced
by Cr(VI) in the presence of ascorbate as a reducing agent to form
Cr(III) (Ex. 35-263). These results were confirmed in a cell system by
treating human lung fibroblasts with soluble Cr(VI), isolating genomic
DNA, and demonstrating dose-dependent guanine-specific arrest in a DNA
polymerase assay (Ex. 35-188). Cr(V) may also form intrastrand
crosslinks since Cr(V) interacts with DNA in vitro (Ex. 35-178). The
Cr(V)-DNA crosslinks are probably readily reduced to Cr(III) in cell
systems. Intrastrand crosslinks have also been implicated in inhibition
of RNA polymerase and DNA topoisomerase, leading to cell cycle arrest,
apoptosis and possibly other disturbances in cell growth that
contribute to the carcinogenic pathway (Ex. 35-149).
DNA strand breaks and oxidative damage result from the one electron
reduction/oxidation cycling of Cr(VI), Cr(V), and Cr(IV). Shi et al.
showed that soluble Cr(VI) in the presence of ascorbate and
H2O2 caused DNA double strand breaks and 8-
hydroxy deoxyguanine (8-OHdG, a marker for oxidative DNA damage) in
vitro (Ex. 35-129). Leonard et al. showed that the DNA strand breaks
were reduced by several experimental conditions including an
O2-free atmosphere, catabolism of H2O2
by catalase, ROS depletion by free radical scavengers, and chelation of
Cr(V). They concluded that the strand breaks and 8-OHdG resulted from
DNA damage caused by hydroxyl radicals from Cr(VI) reduction/oxidation
cycling (Ex. 31-22-17). Generation of ROS-dependant DNA damage could
also be shown with insoluble Cr(VI) (Ex. 35-137). DNA strand breaks and
related damage caused by soluble Cr(VI) have been reported in Chinese
hamster cells (Ex. 35-128), human fibroblasts (Ex. 311), and human
prostate cells (Ex. 35-255). Pretreatment of Chinese hamster cells with
a metal chelator suppressed Cr(V) formation from Cr(VI) and decreased
DNA strand breaks (Ex. 35-197). Chinese hamster cells that developed
resistance to H2O2 damage also had reduced DNA
strand breaks from Cr(VI) treatment compared to the normal phenotype
(Ex. 35-176).
Several researchers have been able to modulate Cr(VI)-induced DNA
damage using cellular reductants such as ascorbate, GSH and the free
radical scavenger tocopherol (vitamin E). This has provided insight
into the relationships between DNA damage, reduced chromium forms and
ROS. Sugiyama et al. showed that Chinese hamster cells pretreated with
ascorbate decreased soluble Cr(VI)-induced DNA strand damage (e.g.,
alkali-labile sites), but enhanced DNA-amino acid crosslinks (Ex. 35-
133). Standeven and Wetterhahn reported that elimination of ascorbate
from rat lung cytosol prior to in vitro incubation with soluble Cr(VI)
completely inhibited Cr-DNA binding (Ex. 35-180). However, not all
types of Cr-DNA binding are enhanced by ascorbate. Bridgewater et al.
found that high ratios of ascorbate to Cr(VI) actually decreased
intrastrand crosslinks in vitro while low ratios induced their
formation (Ex. 35-263). This finding is consistent with research by
Stearns and Watterhahn who showed that excessive ascorbate relative to
Cr(VI) leads to two-electron reduction of Cr(III) and formation of
Cr(III)-DNA monoadducts and DNA-Cr(III)-amino acid crosslinks (Ex. 35-
166). Low amounts of ascorbate primarily cause one-electron reduction
to intermediates Cr(V) and Cr(IV) that form crosslinks with DNA and ROS
responsible for DNA strand breaks, alkali-labile sites, and clastogenic
damage. This explains the apparent paradox that extracellular Cr(VI)
reduction by ascorbate to Cr(III) reduces Cr(VI)-induced DNA binding
but intracellular Cr(VI) reduction by ascorbate to Cr(III) enhances Cr-
DNA binding. The aforementioned studies used soluble forms of Cr(VI),
but Blankenship et al. showed that ascorbate pretreatment inhibited
chromosomal aberrations in Chinese hamster ovary cells caused by both
insoluble lead chromate particles as well as soluble Cr(VI) (Ex. 35-
115). Pretreatment with the free radical scavenger tocopherol also
inhibits chromosomal aberrations and alkali-labile sites in Cr(VI)-
treated cells (Exs. 35-115; 35-128).
Studies of the different types of DNA damage caused by Cr(VI) and
the modulation of that damage inside the cell demonstrate that Cr(VI)
itself is not biologically active. Cr(VI) must undergo intracellular
reduction to Cr(V), Cr(IV), and Cr(III) before the damage to DNA can
occur. The evidence suggests that Cr(III) can cause DNA-Cr-amino acid,
DNA-Cr-DNA crosslinks and Cr-DNA monoadducts. Cr(V) and possibly Cr(IV)
contribute to intrastrand crosslinks and perhaps other Cr-DNA binding.
ROS generated during intracellular reduction of Cr(VI) lead to lesions
such as chromosomal aberrations, DNA strand breaks, and oxidative DNA
damage. The specific DNA lesions responsible for neoplastic
transformation have yet to be firmly established so all forms of DNA
damage should, at this time, be regarded as potential contributors to
carcinogenicity.
d. Cr(VI)-induced Disturbances in the Regulation of Cell
Replication. Recent research has begun to elucidate how Cr(VI)-induced
oxidative stress and DNA lesions trigger cell signaling pathways that
regulate the cell growth cycle. The complex regulation of the cell
growth cycle by Cr(VI) involves activation of the p53 protein and other
transcription factors that respond to oxidative stress and DNA damage.
The cellular response ranges from a temporary pause in the cell cycle
to terminal growth arrest (i.e., viable cells that have lost the
ability to replicate) and a programmed form of cell death, known as
apoptosis. Apoptosis involves alterations in mitochondrial
permeability, release of cytochrome c and the action of several kinases
and caspases. Less is known about the molecular basis of terminal
growth arrest. Terminal growth arrest and apoptosis serve to eliminate
further growth of cells with unrepaired Cr(VI)-induced genetic damage.
However, it is believed that cells which escape these protective
mechanisms and regain replicative competence eventually become
resistant to normal growth regulation and can transform to a neoplastic
phenotype (Exs. 35-121; 35-122; 35-120).
Blankenship et al. first described apoptosis as the primary mode of
cell death following a two hour treatment of Chinese hamster ovary
cells with high concentrations (>150 [mu]M) of soluble Cr(VI) (Ex. 35-
144). Apoptosis also occurs in human lung cells following short-term
treatment with soluble Cr(VI)(Ex. 35-125) as well as longer term treatment
(e.g., 24 hours) with lower concentrations of soluble Cr(VI) (e.g., 10 [mu]M)
and insoluble Cr(VI) in the form of lead chromate (Ex. 35-166). Ye et al.
found that the Cr(VI) treatment that caused apoptosis also activated expression
of p53 protein (Ex. 35-125). This apoptotic response was substantially
reduced in a p53-deficient cell line treated with Cr(VI), suggesting
that the p53 activation was required for apoptosis. Other studies using
p53 null cells from mice and humans confirmed that Cr(VI)-induced
apoptosis is p53-dependent (Ex. 35-225).
The p53 protein is a transcription factor known to be activated by
DNA damage, lead to cell cycle arrest, and regulate genes responsible
for either DNA repair or apoptosis. Therefore, it is likely that the
p53 activation is a response to the Cr(VI)-induced DNA damage.
Apoptosis (i.e., programmed cell death) is triggered once the Cr(VI)-
induced DNA damage becomes too extensive to successfully repair. In
this manner, apoptosis serves to prevent replication of genetically
damaged cells.
Several researchers have gone on to further elucidate the molecular
pathways involved in Cr(VI)-induced apoptosis. ROS produced by
intracellular Cr(VI) reduction/oxidation cycling have been implicated
in the activation of p53 and apoptosis (Exs. 35-255; 35-122). Using
specific inhibitors, Pritchard et al. showed that mitochondrial release
of cytochrome c is critical to apoptotic death from Cr(VI) (Ex. 35-
159). Cytochrome c release from mitochondria could potentially result
from either direct membrane damage caused by Cr(VI)-induced ROS or
indirectly by enhanced expression of the p53-dependent apoptotic
proteins, Bax and Nova, known to increase mitochondrial membrane
permeability.
Cr(VI) causes cell cycle arrest and reduces clonogenic potential
(i.e., normal cell growth) at very low concentrations (e.g., 1 [mu]M)
where significant apoptosis is not evident. Xu et al. showed that human
lung fibroblasts treated with low doses of Cr(VI) caused guanine-
guanine intrastrand crosslinks, guanine-specific polymerase arrest, and
inhibited cell growth at the G1/S phase of the cell cycle
(Ex. 35-188). Zhang et al. described a dose-dependent increase in
growth arrest at the G2/M phase of the cell cycle in a human
lung epithelial cell line following 24 hour Cr(VI) treatment over a
concentration range of 1 to 10 [mu]M (Ex. 35-135). The cell cycle
arrest could be partially eliminated by reducing production of Cr(VI)-
induced ROS. Apoptosis was not detected in these cells until a
concentration of 25 [mu]M Cr(VI) had been reached. These data suggest
that low cellular levels of Cr(VI) are able to cause DNA damage and
disrupt the normal cell growth cycle.
Pritchard et al. studied the clonogenicity over two weeks of human
fibroblasts treated 24 hours with soluble Cr(VI) concentrations from 1
to 10 [mu]M (Ex. 35-120). They reported a progressive decline in cell
growth with increasing Cr(VI) concentration. Terminal growth arrest
(i.e., viable cells that have lost the ability to replicate) was
primarily responsible for the decrease in clonogenic survival below 4
[mu]M Cr(VI). At higher Cr(VI) concentrations, apoptosis was
increasingly responsible for the loss in clonogenicity. Pritchard et
al. and other research groups have suggested that a subset of cells
that continue to replicate following Cr(VI) exposure could contain
unrepaired genetic damage or could have become intrinsically resistant
to processes (e.g., apoptosis, terminal growth arrest) that normally
control their growth (Exs. 35-121; 35-122; 35-120). These surviving
cells would then be more prone to neoplastic progression and have
greater carcinogenic potential.
e. Summary. Respirable chromate particulates are taken up by target
cells in the bronchoalveolar region of the lung, become intracellularly
reduced to several reactive genotoxic species able to damage DNA,
disrupt normal regulation of cell division and cause neoplastic
transformation. Scientific studies indicate that both water soluble and
insoluble Cr(VI) can be transported into the cell. In fact, cell
surface interactions with slightly soluble and insoluble chromates may
create a concentrated microenvironment of chromate ion, especially in
the case of the slightly soluble Cr(VI) compounds that more readily
dissociate. The higher concentration of chromate ion in close proximity
to the lung cells will likely result in higher intracellular Cr(VI)
than would occur from the highly water-soluble chromates. This is
consistent with the studies of respiratory tract carcinogenesis in
animals that indicate the most tumorigenic chromates had low to
moderate water solubility. Once inside the cell, Cr(VI) is converted to
several lower oxidation forms able to bind to and crosslink DNA. ROS
are produced during intracellular reduction/oxidation of Cr(VI) that
further damage DNA. These structural lesions are functionally
translated into a impaired DNA replication, mutagenesis, and altered
gene expression that ultimately lead to neoplastic transformation.
9. Conclusion
In the NRPM, OSHA preliminarily concluded that the weight of
evidence supports the determination that all Cr(VI) compounds should be
regarded as carcinogenic to workers (69 FR at 59351). This conclusion
included the highly water soluble chromates, such as sodium chromate,
sodium dichromate, and chromic acid; chromates of slight and
intermediate water solubility such as calcium chromate, strontium
chromates, and many zinc chromates (e.g. zinc yellow); and chromates
that have very low water solubility and are generally considered to be
water insoluble such as barium chromate and lead chromates. The
strongest evidence supporting this conclusion comes from the many
cohort studies reporting excess lung cancer mortality among workers
engaged in the production of soluble chromates (Exs. 7-14; 31-22-11;
23; 31-18-4), chromate pigments (Exs. 7-36; 7-42; 7-46), and chrome
plating (Exs. 35-62; 35-271). Chromate production workers were
principally exposed to the highly soluble sodium chromate and
dichromate (Ex. 35-61) although lesser exposure to other chromates,
such as highly soluble chromic acid and slightly soluble calcium
chromate probably occurred. Pigment production workers were principally
exposed Cr(VI) in the form of lead and zinc chromates. Significantly
elevated lung cancer mortality was found in two British chromium
electroplating cohorts (Exs. 35-62; 35-271). These workers were exposed
to Cr(VI) in the form of chromic acid mist. Therefore, significantly
elevated lung cancer rates have been observed in working populations
exposed to a broad range of Cr(VI) compounds.
Cellular research has shown that both highly water soluble (e.g.
sodium chromate) Cr(VI) and water insoluble (e.g. lead chromate) Cr(VI)
enter lung cells (see Section V.8.a) and undergo intracellular
reduction to several lower oxidation forms able to bind to and
crosslink DNA as well as generate reactive oxygen species that can
further damage DNA (see Section V.8.b). Soluble and insoluble Cr(VI)
compounds are reported to cause mutagenesis, clastogenesis, and
neoplastic transformation across multiple assays in a wide range of
experimental systems from prokaryotic organisms to human cells in vitro
and animals in vivo (see Section V.8.c).
The carcinogenicity of various Cr(VI) compounds was examined after
instillation in the respiratory tract of rodents. Slightly water
soluble Cr(VI) compounds, strontium chromate, calcium chromate, and some
zinc chromates produced a greater incidence of respiratory tract tumors than
highly water soluble (e.g. sodium dichromate and chromic acid) and
water insoluble (e.g. barium chromate and lead chromates) Cr(VI)
compounds under similar experimental protocol and conditions (see
Section V.7). This likely reflects the greater tendency for chromates
of intermediate water solubility to provide a persistent high local
concentration of solubilized Cr(VI) in close proximity to the target
cell. Highly soluble chromates rapidly dissolve and diffuse in the
aqueous fluid lining the epithelia of the lung. Thus, these chromates
are less able to achieve the higher local concentrations within close
proximity of the lung cell surface than the slightly water soluble
chromates. However, it has been shown that water-soluble Cr(VI) can
still enter lung cells, damage DNA, and cause cellular effects
consistent with carcinogenesis (Ex. 31-22-18; 35-125; 35-135; 35-142).
Like the slightly water soluble chromates, water insoluble Cr(VI)
particulates are able to come in close contact with the lung cell
surface and slowly dissolve into readily absorbed chromate ion. For
example, water insoluble lead chromate has been shown to enter human
airway cells both through extracellular solubilization as chromate ion
(Exs. 35-66; 35-327; 47-12-3) as well as internalization as
unsolubilized particulate (Exs. 35-66; 47-19-7). However, the rate of
solubilization and uptake of water insoluble Cr(VI) is expected to be
more limited than chromates with moderate solubility. Once chromate ion
is inside lung cells, studies have shown that similar cellular events
believed critical to initiating neoplastic transformation occur
regardless of whether the source is a highly soluble or insoluble
Cr(VI) compound (Ex. 35-327).
a. Public Comment on the Carcinogenicity of Cr(VI) Compounds
In the NRPM, OSHA requested comment on whether currently available
epidemiologic and experimental studies supported the determination that
all Cr(VI) compounds possess carcinogenic potential and solicited
additional information that should be considered in evaluating relative
carcinogenic potency of the different Cr(VI) compounds (69 FR 59307).
Several comments supported the view that sufficient scientific evidence
exists to regard all Cr(VI) compounds as potential occupational
carcinogens (Exs. 38-106-2; 38-222; 39-73-2; 40-10-2; 42-2). The AFL-
CIO stated that " * * * the agency has fully demonstrated that Cr(VI)
is a human carcinogen and that exposed workers are at risk of
developing lung cancer" (Ex. 38-222). NIOSH stated that "the
epidemiologic and experimental studies cited by OSHA support the
carcinogenic potential of all Cr(VI) compounds (i.e. water soluble,
insoluble, and slightly soluble)" (Ex. 40-10-2, p. 4). Peter Lurie of
Public Citizen testified:
As we heard repeatedly in the course of this hearing, scientific
experts, in fact, agree. They agree that the most reasonable
approach to the regulation is to consider them all [Cr(VI)
compounds] to be carcinogenic (Tr. 710).
Several commenters agreed that the evidence supported the
qualitative determination that Cr(VI) compounds were carcinogenic but
wished to make clear that the information was inadequate to support
quantitative statements about relative potency of the individual
chromates (Exs. 38-106-2; 40-10-2; 42-2). For example, the Boeing
Company in their technical comments stated:
The available data does support the conclusion that the low
solubility hexavalent chromium compounds [e.g. strontium chromate]
can cause cancer but evidence to support a quantitative comparison
of carcinogenic potency based on differences in solubility is
lacking (Ex. 38-106-2, p. 18).
Pigment Manufacturers' Comments on Carcinogenicity of Lead Chromate--
One group that did not regard all Cr(VI) compounds as occupational
carcinogens was the color pigment manufacturers who manufacture and
market lead chromate pigments which are primarily used in industrial
coatings and colored plastic articles. The color pigment manufacturers
maintain that their lead chromate products are unreactive in biological
systems, are not absorbed into the systemic circulation by any route,
and can not enter lung cells (Ex. 38-205, p. 14). Their principal
rationale is that lead chromate is virtually insoluble in water, is
unable to release chromate ion into aqueous media, and therefore, is
incapable of interacting with biological systems (Exs. 38-205, p. 95;
38-201-1, p. 9). The color pigment manufacturers assert that their lead
chromate pigment products are double encapsulated in a resin/plastic
matrix surrounded by a silica coating and that the encapsulated pigment
becomes even less "bioavailable" than unencapsulated "less
stabilized" lead chromates. They believe the extreme stability and
non-bioavailable nature of their products makes them a non-carcinogenic
form of Cr(VI) (Ex. 38-205, p. 106).
According to the Color Pigment Manufacturers Association (CPMA),
several pieces of scientific evidence support their position, namely,
the lack of a significant excess of lung cancer mortality in three
cohorts of pigment workers engaged in the production of water-insoluble
lead chromate (Ex. 38-205, pp. 88-91) and the lack of statistically
significant elevated tumor incidence following a single instillation of
lead chromate in the respiratory tract of rats (Ex. 38-205, pp. 88-92).
They dismiss as irrelevant other animal studies that produced
statistically significant increases in tumors when lead chromate was
repeatedly injected by other routes. In addition, CPMA claims that the
lead chromate used in cellular studies that report genotoxicity was
reagent grade, was contaminated with soluble chromate, and was
inappropriately solubilized using strong acids and bases prior to
treatment (Exs. 38-205, pp. 93-94; 47-31, pp. 9-13). They are
especially critical of studies conducted by the Environmental and
Genetic Toxicology group at the University of Southern Maine that
report lead chromate particulates to be clastogenic in human lung cells
(Exs. 34-6-1; 38-205, pp. 98-102 & appendix D; 47-22). Instead, they
rely on two in vitro studies of lead chromate pigments that report a
lack of genotoxicity in cultured bacterial and hamster ovary cells,
respectively (Exs. 47-3 Appendix C; 38-205, p. 94).
OSHA addresses many of the CPMA claims in other sections of the
preamble. The bioavailability issue of encapsulated lead chromate is
addressed in Section V.A.2. The CPMA request to consider the lack of
excess lung cancer mortality among pigment workers exposed exclusively
to lead chromate is discussed in Section V.B.2. The CPMA assertions
that animal studies are evidence that lead chromates are not
carcinogenic to workers are addressed in Section V.B.7. The studies
documenting uptake of lead chromate into lung cells are described in
Section V.B.8.a. Section V.B.8.c describes evidence that lead chromate
is genotoxic. As requested by CPMA, OSHA will pull these responses
together and expand on their concerns below.
Lung Cancer Mortality in Pigments Workers Exposed to Lead
Chromate--Comments and testimony from NIOSH and others cite evidence of
excess lung cancer among pigment workers and support the results of
OSHA's preliminary risk assessment for color pigments in general and
for lead chromate in particular (Tr. 135-146, 316, 337, Ex. 40-18-1, p.
2). However, comments submitted by the CPMA and the Dominion Colour
Corporation (DCC) attributed the excess lung cancer risk observed in
pigment worker studies to zinc chromate (Tr. 1707, 1747, Exs. 38-201-1,
p. 13; 38-205, p. 90; 40-7, p. 92). For example, the CPMA stated that:
When lead chromate and zinc chromate exposures occur
simultaneously, there appears to be a significant cancer hazard.
However, when lead chromate pigments alone are the source of
chromium exposure, a significant carcinogenic response has never
been found (Ex. 40-7, p. 92).
The latter statement refers to the Davies et al. (1984) study of
British pigment workers, the Cooper et al. (1983) study of U.S. pigment
workers, and the Kano et al. (1993) study of pigment workers in Japan,
all of which calculated separate observed and expected lung cancer
deaths for workers exposed exclusively to lead chromate (Ex. 38-205, p.
89). DCC and the Small Business Administration's Office of Advocacy
similarly stated that the excess lung cancer risk observed among
workers exposed to both zinc chromate and lead chromate cannot
necessarily be attributed to lead chromate (Exs. 38-201-1, p. 13; 38-7,
p. 4).
OSHA agrees with CPMA and DCC that the excess lung cancer observed
in most pigment worker studies taken alone cannot be considered
conclusive evidence that lead chromate is carcinogenic. Given that the
workers were exposed to both zinc chromate and lead chromate, it is not
possible to draw strong conclusions about the effects of either
individual compound using only these studies. However, based on the
overall weight of available evidence, OSHA believes that the excess
lung cancer found in these studies is most likely attributable to lead
chromate as well as zinc chromate exposure. Lead chromate was the
primary source of Cr(VI) for several worker cohorts with excess lung
cancer (e.g., Davies et al. (1984), Factory A; Hayes et al. (1989); and
Deschamps et al. (1995)) (Exs. 7-42; 7-46; 35-234), and as previously
discussed, there is evidence from animal and mechanistic studies
supporting the carcinogenicity of both zinc chromate and lead chromate.
Considered in this context, the elevated risk of lung cancer observed
in most chromate pigment workers is consistent with the Agency's
determination that all Cr(VI) compounds--including lead chromate--
should be regarded as carcinogenic.
Moreover, OSHA disagrees with the CPMA and DCC interpretation of
the data on workers exposed exclusively to lead chromate. In the
Preamble to the Proposed Rule, OSHA stated that "[t]he number of lung
cancer deaths [in the Davies, Cooper, and Kano studies] is too small to
be meaningful" with respect to the Agency's determination regarding
the carcinogenicity of lead chromate (FR 69 at 59332). The CPMA
subsequently argued that:
[b]y this rationale, OSHA could never conclude that a compound
such as lead chromate pigment exhibits no carcinogenic potential
because there can never be enough lung cancer deaths to produce a
"meaningful" result. This is an arbitrary and obviously biased
assessment which creates an insurmountable barrier. Since the lead
chromate pigments did not create an excess of lung cancer, there
cannot be a significant enough mortality from lung cancer to be
meaningful (Ex. 38-205, p. 90).
OSHA believes that these comments reflect a misunderstanding of the
sense in which the Davies, Cooper, and Kano studies are too small to be
meaningful, and also a misunderstanding of the Agency's position.
Contrary to CPMA's argument, a study with no excess in lung cancer
mortality can provide evidence of a lack of carcinogenic effect if the
confidence limits for the measurement of effect are close to the null
value. In other words, the measured effect must be close to the null
and the study must have a high level of precision. In the case of the
Davies, Cooper, and Kano studies, the standardized mortality ratio
(SMR) is the measurement of interest and the null value is an SMR of 1.
Table V.10 below shows that the SMRs for these study populations are
near or below 1; however, the 95% confidence intervals for the SMRs are
quite wide, indicating that the estimated SMRs are imprecise. The Kano
data, for example, are statistically consistent with a "true" SMR as
low as 0.01 or as high as 2.62. The results of these studies are too
imprecise to provide evidence for or against the hypothesis that lead
chromate is carcinogenic.

This lack of precision may be partly explained by the small size of
the studies, as reflected in the low numbers of expected lung cancers.
However, it is the issue of precision, and not the number of lung
cancer deaths per se, that led OSHA to state in the preamble to the
proposed rule that the Davies, Cooper, and Kano studies cannot serve as
the basis of a meaningful analysis of lead chromate carcinogenicity
(Exs. 7-42; 2-D-1; 7-118). In contrast, a study population that has
confidence limits close to or below 1 would provide evidence to support
the DCC claim that " * * * if lead chromate pigments possess any
carcinogenic potential at all, it must be extremely small" (Ex. 38-
201-1, p. 14) at the exposure levels experienced by that population.
While this standard of evidence has not been met in the epidemiological
literature for pigment workers exposed exclusively to lead chromate
(i.e., the Davies, Cooper, and Kano studies), it is hardly an
"insurmountable barrier" that sets up an impossible standard of proof
for those who contend that lead chromate is not carcinogenic.
Some comments suggested that the Davies, Cooper, and Kano studies
should be combined to derive a summary risk measure for exposure to
lead chromate (see e.g. Ex. 38-201-1, pp. 13-14). However, OSHA
believes that these studies do not provide a suitable basis of meta-analysis.
There is little information with which to assess factors recognized by
epidemiologists as key to meta-analysis, for example sources of bias or
confounding in the individual studies and comparability of exposures and
worker characteristics across studies, and to verify certain conditions
required for comparability of SMRs across these studies (see e.g. Modern
Epidemiology, Rothman and Greenland, p. 655). In addition, the
inclusion criteria and length of follow-up differ across the three
studies. Finally, each of the studies is extremely small. Even if it
were appropriate to calculate a 'summary' SMR based on them, the
precision of this SMR would not be much improved compared to those of
the original studies.
In their written testimony, DCC suggested that OSHA should
aggregate the data from the Davies, Cooper, and Kano studies in order
to determine whether there is a discrepancy between the results of
these three studies, taken together, and OSHA's preliminary risk
assessment (Ex. 38-201-1, pp. 13-14). DCC performed a calculation to
compare OSHA's risk model with the observed lung cancer in the three
cohorts. DCC stated that:
OSHA estimates a chromate worker's risk of dying from lung
cancer due to occupational exposure as about one chance in four * *
* [Assuming that there were about] 200 workers in the Kano study,
the total in the three studies would be 600. A calculation of one
quarter would be 150 deaths. To compensate for a working life of
less than OSHA's 45 years [an assumption of 20 years] provides * * *
a refined estimate of about 70 deaths. An observed number less than
this could be due either to exposures already in practice averaging
much less than the current PEL of 52, or to lead chromate having
much less potential (if any) for carcinogenicity than other
chromates. In any event the actual incidence of death from lung
cancer would appear to be no more than one tenth of OSHA's best
estimate (Ex. 38-201-1, pp. 15-16).
The method suggested by DCC is not an appropriate way to assess the
carcinogenicity of lead chromate, to identify a discrepancy between the
pigment cohort results and OSHA's risk estimates, or to determine an
exposure limit for lead chromate. Among other problems, DCC's
calculation does not make a valid comparison between OSHA's risk
estimates and the results of the Davies, Cooper, and Kano studies.
OSHA's 'best estimate' of lung cancer risk for any given Cr(VI)-exposed
population depends strongly on factors including exposure levels,
exposure duration, population age, and length of follow-up. The 'one in
four' prediction cited by DCC applies to one specific risk scenario
(lifetime risk from 45 years of occupational exposure at the previous
PEL of 52 [mu]g/m\3\). OSHA's best estimate of risk would be lower for
a population with lower exposures (as noted by DCC), shorter duration
of exposure, or less than a lifetime of follow-up. Without adequate
information to adjust for each of these factors, a valid comparison
cannot be drawn between OSHA's risk predictions and the results of the
lead chromate cohort studies.
The importance of accounting for cohort age and follow-up time may
be illustrated using information provided in the Cooper et al. study.
As shown in Table V-11 below, approximately three-fourths of the Cooper
et al. Plant 1 cohort members were less than 60 years old at the end of
follow-up.
For a population of 600 with approximately the same distribution of
follow-up time as described in the Cooper et al. publication (e.g.,
0.4% of workers are followed to age 84, 2% to age 79, etc.), OSHA's
risk model predicts about 3-15 excess lung cancers (making the DCC
assumption that workers are exposed for 20 years at 52 [mu]g/m\3\),
rather than the 70 deaths calculated by the DCC. If the workers were
typically exposed for less than 20 years or at levels lower than 52
[mu]g/m\3\, OSHA s model would predict still lower risk. A precise
comparison between OSHA's risk model and the observed lung cancer risk
in the Davies, Cooper and Kano cohorts is not possible without
demographic, work history and exposure information on the lead chromate
workers. (In particular, note that year 2000 background lung cancer
rates were used in the calculation above, as it was not feasible to
reconstruct appropriate reference rates without work history
information on the cohorts.) However, this exercise illustrates that
DCC's assertion of a large discrepancy between OSHA's risk model and
the available data on workers exposed exclusively to lead chromate is
not well-founded. To make a valid comparison between the OSHA risk
model and the lung cancer observed in the lead chromate cohorts would
require more information on exposure and follow-up than is available
for these cohorts.
OSHA received comments and testimony from NIOSH and others
supporting of the Agency's interpretation of the epidemiological
literature on Cr(VI) color pigments, including lead chromate (Tr. 135-
146, 316, 337, Ex. 40-18-1, p. 2). At the hearing, Mr. Robert Park of
NIOSH stated that the available studies of workers exposed to chromate
pigments show " * * * a general pattern of excess [lung cancer] * * *
" and pointed out that "[i]n several of the studies, lead [chromate]
was by far the major component of production, like 90 percent * * * So
I don't think there is any epidemiological evidence at this point that
gets lead off the hook" (Tr. 337). Regarding the lack of statistically
significant excess lung cancer in several pigment worker cohorts, Mr.
Park identified study attributes that may have obscured an excess in
lung cancer, such as the high percentage of workers lost to follow-up
among immigrant workers in the Davies et al. study (Tr. 337) or a
healthy worker effect in the Hayes et al. study (Tr. 316). Dr. Paul
Schulte of NIOSH explained that
* * * a lot of these studies that appear to be negative were
either of low power or had [some] other kind of conflicting
situation [so] that we can't really consider them truly negative
studies (Tr. 338).
Dr. Herman Gibb testified that the epidemiological studies relied on by
CPMA and DCC to question the carcinogenicity of lead chromate have very
low expected numbers of lung cancer deaths, so they " * * * really
don't have a lot of ability to be able to detect a risk" (Tr. 135-
136). Public Citizen agreed with OSHA's preliminary conclusion that
lead chromate is carcinogenic. Based on the major pigment worker
cohorts identified by OSHA in the Preamble to the Proposed Rule, Public
Citizen's Health Research Group concluded that
* * * inadequately-powered studies, the standardized mortality
ratios for exposed workers are significantly elevated (range 1.5-
4.4) and a relationship between extent of exposure (whether measured
by duration of exposure or factory) generally emerges; [moreover,]
[t]hese studies must be placed in the context * * * of the animal
carcinogenicity studies * * * and the mechanistic studies reviewed
by OSHA (Ex. 40-18-1, p. 2).
Tumor Incidence in Experimental Animals Administered Lead Chromate--
CPMA also claims that the absence of evidence for carcinogenicity found
among the three cited cohorts of lead chromate pigment workers " * * *
is further confirmed by the rat implantation studies of Levy" (Ex. 38-
205, p. 98). They argue that these studies which involved implantation
into rat lungs " * * * indicated no increased incidence of tumors for
lead chromate pigment, although more soluble chromates exhibited
varying degrees of carcinogenicity" (Ex. 38-205, p. 93). They
dismissed other animal studies involving intramuscular and subcutaneous
injection of lead chromate which did report increased incidence of
tumors because they believe these techniques
* * * are of questionable relevance in relation to human
workplace exposure conditions in industry, whereas tests involving
implantation in rat lung * * * are relevant to inhalation in
industrial exposures (Ex. 38-205, p. 93).
In a more recent submission, CPMA remarked that the intramuscular and
subcutaneous injection studies with lead chromate were contradictory
and " * * * problematic in that false positive results frequently
occur during the study procedure (Ex. 47-31, p. 13).
The rat implantation studies of Levy involved the surgical
placement of a Cr(VI)-containing pellet in the left bronchus of an
anesthetized rat (Exs. 10-1; 11-12; 11-2). This pellet procedure was an
attempt to deliver Cr(VI) compounds directly to the bronchial
epithelium and mimic continuous chronic in vivo dosing at the tissue
target site in order to assess the relative ability of different Cr(VI)
compounds to induce bronchogenic carcinoma. Histopathological
evaluation of the rat lung was conducted after a two year exposure
time. In most cases, approximately 100 rats were implanted with a
single pellet for each Cr(VI) test compound. The total lifetime dose of
Cr(VI) received by the animal was generally between 0.2 and 1.0 mg
depending on the compound. The amount of Cr(VI) that actually leached
from the cholesterol pellet and remained near the lung tissue was never
determined. At least 20 different commercially relevant Cr(VI)
compounds ranging from water insoluble to highly water soluble were
tested using this intrabronchial implantation protocol.
The results of these studies are described in preamble section
V.B.7 and tables V-7, V-8, and V-9. Reagent grade lead chromate and six
different lead chromate pigments were tested. The lead chromate
pigments were a variety of different chrome yellows, including a silica
encapsulated chrome yellow, and molybdenum orange. The incidence of
bronchogenic cancer in the rats under this set of experimental
conditions was one percent or less for all the lead chromates tested.
This incidence was not statistically different from the negative
controls (i.e. rats implanted with a cholesterol pellet containing no
test compound) or rats administered either the water-insoluble barium
chromate or the highly soluble chromic acid and sodium dichromate. The
percent incidence of bronchogenic cancer in lead chromate-treated rats
was substantially less than that of rats treated with slightly soluble
strontium chromates (about 52 percent) and calcium chromate (24
percent). The type of bronchogenic cancer induced in these experiments
was almost entirely squamous cell carcinomas.
OSHA does not agree with the CPMA position that absence of a
significant tumor incidence in the intrabronchial implantation studies
confirms that lead chromates lack carcinogenic activity and, therefore,
should not be subject to the OSHA Cr(VI) standard. The bioassay
protocol used approximately 100 test animals per experimental group.
This small number of animals limits the power of the bioassay to detect
tumor incidence below three to four percent with an acceptable degree
of statistical confidence. Three of the lead chromates, in fact,
produced a tumor incidence of about one percent (e.g. 1 tumor in 100
rats examined) which was not statistically significant. The researchers
only applied a single 2 mg [approximately 0.3 mg Cr(VI)] dose of lead
chromate to the bronchus of the rats. Since it was not experimentally
confirmed that the lead chromate pigments were able to freely leach
from the cholesterol pellet, the amount of Cr(VI) actually available to
the lung tissue is not entirely clear. Therefore, OSHA believes a more
appropriate interpretation of the study findings is that lead chromates
delivered to the respiratory tract at a dose of about 0.3 mg Cr(VI)
(maybe lower) lead to a less than three percent tumor incidence.
However, OSHA agrees that the intrabronchial implantation protocol
does provide useful information regarding the relative carcinogenicity
of different Cr(VI) compounds once they are delivered and deposited in
the respiratory tract. No other study examines the carcinogenicity of
such a broad range of commercial Cr(VI) compounds under the same
experimental conditions in the relevant target organ to humans (i.e.
respiratory tract) following in vivo administration. OSHA agrees with
CPMA that the results of this study provide credible
evidence that water insoluble lead chromates are less carcinogenic than
some of the more moderately soluble chromates. Specifically, this
includes the slightly soluble zinc chromates (e.g. zinc yellow, zinc
potassium chromates, basic zinc chromates) as well as strontium
chromate and calcium chromate. Intrabronchial implantation of chromic
acid and other highly soluble Cr(VI) salts, such as sodium chromates,
did not induce a significant number of tumors. Therefore, these
experiments do not indicate lead chromate are less carcinogenic than
the highly water soluble Cr(VI) compounds.
If the histopathology data from the intrabronchial implantation is
examined more closely, all lead chromates increased the incidence of
squamous metaplasia relative to controls, and, for some lead chromates,
squamous dysplasia of the bronchial epithelium occurred (Table 2, Ex.
11-2). Squamous metaplasia and dysplasia are generally considered to be
transformed cellular states from which a neoplasm (e.g. carcinomas) can
arise (Ex. 11-12). Increased squamous metaplasia was common among all
tested Cr(VI) compounds but not among Cr(III)-containing materials or
the negative controls (Ex. 11-12). The increased metaplasia induced by
lead chromates is unlikely to be due to bronchial inflammation since
the degree of inflammation was no greater than that observed in the
cholesterol-implanted controls (Table 2, Ex. 11-2).
The squamous metaplasia and dysplasia in the rat lung model
following low dose lead chromate administration is consistent with a
low carcinogenic response (e.g. incidence of one percent or less) not
able to be detected under the conditions of the animal bioassay. This
explanation is supported by studies (discussed later in the section)
that show lead chromate can enter lung cells, damage DNA, and cause
genotoxic events leading to neoplastic transformation.
Lead chromate carcinogenicity is also supported by the animal
studies that CPMA dismisses as problematic and of questionable
relevance. These studies administered lead chromates to rodents by
either the subcutaneous (Exs. 8-25, 5-2, 8-37) or intramuscular routes
(Ex. 10-2). While OSHA agrees that these routes may be less relevant to
occupational inhalation than implantation in the respiratory tract, the
studies exposed rats to a larger dose of lead chromate. The higher
amounts of Cr(VI) produced a significant incidence of tumors at the
injection site (see section V.B.7.c).
The lead chromate pigments, chrome yellow and chrome orange,
induced injection site rhabdomyosarcomas and fibrosarcomas in 65
percent of animals following a single 30 mg injection in a saline
suspension (Ex. 8-37). The rats received a roughly ten fold higher dose
of Cr(VI) than in the intrabronchial bioassay. Rats injected with
saline alone did not develop injection site tumors. Only two percent or
less of rats receiving equal quantities of the inorganic pigments iron
yellow and iron red developed these tumors. The iron oxides are not
considered to be carcinogenic and do not give a significant neoplastic
response in this bioassay. OSHA has no reason to believe the
experimental procedure was problematic or given to frequent false
positives.
A similarly high incidence (i.e. 70 percent) of the same injection
site sarcomas were found in an independent study in which rats were
injected intramuscularly with reagent grade lead chromate once a month
for nine months (Ex. 10-2). Each injection contained approximately 1.3
mg of Cr(VI) and the total dose administered was over 30 times higher
than the intrabronchial implantation. The lead chromate was
administered in a glycerin vehicle. The vehicle produced less than a
two percent incidence of injection site sarcomas when administered
alone.
Contrary to statements by Eurocolour (Ex. 44-3D), lead chromate did
produce a low incidence of site-of-contact tumors in rats in an earlier
study when administered by either intramuscular or intrapleural
implantation (Ex. 10-4). There was no tumor incidence in the control
animals. The dose of lead chromate in this early publication was not
stated.
Based on the increase in pre-neoplastic changes from the single low
dose intrabronchial implantation and the high incidence of malignant
tumors resulting from larger doses administered by subcutaneous and
intramuscular injection, it is scientifically reasonable to expect that
larger doses of lead chromate may have produced a higher incidence of
tumors in the more relevant intrabronchial implantation procedure. The
highly soluble sodium dichromate produced a small (statistically
insignificant) incidence of squamous cell carcinoma (i.e. one percent)
upon single low dose intrabronchial implantation similar to the lead
chromates (Ex. 11-2). In another study, sodium dichromate caused a
significant 17 percent increase in the incidence of respiratory tract
tumors when instilled once a week for 30 months in the trachea of rats
(Ex. 11-7). The weekly-administered dose for this repeated instillation
was about \1/5\th the dose of that used in the intrabronchial
implantation assay but the total administered dose after 30 months was
about 25 times higher. Rats that received a lower total dose of sodium
dichromate or the same total dose in more numerous instillations (i.e.
lower dose rate) developed substantially fewer tumors that were
statistically indistinguishable from the saline controls. A third study
found a 15 percent increase (not statistically significant) in lung
tumor incidence when rats repeatedly inhaled aerosolized sodium
dichromate for 18 months at the highest air concentrations tested (Ex.
10-11). These sodium dichromate studies are further described in
section V.B.7.a. The findings suggest that the lack of significant
carcinogenic activity in the intrabronchial implantation study
reflects, in part, the low administered dose employed in the bioassay.
In his written testimony to OSHA, Dr. Harvey Clewell directly
addressed the issue of interpreting the absence of carcinogenicity in
an animal study as it relates to significant risk.
First, the ability to detect an effect depends on the power of
the study design. A statistically-based No Observed Adverse Effect
Level (NOAEL) in a toxicity study does not necessarily mean that
there is no risk of adverse effect. For example, it has been
estimated that a NOAEL in a typical animal study can actually be
associated with the presence of an effect in as many as 10% to 30%
of the animals. Thus the failure to observe a statistically
significant increase in tumor incidence at a particular exposure
does not rule out the presence of a substantial carcinogenic effect
at that exposure * * *. Similarly the failure of Levy et al. (1986)
to detect an increase in tumors following intrabronchial
instillation of lead chromate does not in itself demonstrate a lack
of carcinogenic activity for that compound. It only demonstrates a
lower activity than for other compounds that showed activity in the
same experimental design. Presumably this lower activity is
primarily due to its low solubility; evidence of solubilization,
cellular uptake, and carcinogenic activity of this compound [i.e.
lead chromate] is provided in other studies (Maltoni et al. 1974,
Furst et al., 1976, Blankenship et al., 1997; Singh et al., 1999;
Wise et al., 2004) (Ex. 44.5, p. 13-14).
OSHA agrees with Dr. Clewell that the inability to detect a
statistically significant incidence of tumors in one study that
administers a single low dose of lead chromate to a limited number of
animals is not evidence that this Cr(VI) compound lacks carcinogenic
activity. This is especially true when there exists an elevation in
pre-neoplastic lesions and other studies document significant
tumor incidence in animals administered higher doses of lead chromate.
Cellular Uptake and Genotoxicity of Lead Chromate--CPMA disputes
the many studies that report lead chromate to be genotoxic or
clastogenic in cellular test systems (Exs. 35-162; 12-5; 35-119; 35-
188; 35-132; 35-68; 35-67; 35-115; 35-66; 47-22-1; 47-12-3; 35-327; 35-
436). They claim that the studies inappropriately solubilized the lead
chromate " * * * in non-biological conditions such as strong alkali or
strong acid that causes the chemical breakdown of the lead chromate
crystal" (Ex. 38-205, p. 94) and the "lead chromate had been
dissolved * * * using aggressive substances" (Ex. 38-205, p. 99). In a
later submission, CPMA states state that some of the cellular studies
used reagent grade lead chromate that is only >=98 percent pure and may
contain up to 2 percent soluble chromate (Ex. 47-31, p. 11). They
speculate that the interactions (e.g. chromate ion uptake, chromosomal
aberrations, DNA adducts, etc.) described in studies using cell
cultures treated with lead chromate are either due to the presumed
contamination of soluble chromate or some other undefined "reactive
nature" of lead chromate. CPMA adds that " * * * the studies
referenced by OSHA [that use reagent grade lead chromate] have no
relevance to occupational exposures to commercial lead chromate
pigments" (Ex. 38-205, p. 11-12).
OSHA agrees that studies involving lead chromate pre-solubilized in
solutions of hydrochloric acid, sodium hydroxide or other strong acids
and bases prior to treatment with cells are not particularly relevant
to the inhalation of commercial lead chromate particulates. However,
several relevant cellular studies have demonstrated that lead chromate
particulates suspended in biological media and not can enter lung
cells, damage DNA, and cause altered gene expression as described
below.
Beginning in the late 1980s, there has been a consistent research
effort to characterize the genotoxic potential of lead chromate
particulate in mammalian cells. The lead chromate was not pre-
solubilized prior to cell treatment in any of these investigations. In
most of the studies, lead chromate particles were rinsed with water and
then acetone. The rinses cleansed the particles of water- and acetone-
soluble contaminants before cell treatment. This served to remove any
potential water-soluble Cr(VI) present that might confound the study
results. In most instances, the lead chromate particles were filtered,
stirred or sonicated in suspension to break up the aggregated particles
into monomeric lead chromate particulates. These lead chromate
particulates were primarily less than 5 [mu]m in diameter. This is
consistent with the inhaled particle size expected to deposit in the
bronchial and alveolar regions of the lung where lung cancer occurs.
Air-dried lead chromate particulates were introduced to the cell
cultures in a suspension of either saline-based media or acetone. Lead
chromate particulate is considered to be insoluble in both solvents so
significant solubilization is not expected during the process of
creating a homogenous suspension.
The initial research showed that lead chromate particulate
morphologically transformed mouse and hamster embryo cells (Exs. 35-
119; 12-5). One study tested a variety of lead chromate pigments of
different types (e.g. chrome yellows, chrome oranges, molybdate
oranges) as well as reagent grade lead chromate (Ex. 12-5). The
transformed cells displayed neoplastic properties (e.g. growth in soft
agar) and were tumorigenic when injected into animals (Ex. 35-119; 12-
5). While lead chromate particulate transformed mouse embryo cells, it
is important to note that lead chromate particulate was not found to be
mutagenic in these cells suggesting that other types of genetic lesions
(e.g. clastogenicity) may be involved (Ex. 35-119).
Follow-on research established that lead chromate particulate
caused DNA-protein crosslinks, DNA strand breaks, and chromosomal
aberrations (i.e. chromatid deletions and achromatic lesions combined)
in mammalian cells rather than DNA nucleotide binding often associated
with base substitution and frameshift mutations captured in a standard
Ames assay (Exs. 35-132; 35-188). This distinguishes lead chromate
particulate from high concentrations of soluble Cr(VI) compounds or
pre-solubilized lead chromate which can cause these mutations.
Lead chromate particulate enters mammalian embryo cells by two
distinct pathways (Ex. 35-68). It partially dissolves in the culture
medium (i.e. biological saline solution) to form chromate ion, which is
then transported into the cell. The rate of particle dissolution was
shown to be time- and concentration-dependent. The measured chromate
ion concentration was consistent with that predicted from the lead
chromate solubility constant in water. Lead chromate particulates were
shown to adhere to the embryo cell surface enhancing chromate ion
solubilization leading to sustained intracellular chromium levels and
measurable chromosomal damage (Ex. 35-67).
Lead chromate particulates are also internalized into embryo cells,
without dissolution, by a phagocytic process (Ex. 35-68). The lead
chromate particles appeared to remain undissolved in tight vacuoles
(i.e. phagosomes) within the cell over a 24 hour period. Treatment of
embryo cells with lead chromate particulates in the presence of a
reducing agent (i.e. ascorbate) substantially reduced cellular uptake
of dissolved chromate ions and the chromosomal damage, but did not
impact the internalization of lead chromate particulates (Ex. 35-68).
This suggests that chromosomal damage by lead chromate was the result
of extracellular particle dissolution and not internalization under the
particular experimental conditions. Embryo cell treatment with large
amounts of lead glutamate that produced high intracellular lead in the
absence of Cr(VI) did not cause chromosomal damage further implicating
intracellular chromium as the putative clastogenic agent (Ex. 35-67).
As the ability to maintain human tissue cells in culture improved
in the 1990s, dissolution and internalization of lead chromate
particulates, uptake of chromate ion, and the resulting chromosomal
damage were verified in human lung cells (Exs. 35-66; 47-22-1; 47-12-3;
35-327; 35-436). Lead chromate particulates are internalized, form
chromium adducts with DNA, and trigger dose-dependent apoptosis in
human small airway epithelial cells (Ex. 35-66). They also cause dose-
dependent increases in intracellular chromium, internalized lead
chromate particulates and chromosomal damage in human lung fibroblasts
(Exs. 47-22-1; 47-12-3). The chromosomal damage from lead chromate in
these human lung cells is dependent on the extracellular dissolution
and cell uptake of the chromate, rather than lead, in a manner similar
to dilute concentrations of the highly soluble sodium chromate (Ex. 47-
12-3; 35-327). Another water insoluble Cr(VI) compound, barium chromate
particulate, produces very similar responses in human lung fibroblasts
(Ex. 35-328). Human lung macrophages can phagocytize lead chromate
particulates and trigger oxidation-reduction of Cr(VI) to produce
reactive oxygen species capable of damaging DNA and altering gene
expression (Ex. 35-436).
OSHA finds these recent studies to be carefully conceived and
executed by reputable academic laboratories. The scientific findings
have been published in well-respected peer reviewed
molecular cancer and toxicology journals, such as Carcinogenesis (Exs.
12-5, 35-68), Cancer Research (Ex. 35-119), Toxicology and Applied
Pharmacology (Exs. 35-66; 25-115), and Mutation Research (Exs. 35-132;
47-22-1; 35-327). Contrary to statements by CPMA, the results indicate
that lead chromate particulates are able to dissociate in the presence
of biological media without the aid of aggressive substances. The
resulting chromate ion is bioavailable to enter lung cells, damage
genetic material and initiate events critical to carcinogenesis. These
effects can not be attributed to small amounts of soluble chromate
contaminants since these substances are usually removed as part of the
test compound preparation prior to cell treatment.
As one of the study authors, Dr. John Wise of the University of
Southern Maine, stated in his post-hearing comments:
At no time did we dissolve lead chromate particles prior to
administration. At the initial onset of the administration of lead
chromate particles in our studies, the cells encountered intact lead
chromate particles. Any dissolution that occurred was the natural
result of the fate of lead chromate particles in a biological
environment (Ex. 47-12, p. 3).
Other scientists concurred that the methods and findings of the
cellular research with lead chromate were reasonable. Dr. Kathleen
MacMahon, a biologist from NIOSH stated:
NIOSH believes that the methods that were used in the [lead
chromate] studies were credible and we support the results and
conclusions from those studies (Tr. 342).
Dr. Clewell said:
As I recall, it [lead chromate particles] was suspended in
acetone and ultrasonically shaken to reduce it to submicron
particles, which seems like a reasonably good thing to do. There are
actually a couple of studies besides the Wise studies that have
looked at the question of the uptake of lead chromate. I have looked
at those studies and I don't really see any basic flaws in what they
did. It is obviously a challenge to reproduce inhalation exposure in
vitro (Tr. 180-181).
Chromosal Aberrations and Lead Chromate--Several submissions contained
testimony from another researcher, Dr. Earle Nestmann of CANTOX Health
Sciences International, that criticized the methodology and findings of
a study published by the research group at the University of Southern
Maine (Exs. 34-6-1; 38-205D; 47-12-1; 47-22). Dr. Nestmann viewed as
inappropriate the practice of combining the chromatid deletions and
achromatic lesions together as chromosomal aberrations. He indicated
the standard practice was to score these two types of lesions
separately and that only the deletions had biological relevance.
According to Dr. Nestmann, achromatic lesions are chromatid gaps (i.e.
lesion smaller than the width of one chromatid) that have no
clastogenic significance and serve to inflate the percentage of cells
with chromosomal aberrations (i.e. chromatid deletions or breaks). Dr.
Nestmann criticized the studies for not including a positive control
group that shows the experimental system responds to a 'true'
clastogenic effect (i.e. a compound that clearly increases chromosomal
deletions without contribution from chromatid gaps).
Dr. John Wise, the Director of the research laboratory at the
University of Southern Maine, responded that distinguishing chromatid
gaps from breaks is a subjective distinction (e.g. requiring judgment
as to the width of a lesion relative to the width of a chromatid) and
pooling these lesions simply reduces this potential bias (Ex. 47-12;
47-12-1). He stated that there is no consensus on whether gaps should
or should not be scored as a chromosomal aberration and that gaps have
been included as chromosomal aberrations in other publications. Dr.
Wise also points out that achromatic lesions have not been shown to
lack biological significance and that the most recent research
indicates that they may be related to DNA strand breaks, a
scientifically accepted genotoxic endpoint. Dr. Wise further believed
that a positive control was unnecessary in his experiments since the
purpose was not to determine whether lead chromate was a clastogenic
agent, which had already been established by other research. Rather,
the purpose of his studies was to assess Cr(VI) uptake and chromosomal
damage caused by water-insoluble lead chromate compared to that of
highly water soluble sodium chromate using a relevant in vitro cell
model (i.e. human lung cells).
OSHA is not in a position to judge whether achromatic lesions
should be scored as a chromosomal aberration. However, OSHA agrees with
Dr. Nestmann that combining gaps and breaks together serves to increase
the experimental response rate in the studies. Given the lack of
consensus on the issue, it would have been of value to record these
endpoints separately. OSHA is not aware of data that show achromatic
gaps to be of no biological significance. The experimental data cited
above indicate that soluble and insoluble Cr(VI) compounds clearly
increase achromatic gaps in a concentration-dependent manner. The
chromatid lesions (gaps and breaks) may be chromosomal biomarkers
indicative of genetic damage that is critical to neoplastic
transformation. Furthermore, OSHA agrees with Dr. Wise that other
evidence establishes lead chromate as an agent able to cause DNA damage
and transform cells. The Agency considers the use of sodium chromate-
treated cells in the above set of experiments to be the appropriate
comparison group and does not find the absence of an additional
positive control group to be a technical deficiency of the studies.
OSHA considers the research conducted at the University of Southern
Maine documenting chromosomal damage in human lung cells following
treatment with lead chromate particulates to be consistent with results
from other studies (see Section V.B.8) and, thus, contributes to the
evidence that water insoluble lead chromate, like other chromates, is
able to enter lung cells and damage DNA.
In post-hearing comments, CPMA provided a Canadian research
laboratory report that tested the lead chromate Pigment Yellow 34 for
chromosomal aberrations in a hamster embryo cell system (Ex. 47-3,
appendix C). The research was sponsored by DCC and its representative
Dr. Nestmann. Lead chromate particles over the concentration range of
0.1 [mu]/cm2 to 10 [mu]/cm2 were reported to not
induce chromosomal aberrations under the experimental test conditions.
Chromatid structural and terminal gaps were not scored as aberrations
in this study, even though the percentage of cells with these lesions
increased in a dose-dependent manner from two percent in the absence of
lead chromate to over thirteen percent in cells treated with 1 [mu]/
cm2 lead chromate pigment particles.
This result is consistent with other experimental data that show
lead chromate particulates cause chromosomal lesions when administered
to mammalian embryo cells (Exs. 35-188; 35-132; 35-68; 35-67). The key
difference is how the various researchers interpreted the data. The
George Washington University group (i.e. Pateirno, Wise, Blankenship et
al.) considered the dose-dependent achromatic lesions (i.e. chromatid
gaps) as a clastogenic event and included them as chromosomal damage.
The Canadian test laboratory (i.e. Nucrotechnics) reported achromatic
lesions but did not score them as chromosomal aberrations. Reporting
achromatic lesions but not scoring them as chromosomal aberrations is
consistent with regulatory test guidelines as currently recommended by
EPA and OECD. The Nucrotechnics data suggest that the tested lead chromate
pigment caused a similar degree of chromosomal damage (i.e. dose-dependent
achromatic lesions and chromosomal aberrations combined) in mammalian cells.
This result was similar to results produced by reagent grade lead chromate in
previous studies.
Mutagenicity and Lead Chromate--CPMA also relied on a study that
reported a lack of mutagenicity for lead chromate pigments in a
bacterial assay using Salmonella Typhimurium TA 100 (Ex. 11-6). As
previously mentioned, this assay specifically measures point and
frameshift mutations usually caused by DNA adduct formation. The assay
is not sensitive to chromosomal damage, DNA strand breaks, or DNA
crosslinks most commonly found with low concentrations of Cr(VI)
compounds. Large amounts (50 to 500 [mu]g/plate) of highly soluble
sodium dichromate and slightly soluble calcium, strontium, and zinc
chromates, were found to be mutagenic in the study, but not the water
insoluble barium chromate and lead chromate pigments. However,
mutagenicity was observed when the acidic chelating agent,
nitrilotriacetic acid (NTA), was added to the assay to help solubilize
the water insoluble Cr(VI) compounds. The chelating agent was unable to
solubilize sufficient amounts of lead chromate pigments to cause
bacterial mutagenicity, if these pigments were more than five percent
encapsulated (weight to weight) with amorphous silica.
OSHA finds the results of this study to be consistent with the
published literature that shows Cr(VI) mutagenicity requires high
concentrations of solubilized chromate ion (Exs. 35-118; 35-161). Large
amounts of water-soluble and slightly soluble Cr(VI) compounds produce
a mutagenic response in most studies since these Cr(VI) compounds can
dissociate to achieve a high concentration of chromate ion. Insoluble
lead chromate usually needs to be pre-solubilized under acidic or
alkaline conditions to achieve sufficient chromate ion to cause
mutagenicity (Ex. 35-162). The above study found highly and slightly
soluble chromates to be mutagenic as well as water insoluble lead
chromate pigments pre-solubilized with NTA. The lack of mutagenicity
for silica encapsulated lead chromate pigments under these experimental
conditions is likely the result of their greater resistance to acidic
digestion than unencapsulated lead chromate pigment.
Failure to elicit a mutagenic response in a bacterial assay, with
or without NTA, is not a convincing demonstration that chromate ion can
not partially dissociate from encapsulated lead chromate in biological
media, enter mammalian cells, and elicit other types of genotoxicity.
As described above, chromosomal damage, believed to result from DNA
strand breaks and crosslinks, appears to be the critical genotoxic
endpoint for low concentrations of Cr(VI) compounds. Research has shown
that lead chromate and lead chromate pigment particulates in biological
media can cause chromosomal lesions and cell transformation without the
aid of strongly acidic or basic substances (Exs. 12-5; 35-119; 35-188;
35-132; 35-68; 35-67; 47-12-3; 35-327). While silica-encapsulated lead
chromate pigments have not been as thoroughly investigated as the
unencapsulated pigments or reagent grade lead chromate, one study
reported that lead silicochromate particles did have low solubility in
biological culture media and transformed hamster embryo cells (Ex. 12-
5).
Information is not available in the record to adequately
demonstrate the efficiency and stability of the encapsulation process,
despite OSHA statements that such information would be of value in its
health effects evaluation and its request for such information (69 FR
59315-59316, 10/4/2004; Ex. 2A). In the absence of data to the
contrary, OSHA believes it prudent and plausible that encapsulated lead
chromate pigments are able to partially dissociate into chromate ion
available for lung cell uptake and/or be internalized in a manner
similar to other lead chromate particulates. The resulting
intracellular Cr(VI) leads to genotoxic damage and cellular events
critical to carcinogenesis.
Public Comments on Carcinogenicity of Slightly Water Soluble Cr(VI)
Compounds--In its written comments to the NPRM, Boeing Corporation
stated that "there is no persuasive scientific evidence for OSHA's
repeated assertion that low solubility hexavalent chromium compounds
[e.g. strontium and zinc chromates] are more potent carcinogens than
[highly] soluble [Cr(VI)] compounds" (Ex. 38-106, p. 2). Boeing and
others in the aerospace industry are users of certain slightly soluble
Cr(VI) compounds, particularly strontium chromate, found in the
protective coatings applied to commercial and military aircraft.
Boeing argues that OSHA, along with IARC, ACGIH and others, have
exclusively relied on intrabronchial implantation studies in animals
that are both not representative of inhalation exposures in the
workplace and are not consistent with the available animal inhalation
data (Ex. 38-106-2, p. 26). Boeing asserts that there is no evidence
that slightly soluble chromates behave differently in terms of their
absorption kinetics than highly soluble chromates when instilled in the
lungs of rats (Ex. 38-106-2, p. 19). Boeing believes the OSHA position
that slightly soluble Cr(VI) compounds are retained in the lung,
associate with cells, and cause high uptake or high local
concentrations to be inconsistent with other data showing these Cr(VI)
compounds quickly disperse in water (Ex. 38-106-2, p. 26). Boeing
concludes:
There is no basis for the conclusion that low solubility [i.e.
slightly soluble] chromates could be more potent than [highly]
soluble, and some evidence the opposite may be the case. As a worst
case OSHA should conclude that there is inadequate evidence to
conclude that [highly] soluble and low-solubility compounds differ
in carcinogenic potency. It is critical that OSHA maintain a
distinction between low-solubility chromates and highly insoluble
chromates based on this data. (Ex. 38-106-2, p. 26)
As noted earlier, OSHA as well as other commenters agree with
Boeing that the animal intrabronchial and intratracheal instillation
studies are not appropriate for quantitatively predicting lung cancer
risk to a worker breathing Cr(VI) dust and aerosols. However, many
stakeholders disagreed with the Boeing view and believed these animal
studies can be relied upon as qualitative evidence of relative
carcinogenic potency. CPMA, which relies on the rat intrabronchial
implantation results as evidence that lead chromate is non-
carcinogenic, states "tests involving implantation in rat lung, as
carried out by Levy et al. in 1986, are relevant to inhalation in
industrial exposures" (Ex. 38-205, p. 93). In their opening statement
NIOSH agreed with the preliminary OSHA determination that "the less
water soluble [Cr(VI)] compounds may be more potent than the more water
soluble [Cr(VI)] compounds" (Tr. 299). NIOSH identified the rat
intrabronchial implantation findings as the basis for their position
that the slightly soluble Cr(VI) compounds appear to be more
carcinogenic than the more soluble and insoluble Cr(VI) compounds (Tr.
334). Dr. Clewell testified that:
Some animal studies suggest the solubility of hexavalent
chromium compounds influences their carcinogenic potency with
slightly soluble compounds having the higher potencies than highly
soluble or insoluble compounds. However, the evidence is inadequate
to conclude that specific hexavalent chromium compounds are not
carcinogenic. Moreover the designs of the studies were not
sufficient to quantitatively estimate comparative potencies (Ex. 44-5, p. 15).
Respiratory Tract Instillation of Slightly Soluble Cr(VI) Compounds in
Rats--OSHA agrees that animal intrabronchial and intratracheal
implantation studies provide persuasive evidence that slightly soluble
Cr(VI) are more carcinogenic than the highly soluble Cr(VI) compounds.
As mentioned previously, these studies provide useful information
regarding the relative carcinogenicity of different Cr(VI) compounds
once they are delivered and deposited in the respiratory tract. For
example, one study examined the carcinogenicity of over twenty
different Cr(VI) compounds in rats, spanning a broad range of
solubilities, under the same experimental conditions in the relevant
target organ to humans (i.e. respiratory tract) following in vivo
administration (Ex. 11-2). A single administration of each Cr(VI) test
compound was instilled in the lower left bronchus of approximately 100
rats. The results were dramatic. Roughly 50 and 25 percent of the rats
receiving the slightly soluble strontium and calcium chromates,
respectively, developed bronchogenic carcinoma. No other Cr(VI)
compounds produced more than five percent tumor incidence. The highly
soluble sodium dichromate under the same experimental conditions caused
bronchogenic carcinoma in only a single rat.
The higher relative potency of the slightly soluble calcium
chromate compared to the highly soluble sodium dichromate was confirmed
in another study in which each test compound was instilled at a low
dose level (i.e., 0.25 mg/kg) in the trachea of 80 rats five times
weekly for 30 months (Ex. 11-7). Using this experimental protocol, 7.5
percent of the slightly soluble calcium chromate-treated animals
developed brochioalveolar adenomas while none of the highly soluble
sodium dichromate-treated rats developed tumors. The tumor incidence at
this lower dose level occurred in the absence of serious lung pathology
and is believed to reflect the tumorigenic potential of the two Cr(VI)
compounds at workplace exposures of interest to OSHA. On the other
hand, a five-fold higher dose level that caused severe damage and
chronic inflammation to the rat lungs produced a similar fifteen
percent lung tumor incidence in both calcium and sodium chromate
treated rats. OSHA, as well as the study authors, believe the later
tumor response with the higher dose level did not result from direct
Cr(VI) interaction with cellular genes, but, instead, was primarily
driven by the cellular hyperplasia secondary to the considerable damage
to the lung tissue. Boeing also seems to attribute this result to
tissue damage stating "most of the tumors were found in areas of
chronic inflammation and scarring, suggesting an effect that is
secondary to tissue damage" (Ex. 38-106-2, p. 21).
OSHA does not agree with some study interpretations advanced by
Boeing in support of their position that slightly soluble Cr(VI)
compounds are no more carcinogenic than highly soluble Cr(VI). For
example, Boeing claims that the intrabronchial implantation experiments
cannot be relied upon because the results do not correspond to findings
from animal inhalation studies (Ex. 38-106-2, p. 24-25). The primary
basis for the Boeing comparison were two rodent bioassays that reported
tumor incidence from the inhalation of different Cr(VI) compounds (Exs.
10-8; 10-11). In one study over 200 mice inhaled slightly soluble
calcium chromate powder for five hours per day, five days per week for
roughly two years (Ex. 10-8). In the other study, 19 rats inhaled an
aqueous sodium dichromate liquid aerosol virtually around the clock for
22 hours a day, seven days a week for eighteen months (Ex. 10-11). The
two studies reported a similar tumor incidence despite the lower total
weekly Cr(VI) dose of sodium dichromate in the second study. OSHA
believes the vastly different experimental protocols employed in these
studies do not allow for a legitimate comparison of carcinogenic
potency between Cr(VI) compounds. First, mouse and rat strains can
differ in their susceptibility to chemical-induced lung tumors. Second,
the proportion of respirable Cr(VI) may differ between a liquid aerosol
of aqueous sodium dichromate mist and an aerosol solid calcium chromate
particles suspended in air. Third, the opportunity for Cr(VI) clearance
will undoubtedly differ between a Cr(VI) dose inhaled nearly
continuously (e.g., 22 hours per day, seven days a week) and inhaled
intermittently (e.g., five hours a day, five days a week) over the
course of a week. These experimental variables can be expected to have
a major influence on tumor response and, thus, will obscure a true
comparison of carcinogenic potency. Boeing acknowledges that "these
[inhalation] studies used very different protocols and are not directly
comparable" (Ex. 38-106-2, p.24). On the other hand, slightly soluble
Cr(VI) compounds were found to cause a greater incidence of lung tumors
than highly soluble Cr(VI) compounds in two independent studies in
which the test compounds were instilled under the same dosing regime in
the same rodent models in research specifically designed to assess
relative Cr(VI) carcinogenic potency (Exs. 11-2; 11-7). Therefore, OSHA
believes any apparent lack of correspondence between animal inhalation
and instillation studies is due to an inability to compare inhalation
data from vastly different experimental protocols and should not
diminish the relevance of the instillation findings.
Epidemiological Studies of Slightly Soluble Cr(VI) Compounds--
Boeing further argues that the greater carcinogenic potency experienced
by rats intrabronchially instilled with slightly soluble chromates
compared to rats instilled with highly soluble and water-insoluble
Cr(VI) compounds "do not correspond qualitatively to observed lung
cancer in occupational exposure" (Ex. 38-106-2, p. 21). Several other
industry stakeholders disagree. In explaining the excess lung cancer
mortality among pigment production workers, CPMA commented:
[water-insoluble] Lead chromate pigments must be differentiated
from [slightly soluble] zinc chromate corrosion inhibitor additives,
which are consistently shown to be carcinogenic in various studies.
When [water insoluble] lead chromate and [slightly soluble] zinc
chromate exposures occur simultaneously, there appears to be a
significant cancer hazard. However, when lead chromate pigments
alone are the source of chromium exposure, a significant cancer
response has never been found (Ex. 38-205, p. 91).
In explaining the excess lung cancer mortality among chromate
production workers in the Gibb and Luippold cohorts, the Electric Power
Research Institute states that:
One important distinction is that workers of the historical
chromate production industry were exposed to sparingly soluble forms
of calcium chromate in the roast mix, which are recognized to have
greater carcinogenic potential as compared to soluble forms of
Cr(VI) based on animal implantation studies (Ex. 38-8, p. 12).
Deborah Proctor of Exponent also testified:
Several studies of chromate production worker cohorts have
demonstrated that the excess cancer risk is reduced when less lime
is added to the roast mixture, reducing worker exposure to the
sparingly soluble calcium chromate compounds" (Ex. 40-12-5).
OSHA believes there is merit to the above comments that workplace
exposure to slightly soluble Cr(VI) compounds may have contributed to
the higher lung cancer mortality in both pigments workers producing
mixed zinc and lead chromate pigments as well as
chromate production workers exposed to calcium chromate from high lime
production processes in the 1930s and 1940s. Other factors, such as
greater Cr(VI) exposure, probably also contributed to the higher lung
cancer mortality observed in these cohorts. In any case, these
epidemiological findings support the Boeing contention that the
epidemiological findings are inconsistent with the results from animal
intrabronchial implantation studies (Ex. 38-106-2, p. 26).
Clearance, Retention, and Dissolution of Slightly Soluble Cr(VI)
Compounds in the Lung--Boeing argues that animal experiments that
examined the absorption, distribution and excretion of Cr(VI) compounds
after intratracheal instillation of Cr(VI) compounds in rats do not
show that highly soluble Cr(VI) is cleared more rapidly or retained in
the lung for shorter periods than slightly soluble Cr(VI) compounds
(Ex. 38-106-2, p. 18-19). The results of one study found that larger
amounts of water-insoluble lead chromate were retained in the lungs of
rats at both 30 minutes and at 50 days after instillation than for
highly soluble sodium chromate or slightly soluble zinc chromate (Ex.
35-56). Although the authors concluded that slightly soluble zinc
chromate was more slowly absorbed from the lung than the highly soluble
sodium chromate, the excretion and distribution of the absorbed
chromium from the zinc and sodium chromate instillations was similar.
Furthermore, there was little difference in the amounts of zinc and
sodium chromate retained by the lung at the two extreme time points
(e.g., 30 minutes and 50 days) measured in the study. OSHA agrees with
Boeing that these findings indicate slower clearance and longer
retention in the lung of the water insoluble lead chromate relative to
highly soluble sodium chromate, but not in the case of the slightly
soluble zinc chromate. Slower clearance and longer residence time in
the lung will generally enhance carcinogenic potential assuming other
dosimetric variables such as lung deposition, Cr(VI) concentration at
the lung cell surface, and dissociation into chromate ion are
unchanged.
Boeing asserts that a study of strontium chromate dissociation from
paint primer contradicts the notion that slightly soluble are more
likely than highly soluble Cr(VI) compounds to concentrate and
dissociate at the lung cell surface (Ex. 38-106-2, p. 25). This
experimental research found that roughly 75 and 85 percent of strontium
chromate contained in metal surface primer coating particles was
solubilized in water after one and 24 hours, respectively (Ex. 31-2-1).
The primer particles were generated using a high volume, low pressure
spray gun according to manufacturer specifications, and collected in
water impingers. The authors concluded that their study demonstrated
that chromate dissociation from primer particles into the aqueous fluid
lining lung cells would be modestly hindered relative to highly water
soluble Cr(VI) aerosols.
The slower dissociation of the slightly soluble Cr(VI) compound,
strontium chromate, plausibly explains its higher carcinogenicity in
animal implantation studies. The 'modest hindrance' allows the
undissociated chromate to achieve higher concentrations at the surface
of the lung cells facilitating chromate transport into the cell. The
unhindered, instantaneous dispersion of highly water soluble chromates
in aqueous fluid lining of the respiratory tract is less likely to
achieve a high chromate concentration at the lung cell membrane. OSHA
believes the results of the above study support, not contradict, that
slightly soluble Cr(VI) may lead to higher chromium uptake into lung
cells than highly soluble Cr(VI) compounds.
In summary, slightly soluble Cr(VI) compounds have consistently
caused higher lung tumor incidence in animal instillation studies
specifically designed to examine comparative carcinogenic potency in
the respiratory tract. The higher carcinogenic activity of slightly
soluble Cr(VI) is consistent with cellular studies that indicate that
chromate dissociation in close proximity to the lung cell surface may
be a critical feature to efficient chromate ion uptake. This is
probably best achieved by Cr(VI) compounds that have intermediate water
solubility rather than by highly water-soluble Cr(VI) that rapidly
dissolves and diffuses in the aqueous fluid layers lining the
respiratory tract. The higher carcinogenicity of slightly soluble
Cr(VI) may contribute, along with elevated Cr(VI) workplace exposures,
to the greater lung cancer mortality in certain occupational cohorts
exposed to both slightly soluble and other forms of Cr(VI). The vastly
different study protocols employed in the few animal inhalation
bioassays do not allow a valid comparison of lung tumor incidence
between slightly soluble and highly soluble Cr(VI) compounds.
b. Summary of Cr(VI) Carcinogenicity
After carefully considering all the epidemiological, animal and
mechanistic evidence presented in the rulemaking record, OSHA regards
all Cr(VI) compounds as agents able to induce carcinogenesis through a
genotoxic mode of action. This position is consistent with findings of
IARC, EPA, and ACGIH that classified Cr(VI) compounds as known or
confirmed human carcinogens. Based on the above animal and experimental
evidence, OSHA believes that slightly soluble Cr(VI) compounds are
likely to exhibit a greater degree of carcinogenicity than highly water
soluble or water insoluble Cr(VI) when the same dose is delivered to
critical target cells in the respiratory tract of the exposed worker.
In its evaluation of different Cr(VI) compounds, ACGIH recommended
lower occupational exposure limits for the slightly soluble strontium
chromate (TLV of 0.5 [mu]g/m\3\) and calcium chromate (TLV of 1 [mu]g/
m\3\) than either water insoluble (TLV of 10 [mu]g/m\3\) or water
soluble (TLV of 50 [mu]g/m\3\) forms of Cr(VI) based on the animal
instillation studies cited above. While these animal instillation
studies are useful for hazard identification and qualitative
determinations of relative potency, they cannot be used to determine a
reliable quantitative estimate of risk for human workers breathing
these chromates during occupational exposure. This was due to use of
inadequate number of dose levels (e.g., single dose level) or a less
appropriate route of administration (e.g., tracheal instillation).
It is not clear from the animal or cellular studies whether the
carcinogenic potency of water insoluble Cr(VI) compounds would be
expected to be more or less than highly water soluble Cr(VI). However,
it was found that a greater percentage of water insoluble lead chromate
remains in the lungs of rats for longer periods than the highly water
soluble sodium chromate when instilled intratracheally at similar doses
(Ex. 35-56). Since water insoluble lead chromate can persist for long
periods in the lung and increase intracellular levels of Cr and damage
DNA in human lung cells at low doses (e.g., 0.1 [mu]g/cm\2\), OSHA
believes that based on the scientific evidence discussed above it is
reasonable to regard the water insoluble Cr(VI) to be of similar
carcinogenic potency to highly soluble Cr(VI) compounds. No convincing
scientific evidence was introduced into the record that shows lead
chromate to be less carcinogenic than highly soluble chromate
compounds.
C. Non-cancer Respiratory Effects
The following sections describe the evidence from the literature on
nasal irritation, nasal ulcerations, nasal perforations, asthma, and
bronchitis following inhalation exposure to water
soluble Cr(VI) compounds. The evidence clearly demonstrates that
workers can develop impairment to the respiratory system (nasal
irritation, nasal ulceration, nasal perforation, and asthma) after
workplace exposure to Cr(VI) compounds below the previous PEL.
It is very clear from the evidence that workers may develop nasal
irritation, nasal tissue ulcerations, and nasal septum perforations at
occupational exposures level at or below the current PEL of 52 [mu]g/
m\3\. However, it is not clear what occupational exposure levels lead
to the development of occupational asthma or bronchitis.
1. Nasal Irritation, Nasal Tissue Ulcerations and Nasal Septum
Perforations
Occupational exposure to Cr(VI) can lead to nasal tissue
ulcerations and nasal septum perforations. The nasal septum separates
the nostrils and is composed of a thin strip of cartilage. The nostril
tissue consists of an overlying mucous membrane known as the mucosa.
The initial lesion after Cr(VI) exposure is characterized by localized
inflammation or a reddening of the affected mucosa, which can later
lead to atrophy. This may progress to an ulceration of the mucosa layer
upon continued exposure (Ex. 35-1; Ex. 7-3). If exposure is
discontinued, the ulcer progression will stop and a scar may form. If
the tissue damage is sufficiently severe, it can result in a
perforation of the nasal septum, sometimes referred to chrome hole.
Individuals with nasal perforations may experience a range of signs and
symptoms, such as a whistling sound, bleeding, nasal discharge, and
infection. Some individuals may experience no noticeable effects.
Several cohort and cross-sectional studies have described nasal
lesions from airborne exposure to Cr(VI) at various electroplating and
chrome production facilities. Most of these studies have been reviewed
by the Center for Disease Control's Agency for Toxic Substances and
Disease Registry (ATSDR) toxicological profile for chromium (Ex. 35-
41). OSHA reviewed the studies summarized in the profile, conducted its
own literature search, and evaluated studies and comments submitted to
the rulemaking record. In its evaluation, OSHA took into consideration
the exposure regimen and experimental conditions under which the
studies were performed, including exposure levels, duration of
exposure, number of animals, and the inclusion of appropriate control
groups. Studies were not included if they did not contribute to the
weight of evidence either because of inadequate documentation or
because of poor quality. This section only covers some of the key
studies and reviews. OSHA has also identified two case reports
demonstrating the development of nasal irritation and nasal septum
perforations, and these case reports are summarized as well. One case
report shows how a worker can develop the nasal perforations from
direct contact (i.e., touching the inner surface of the nose with
contaminated fingers).
Lindberg and Hedenstierna examined the respiratory symptoms and
effects of 104 Swedish electroplaters (Ex. 9-126). Of the 104
electroplaters, 43 were exposed to chromic acid by inhalation. The
remaining 61 were exposed to a mixture of chromic acid and nitric acid,
hydrochloric acid, boric acid, nickel, and copper salts. The workers
were evaluated for respiratory symptoms, alterations in the condition
of the nasal tissue, and lung function. All workers were asked to fill
out a detailed questionnaire on their history of respiratory symptoms
and function. Physicians performed inspections of the nasal passages of
each worker. Workers were given a pulmonary function test to assess
lung function. For those 43 workers exposed exclusively to chromic
acid, the median exposure time was 2.5 years, ranging from 0.2 to 23.6
years. The workers were divided into two groups, a low exposure group
(19 workers exposed to eight-hour time weighted average levels below 2
[mu]g/m\3\) and a high exposure group (24 workers exposed to eight-hour
time weighted average levels above 2 [mu]g/m\3\). Personal air sampling
was conducted on 11 workers for an entire week at stations close to the
chrome baths to evaluate peak exposures and variations in exposure on
different days over the week. Nineteen office employees who were not
exposed to Cr(VI) were used as controls for nose and throat symptoms,
and 119 auto mechanics (no car painters or welders) whose lung function
had been evaluated using similar techniques to those used on Cr(VI)
exposed workers were used as controls for lung function.
The investigators reported nasal tissue ulcerations and septum
perforations in a group of workers exposed to chromic acid as Cr(VI) at
peak exposure ranging from 20 [mu]g/m\3\ to 46 [mu]g/m\3\. The
prevalence of ulceration/perforation was statistically higher than the
control group. Of the 14 individuals in the 20-46 [mu]g/m\3\ exposure
group, 7 developed nasal ulcerations. In addition to nasal ulcerations,
2 of the 7 also had nasal perforations. Three additional individuals in
this group developed nasal perforations in the absence of ulcerations.
None of the 14 workers in the 20-46 [mu]g/m\3\ exposure group were
reported to have nasal tissue atrophy in the absence of the more
serious ulceration or perforation.
At average exposure levels from 2 [mu]g/m\3\ to 20 [mu]g/m\3\, half
of the workers complained of "constantly running nose," "stuffy
nose," or "there was a lot to blow out." (Authors do not provide
details of each complaint). Nasal tissue atrophy, in the absence of
ulcerations or perforations, was observed in 66 percent of
occupationally exposed workers (8 of 12 subjects) at relatively low
peak levels ranging from 2.5 [mu]g/m\3\ to 11 [mu]g/m\3\. No one
exposed to levels below 1 [mu]g/m\3\ (time-weighted average, TWA)
complained of respiratory symptoms or developed lesions.
The authors also reported that in the exposed workers, both forced
vital capacity and forced expiratory volume in one second were reduced
by 0.2 L, when compared to controls. The forced mid-expiratory flow
diminished by 0.4 L/second from Monday morning to Thursday afternoon in
workers exposed to chromic acid as Cr(VI) at daily TWA average levels
of 2 [mu]g/m\3\ or higher. The effects were small, not outside the
normal range and transient. Workers recovered from the effects after
two days. There was no difference between the control and exposed group
after the weekend. The workers exposed to lower levels (2 [mu]g/m\3\ or
lower, TWA) showed no significant changes.
Kuo et al. evaluated nasal septum ulcerations and perforations in
189 electroplaters in 11 electroplating factories (three factories used
chromic acid, six factories used nickel-chromium, and two factories
used zinc) in Taiwan (Ex. 35-10). Of the 189 workers, 26 used Cr(VI),
129 used nickel-chromium, and 34 used zinc. The control group consisted
of electroplaters who used nickel and zinc. All workers were asked to
fill out a questionnaire and were given a nasal examination including a
lung function test by a certified otolaryngologist. The authors
determined that 30% of the workers (8/26) that used chromic acid
developed nasal septum perforations and ulcerations and 38% (10/26)
developed nasal septum ulcers. Using the Mantel Extension Test for
Trends, the authors also found that chromium electroplaters had an
increased likelihood of developing nasal ulcers and perforations
compared to electroplating workers using nickel-chromium and zinc.
Personal sampling of airborne Cr(VI) results indicated the highest
levels (32 [mu]g/m\3\ 35 [mu]g/m\3\, ranging from 0.1
[mu]g/m\3\-119 [mu]g/m\3\) near the electroplating tanks of the Cr(VI)
electroplating
factories (Ex. 35-11). Much lower personal sampling levels were
reported in the "other areas in the manufacturing area" and in the
"administrative area" (TWA 0.16 0.10 [mu]g/m\3\) of the
Cr(VI) electroplating plant. The duration of sampling was not
indicated. The lung function tests showed that Cr(VI) electroplaters
had significantly lower forced vital capacity and forced expiratory
volume when compared to other exposure groups.
Cohen et al. examined respiratory symptoms of 37 electroplaters
following inhalation exposure to chromic acid (Ex. 9-18). The mean
length of employment for the 37 electroplaters was 26.9 months (range
from 0.3 to 132 months). Fifteen workers employed in other parts of the
plant were randomly chosen for the control group (mean length of
employment was 26.1 months; range from 0.1 to 96). All workers were
asked to fill out a questionnaire on their respiratory history and to
provide details about their symptoms. An otolaryngologist then examined
each individual's nasal passages and identified ulcerations and
perforations. Air samples to measure Cr(VI) were collected for
electroplaters. The air sampling results of chromic acid as Cr(VI)
concentrations for electroplaters was a mean of 2.9 [mu]g/m\3\ (range
from non-detectable to 9.1 [mu]g/m\3\). The authors found that 95% of
the electroplaters developed pathologic changes in nasal mucosa.
Thirty-five of the 37 workers who were employed for more than 1 year
had nasal tissue damage. None of these workers reported any previous
job experience involving Cr(VI) exposure. Four workers developed nasal
perforations, 12 workers developed ulcerations and crusting of the
septal mucosa, 11 workers developed discoloration of the septal mucosa,
and eight workers developed shallow erosion of septal mucosa. The
control group consisted of 15 workers who were not exposed to Cr(VI) at
the plant. All but one had normal nasal mucosa. The one individual with
an abnormal finding was discovered to have had a previous Cr(VI)
exposure while working in a garment manufacturing operation as a fabric
dyer for three years. In addition to airborne exposure, the authors
observed employees frequently wiping their faces and picking their
noses with contaminated hands and fingers. Many did not wear any
protective gear, such as gloves, glasses, or coveralls.
Lucas and Kramkowsi conducted a Health Hazard Evaluation (HHE) on
11 chrome platers in an industrial electroplating facility (Ex. 3-84).
The electroplaters worked for about 7.5 years on average. Physicians
evaluated each worker for chrome hole scars, nasal septum ulceration,
mucosa infection, nasal redness, perforated nasal septum, and wheezing.
Seventeen air samples for Cr(VI) exposure were collected in the chrome
area. Cr(VI) air concentrations ranged from 1 to 20 [mu]g/m\3\, with an
average of 4 [mu]g/m\3\. In addition to airborne exposure, the authors
observed workers being exposed to Cr(VI) by direct "hand to nose"
contact, such as touching the nose with contaminated hands. Five
workers had nasal mucosa that became infected, two workers had nasal
septum ulcerations, two workers had atrophic scarring (author did not
provide explanation), possibly indicative of presence of past
ulcerations, and four workers had nasal septum perforations.
Gomes evaluated 303 employees from 81 electroplating operations in
Sao Paulo, Brazil (Ex. 9-31). Results showed that more than two-thirds
of the workers had nasal septum ulcerations and perforations following
exposure to chromic acid at levels greater than 100 [mu]g/m\3\, but
less than 600 [mu]g/m\3\ (precise duration of exposure was not stated).
These effects were observed within one year of employment.
Lin et al. examined nasal septum perforations and ulcerations in 79
electroplating workers from seven different chromium electroplating
factories in Taipei, Taiwan (Ex.35-13). Results showed six cases of
nasal septum perforations, four having scar formations, and 38 cases of
nasal septum ulcerations following inhalation exposure to chromic acid.
Air sampling near the electroplating tanks had the highest range of
chromic acid as Cr(VI) (mean of 28 [mu]g/m\3\; range from 0.7 to 168.3
[mu]g/m\3\). In addition to airborne exposures, the authors also
observed direct "hand to nose" contact where workers placed
contaminated fingers in their nose. The authors attributed the high
number of cases to poor industrial hygiene practices in the facilities.
Five of the seven factories did not have adequate ventilation systems
in place. Workers did not wear any PPE, including respirators.
Bloomfield and Blum evaluated nasal tissue damage and nasal septum
perforations in 23 workers employed at six chromium electroplating
plants (Ex. 9-13). They found that daily exposure to chromic acid as
Cr(VI) at levels of 52 [mu]g/m\3\ or higher can lead to nasal tissue
damage. Three workers developed nasal ulcerations, two workers had
nasal perforations, nine workers had nose bleeds, and nine workers had
inflamed mucosa.
Kleinfeld and Rosso found that seven out of nine of chrome
electroplaters had nasal septum ulcerations (Ex. 9-41). The nine
workers were exposed to chromic acid as Cr(VI) by inhalation at levels
ranging from 93 [mu]g/m\3\ to 728 [mu]g/m\3\. Duration of exposure
varied from two weeks to one year. Nasal septum ulcerations were noted
in some workers who had been employed for only one month.
Royle, using questionnaire responses from 997 British
electroplaters exposed to chromic acid, reported a significant increase
in the prevalence of nasal ulcerations. The prevalence increased the
longer the worker was exposed to chromic acid (e.g., from 14 cases with
exposure less than one year to 62 cases with exposure over five years)
(Ex. 7-50). In all but 2 cases, air samples revealed chromic acid
concentrations of 0.03 mg/m\3\ (i.e., 30 [mu]g/m\3\).
Gibb et al. reported nasal irritations, nasal septum bleeding,
nasal septum ulcerations and perforations among a cohort of 2,350
chrome production workers in a Baltimore plant (Ex. 31-22-12). A
description of the cohort is provided in detail in the cancer health
effects section V.B. of this preamble. The authors found that more than
60% of the cohort had experienced nasal ulcerations and irritations,
and that the workers developed these effects for the first time within
the first three months of being hired (median). Gibb et al. found that
the median annual exposure to Cr(VI) during first diagnosis of
irritated and/or ulcerated nasal septum was 10 [mu]g/m\3\. About 17% of
the cohort reported nasal perforations. Based on historical data, the
authors believe that the nasal findings are attributable to Cr(VI)
exposure.
Gibb et al. also used a Proportional Hazard Model to evaluate the
relationship between Cr(VI) exposure and the first occurrence of each
of the clinical findings. Cr(VI) data was entered into the model as a
time dependent variable. Other explanatory variables were calendar year
of hire and age of hire. Results of the model indicated that airborne
Cr(VI) exposure was associated with the occurrence of nasal septum
ulceration (p = 0.0001). The lack of an association between airborne
Cr(VI) exposure and nasal perforation and bleeding nasal septum may
reflect the fact that Cr(VI) concentrations used in the model represent
annual averages for the job, in which the worker was involved in at the
time of the findings, rather than a short-term average. Annual averages
do not factor in day-to-day fluctuations or extreme episodic
occurrences. Also, the author believed that poor housekeeping
and hygiene practices may have contributed to these health effects as
well as Cr(VI) air borne concentrations.
Based on their hazard model, Gibb et al. estimated the relative
risks for nasal septum ulcerations would increase 1.2 for each 52 [mu]g
of Cr(VI)/m\3\ increase in Cr(VI) air levels. They found a reduction in
the incidence of nasal findings in the later years. They found workers
from the earlier years who did not wear any PPE had a greater risk of
developing respiratory problems. They believe that the reduction in
ulcerations was possibly due to an increased use of respirators and
protective clothing and improved industrial hygiene practices at the
facility.
The U.S. Public Health Service conducted a study of 897 chrome
production workers in seven chromate producing plants in the early
1950s (Ex. 7-3). The findings of this study were used in part as
justification for the current OSHA PEL. Workers were exposed by
inhalation to various water soluble chromates and bichromate compounds.
The total mean exposure to the workers was a TWA of 68 [mu]g/m\3\. Of
the 897 workers, 57% (or 509 workers) were found to have nasal septum
perforations. Nasal septum perforations were even observed in workers
during their first year on the job.
Case reports provide further evidence that airborne exposure and
direct "hand to nose" contact of Cr(VI) compounds lead to the
development of nasal irritation and nasal septum perforations.
For example, a 70-year-old man developed nasal irritation,
incrustation, and perforation after continuous daily exposure by
inhalation to chromium trioxide (doses were not specified, but most
likely quite high given the nature of his duties). This individual
inhaled chromium trioxide daily by placing his face directly over an
electroplating vessel. He worked in this capacity from 1934 to 1982.
His symptoms continued to worsen after he stopped working. By 1991, he
developed large perforations of the nasal septum and stenosis (or
constriction) of both nostrils by incrustation (Ex. 35-8).
Similarly, a 30-year-old female jigger (a worker who prepares the
items prior to electroplating by attaching the items to be plated onto
jigs or frames) developed nasal perforation in her septum following
continuous exposure (doses in this case were not provided) to chromic
acid mists. She worked adjacent to the automated Cr(VI) electroplating
shop. She was also exposed to chromic acid from direct contact when she
placed her contaminated fingers in her nose. Her hands became
contaminated by handling wet components in the jigging and de-jigging
processes (Ex. 35-24).
Evidence of nasal septum perforations has also been demonstrated in
experimental animals. Adachi exposed 23 C57BL mice to chromic acid by
inhalation at concentrations of 1.81 mg Cr(VI)/m\3\ for 120 min per
day, twice a week and 3.63 mg Cr(VI)/m\3\ for 30 minutes per day, two
days per week for up to 12 months (Ex. 35-26). Three of the 23 mice
developed nasal septum perforations in the 12 month exposure group.
Adachi et al. also exposed 50 ICR female mice to chromic acid by
inhalation at concentrations of 3.18 mg Cr(VI)/m\3\ for 30 minutes per
day, two days per week for 18 months (Ex. 35-26-1). The authors used a
miniaturized chromium electroplating system to mimic electroplating
processes and exposures similar to working experience. Nasal septum
perforations were found in six mice that were sacrificed after 10
months of exposure. Of those mice that were sacrificed after 18 months
of exposure, nasal septum perforations were found in three mice.
2. Occupational Asthma
Occupational asthma is considered "a disease characterized by
variable airflow limitation and/or airway hyperresponsiveness due to
causes and conditions attributable to a particular occupational
environment and not to stimuli encountered outside the workplace" (Ex.
35-15). Asthma is a serious illness that can damage the lungs and in
some cases be life threatening. The common symptoms associated with
asthma include heavy coughing while exercising or when resting after
exercising, shortness of breath, wheezing sound, and tightness of chest
(Exs. 35-3; 35-6).
Cr(VI) is considered to be an airway sensitizer. Airway sensitizers
cause asthma through an immune response. The sensitizing agent
initially causes production of specific antibodies that attach to cells
in the airways. Subsequent exposure to the sensitizing agent, such as
Cr(VI), can trigger an immune-mediated narrowing of the airways and
onset of bronchial inflammation. All exposed workers do not become
sensitized to Cr(VI) and the asthma only occurs in sensitized
individuals. It is not clear what occupational exposure levels of
Cr(VI) compounds lead to airway sensitization or the development of
occupational asthma.
The strongest evidence of occupational asthma has been demonstrated
in four case reports. OSHA chose to focus on these four case reports
because the data from other occupational studies do not exclusively
implicate Cr(VI). The four case reports have the following in common:
(1) The worker has a history of occupational exposure exclusively to
Cr(VI); (2) a physician has confirmed a diagnosis that the worker has
symptoms consistent with occupational asthma; and (3) the worker
exhibits functional signs of air restriction (e.g., low forced
expiratory volume in one second or low peak expiratory flow rate) upon
bronchial challenge with Cr(VI) compounds. These case reports
demonstrate, through challenge tests, that exposure to Cr(VI) compounds
can cause asthmatic responses. The other general case reports below did
not use challenge tests to confirm that Cr(VI) was responsible for the
asthma; however, these reports came from workers similarly exposed to
Cr(VI) such that Cr(VI) is likely to have been a contributing factor in
the development of their asthmatic symptoms.
DaReave reported the case of a 48-year-old cement floorer who
developed asthma from inhaling airborne Cr(VI) (Ex. 35-7). This worker
had been exposed to Cr(VI) as a result of performing cement flooring
activities for more than 20 years. The worker complained of dyspnea,
shortness of breath, and wheezing after work, especially after working
in enclosed spaces. The Cr(VI) content in the cement was about 12 ppm.
A bronchial challenge test with potassium dichromate produced a 50%
decrease in forced expiratory volume in one second. The occupational
physician concluded that the worker's asthmatic condition, triggered by
exposure to Cr(VI) caused the worker to develop bronchial constriction.
LeRoyer reported a case of a 28-year-old roofer who developed
asthma from breathing dust while sawing material made of corrugated
fiber cement containing Cr(VI) for nine years (Ex. 35-12). This worker
demonstrated symptoms such as wheezing, shortness of breath, coughing,
rhinitis, and headaches while working. Skin prick tests were all
negative. Several inhalation challenges were performed by physicians
and immediate asthmatic reactions were observed after nebulization of
potassium dichromate. A reduction (by 20%) in the forced expiratory
volume in one second after exposure to fiber cement dust was noted.
Novey et al. reported a case of a 32-year-old electroplating worker
who developed asthma from working with chromium sulfate and nickel
salts (Ex. 35-16). He began experiencing coughs,
wheezing, and dyspnea within the first week of exposure. Separate
inhalation challenge tests given by physicians using chromium sulfate
and nickel salts resulted in positive reactions. The worker immediately
had difficulty breathing and started wheezing. The challenges caused
the forced expiratory volume in 1 second to decrease by 22% and the
forced expiratory volume in 1 second/forced vital capacity ratio to
decrease from 74.5% to 60.4%. The author believes the worker's
bronchial asthma was induced from inhaling chromium sulfate and nickel
salts. Similar findings were reported in a different individual by
Sastre (Ex.35-20).
Shirakawa and Morimoto reported a case of a 50-year-old worker who
developed asthma while working at a metal-electroplating plant (Ex. 35-
21). Bronchial challenge by physicians produced positive results when
using potassium bichromate, followed by a rapid recovery within 5
minutes, when given no exposures. The worker's forced expiratory volume
in one second dropped by 37% after inhalation of potassium bichromate.
The individual immediately began wheezing, coughing with dyspnea, and
recovered without treatment within five minutes. The author believes
that the worker developed his asthma from inhaling potassium
bichromate.
In addition to the case reports confirming that Cr(VI) is
responsible for the development of asthma using inhalation challenge
tests, there are several other case reports of Cr(VI) exposed workers
having symptoms consistent with asthma where the symptoms were never
confirmed by using inhalation challenge tests.
Lockman reported a case of a 41-year-old woman who was
occupationally exposed to potassium dichromate during leather tanning
(Ex. 35-14). The worker developed an occupational allergy to potassium
dichromate. This allergy involved both contact dermatitis and asthma.
The physicians considered other challenge tests using potassium
dichromate as the test agent (i.e., peak expiratory flow rate, forced
expiratory volume in 1 second and methacholine or bronchodilator
challenge), but the subject changed jobs before the physicians could
administer these tests. Once the subject changed jobs, all her symptoms
disappeared. It was not confirmed whether the occupational exposure to
Cr(VI) was the cause of the asthma.
Williams reported a 23-year-old textile worker who was
occupationally exposed to chromic acid. He worked near two tanks of
chromic acid solutions (Ex. 35-23) and inhaled fumes while frequently
walking through the room with the tanks. He developed both contact
dermatitis and asthma. He believes the tank was poorly ventilated and
was the source of the fumes. He stopped working at the textile firm on
the advice of his physician. After leaving, his symptoms improved
greatly. No inhalation bronchial challenge testing was conducted to
confirm that chromic acid was causing his asthmatic attacks. However,
as noted above, chromic acid exposure has been shown to lead to
occupational asthma, and thus, chromic acid was likely to be a
causative agent in the development of asthma.
Park et al. reported a case of four workers who worked in various
occupations involving exposure to either chromium sulfate or potassium
dichromate (Ex. 35-18). Two worked in a metal electroplating factory,
one worked at a cement manufacturer, and the other worked in
construction. All four developed asthma. One individual had a positive
response to a bronchial provocation test (with chromium sulfate as the
test agent). This individual developed an immediate reaction,
consisting of wheezing, coughing and dyspnea, upon being given chromium
sulfate as the test agent. Peak expiratory flow rate decreased by about
20%. His physician determined that exposure to chromium sulfate was
contributing to his asthma condition. Two other individuals had
positive reactions to prick skin tests with chromium sulfate as the
test agent. Two had positive responses to patch tests using potassium
dichromate as the testing challenge agent. Only one out of four
underwent inhalation bronchial challenge testing (with a positive
result to chromium sulfate) in this report.
3. Bronchitis
In addition to nasal ulcerations, nasal septum perforations, and
asthma, there is also limited evidence from reports in the literature
of bronchitis associated with Cr(VI) exposure. It is not clear what
occupational exposure levels of Cr(VI) compounds would lead to the
development of bronchitis.
Royle found that 28% (104/288) of British electroplaters developed
bronchitis upon inhalation exposure to chromic acid, as compared to 23%
(90/299) controls (Ex. 7-50). The workers were considered to have
bronchitis if they had symptoms of persistent coughing and phlegm
production. In all but two cases of bronchitis, air samples revealed
chromic acid at levels of 0.03 mg/m\3\. Workers were asked to fill out
questionnaires to assess respiratory problems. Self-reporting poses a
problem in that the symptoms and respiratory health problems identified
were not medically confirmed by physicians. Workers in this study
believe they were developing bronchitis, but it is not clear from this
study whether the development of bronchitis was confirmed by
physicians. It is also difficult to assess the bronchitis health
effects of chromic acid from this study because the study results for
the exposed (28%) and control groups (23%) were similar.
Alderson et al. reported 39 deaths of chromate production workers
related to chronic bronchitis from three chromate producing factories
(Bolton, Eaglescliffe, and Rutherglen) from 1947 to 1977 (Ex. 35-2).
Neither the specific Cr(VI) compound nor the extent or frequency with
which the workers were exposed were specified. However, workers at all
three factories were exposed to sodium chromate, chromic acid, and
calcium chromate at one time or another. The authors did not find an
excess number of bronchitis related deaths at the Bolton and
Eaglescliffe factories. At Rutherglen, there was an excess number of
deaths (31) from chronic bronchitis with a ratio of observed/expected
of 1.8 (p< 0.001). It is difficult to assess the respiratory health
effects of Cr(VI) compounds from this study because there are no
exposure data, there are no data on smoking habits, nor is it clear the
extent, duration, and amount of specific Cr(VI) compound to which the
workers were exposed during the study.
While the evidence supports an association between bronchitis and
Cr(VI) exposure is limited, studies in experimental animals demonstrate
that Cr(VI) compounds can cause lung irritation, inflammation in the
lungs, and possibly lung fibrosis at various exposure levels. Glaser et
al. examined the effects of inhalation exposure of chromium (VI) on
lung inflammation and alveolar macrophage function in rats (Ex. 31-18-
9). Twenty, 5-week-old male TNO-W-74 Wistar rats were exposed via
inhalation to 25-200 [mu]g Cr(VI)/m3 as sodium dichromate
for 28 days or 90 days for 22 hours per day, 7 days per week in
inhalation chambers. Twenty, 5-week-old male TNO-W-74 Wistar rats also
served as controls. All rats were killed at the end of the inhalation
exposure period. The authors found increased lung weight in the 50-200
[mu]g/m3 groups after the 90-day exposure period. They also
found that 28-day exposure to levels of 25 and 50 [mu]g/m3
resulted in "activated" alveolar macrophages with stimulated
phagocytic activities. A more pronounced effect on the activation of
alveolar macrophages was seen during the 90-day exposure period of 25
and 50 [mu]g/m3.
Glaser et al. exposed 150 male, 8-week-old Wistar rats (10 rats per
group) continuously by inhalation to aerosols of sodium dichromate at
concentrations of 50, 100, 200, and 400 [mu]g Cr(VI)/m3 for
22 hours per day, 7 days a week, for continuous exposure for 30 days or
90 days in inhalation chambers (Ex. 31-18-11). Increased lung weight
changes were noticeable even at levels as low as 50 and 100 [mu]g
Cr(VI)/m3 following both 30 day and 90 day exposures.
Significant accumulation of alveolar macrophages in the lungs was noted
in all of the exposure groups. Lung fibrosis occurred in eight rats
exposed to 100 [mu]g Cr(VI)/m3 or above for 30 days. Most
lung fibrosis disappeared after the exposure period had ceased. At 50
[mu]g Cr(VI)/m3 or higher for 30 days, a high incidence of
hyperplasia was noted in the lung and respiratory tract. The total
protein in bronchoalveolar lavage (BAL) fluid, albumin in BAL fluid,
and lactate dehydrogenase in BAL fluid were significant at elevated
levels of 200 and 400 [mu]g Cr(VI)/m3 in both the 30 day and
90 day exposure groups (as compared to the control group). These
responses are indicative of severe injury in the lungs of animals
exposed to Cr(VI) dose levels of 200 [mu]g Cr(VI)/m3 and
above. At levels of 50 and 100 [mu]g Cr(VI)/m3, the
responses are indicative of mild inflammation in the lungs. The authors
concluded that these results suggest that the severe inflammatory
reaction may lead to more chronic and obstructive lesions in the lung.
4. Summary
Overall, there is convincing evidence to indicate that Cr(VI)
exposed workers can develop nasal irritation, nasal ulcerations, nasal
perforations, and asthma. There is also some limited evidence that
bronchitis may occur when workers are exposed to Cr(VI) compounds at
high levels. Most of the studies involved exposure to water-soluble
Cr(VI) compounds. It is very clear that workers may develop nasal
irritations, nasal ulcerations, and nasal perforations at levels below
the current PEL of 52 [mu]g/m3. However, it is not clear
what occupational exposure levels lead to disorders like asthma and
bronchitis.
There are numerous studies in the literature showing nasal
irritations, nasal perforations, and nasal ulcerations resulting from
Cr(VI) inhalation exposure. It also appears that direct hand-to-nose
contact (i.e., by touching inner nasal surfaces with contaminated
fingers) can contribute to the incidence of nasal damage. Additionally,
some studies show that workers developed these nasal health problems
because they did not wear any PPE, including respiratory protection.
Inadequate area ventilation and sanitation conditions (lack of
cleaning, dusty environment) probably contributed to the adverse nasal
effects.
There are several well documented case reports in the literature
describing occupational asthma specifically triggered by Cr(VI) in
sensitized workers. All involved workers who frequently suffered
symptoms typical of asthma (e.g. dyspnea, wheezing, coughing, etc.)
while working in jobs involving airborne exposure to Cr(VI). In some of
the reports, a physician diagnosed bronchial asthma triggered by Cr(VI)
after specific bronchial challenge with a Cr(VI) aerosol produced
characteristic symptoms and asthmatic airway responses. Several
national and international bodies, such as the National Institute for
Occupational Safety and Health, the World Health Organization's
International Programme on Chemical Safety, and the United Kingdom
Health and Safety Executive have recognized Cr(VI) as an airway
sensitizer that can cause occupational asthma. Despite the widespread
recognition of Cr(VI) as an airway sensitizer, OSHA is not aware of any
well controlled occupational survey or epidemiological study that has
found a significantly elevated prevalence of asthma among Cr(VI)-
exposed workers. The level of Cr(VI) in the workplace that triggers the
asthmatic condition and the number of workers at risk are not known.
The evidence that workers breathing Cr(VI) can develop respiratory
disease that involve inflammation, such as asthma and bronchitis is
supported by experimental animal studies. The 1985 and 1990 Glaser et
al. studies show that animals experience irritation and inflammation of
the lungs following repeated exposure by inhalation to water-soluble
Cr(VI) at air concentrations near the previous PEL of 52 [mu]g/
m3.
D. Dermal Effects
Occupational exposure to Cr(VI) is a well-established cause of
adverse health effects of the skin. The effects are the result of two
distinct processes: (1) Irritant reactions, such as skin ulcers and
irritant contact dermatitis, and (2) delayed hypersensitivity
(allergic) reactions. Some evidence also indicates that exposure to
Cr(VI) compounds may cause conjunctivitis.
The mildest skin reactions consist of erythema (redness), edema
(swelling), papules (raised spots), vesicles (liquid spots), and
scaling (Ex. 35-313, p. 295). The lesions are typically found on
exposed areas of the skin, usually the hands and forearms (Exs. 9-9; 9-
25). These features are common to both irritant and allergic contact
dermatitis, and it is generally not possible to determine the etiology
of the condition based on histopathologic findings (Ex. 35-314).
Allergic contact dermatitis can be diagnosed by other methods, such as
patch testing (Ex. 35-321, p. 226). Patch testing involves the
application of a suspected allergen to the skin, diluted in petrolatum
or some other vehicle. The patch is removed after 48 hours and the skin
examined at the site of application to determine if a reaction has
occurred.
Cr(VI) compounds can also have a corrosive, necrotizing effect on
living tissue, forming ulcers, or "chrome holes" (Ex. 35-315). This
effect is apparently due to the oxidizing properties of Cr(VI)
compounds (Ex. 35-318, p. 623). Like dermatitis, chrome ulcers
generally occur on exposed areas of the body, chiefly on the hands and
forearms (Ex. 35-316). The lesions are initially painless, and are
often ignored until the surface ulcerates with a crust which, if
removed, leaves a crater two to five millimeters in diameter with a
thickened, hardened border. The ulcers can penetrate deeply into tissue
and become painful. Chrome ulcers may penetrate joints and cartilage
(Ex. 35-317, p. 138). The lesions usually heal in several weeks if
exposure to Cr(VI) ceases, leaving a flat, atrophic scar (Ex. 35-318,
p.623). If exposure continues, chrome ulcers may persist for months
(Ex. 7-3).
It is generally believed that chrome ulcers do not occur on intact
skin (Exs. 35-317, p. 138; 35-315; 35-25). Rather, they develop readily
at the site of small cuts, abrasions, insect bites, or other injuries
(Exs. 35-315; 35-318, p. 138). In experimental work on guinea pigs,
Samitz and Epstein found that lesions were never produced on undamaged
skin (Ex. 35-315). The degree of trauma, as well as the frequency and
concentration of Cr(VI) application, was found to influence the
severity of chrome ulcers.
The development of chrome ulcers does not appear to be related to
the sensitizing properties of Cr(VI). Edmundson provided patch tests to
determine sensitivity to Cr(VI) in 56 workers who exhibited either
chrome ulcers or scars (Ex. 9-23). A positive response to the patch
test was found in only two of the workers examined.
Parkhurst first identified Cr(VI) as a cause of allergic contact
dermatitis in 1925 (Ex. 9-55). Cr(VI) has since been
confirmed as a potent allergen. Kligman (1966) used a maximization test
(a skin test for screening possible contact allergens) to assess the
skin sensitizing potential of Cr(VI) compounds (Ex. 35-327). Each of
the 23 subjects was sensitized to potassium dichromate. On a scale of
one to five, with five being the most potent allergen, Cr(VI) was
graded as five (i.e., an extreme sensitizer). This finding was
supported by a guinea pig maximization test, which assigned a grade of
four to potassium chromate using the same scale (Ex. 35-328).
1. Prevalence of Dermal Effects
Adverse skin effects from Cr(VI) exposure have been known since at
least 1827, when Cumin described ulcers in two dyers and a chromate
production worker (Ex. 35-317, p. 138). Since then, skin conditions
resulting from Cr(VI) exposure have been noted in a wide range of
occupations. Work with cement is regarded as the most common cause of
Cr(VI)-induced dermatitis (Exs. 35-313, p. 295; 35-319; 35-320). Other
types of work where Cr(VI)-related skin effects have been reported
include chromate production, chrome plating, leather tanning, welding,
motor vehicle assembly, manufacture of televisions and appliances,
servicing of railroad locomotives, aircraft production, and printing
(Exs. 31-22-12; 7-50; 9-31; 9-100; 9-63; 9-28; 9-95; 9-54; 35-329; 9-
97; 9-78; 9-9; 35-330). Some of the important studies on Cr(VI)-related
dermal effects in workers are described below.
a. Cement Dermatitis
Many workers develop cement dermatitis, including masons, tile
setters, and cement workers (Ex. 35-318, p. 624). Cement, the basic
ingredient of concrete, may contain several possible sources of
chromium (Exs. 35-317, p.148; 9-17). Clay, gypsum, and chalk that serve
as ingredients may contain traces of chromium. Ingredients may be
crushed using chrome steel grinders that, with wear, contribute to the
chromium content of the concrete. Refractory bricks in the kiln and ash
residues from the burning of coal or oil to heat the kiln serve as
additional sources. Trivalent chromium from these sources can be
converted to Cr(VI) in the kiln (Ex. 35-317. p. 148).
The prevalence of cement dermatitis in groups of workers with
regular contact with wet cement has been reported to be from 8 to 45
percent depending on the countries of origin, type of construction
industry, and criteria used to diagnose dermatitis (Exs. 46-74, 9-131;
35-317, 9-57, 40-10-10). Cement dermatitis can be caused by direct
irritation of the skin, by sensitization to Cr(VI), or both (Ex. 35-
317, p. 147). The reported proportion of allergic and irritant contact
dermatitis varies considerably depending on the information source. In
a review of 16 different data sets, Burrows (1983) found that, on
average, 80% of cement dermatitis cases were sensitized to Cr(VI) (Ex.
35-317, p. 148). The studies were mostly conducted prior to 1970 on
European construction workers. More recent occupational studies suggest
that Cr(VI) allergy may make up a smaller proportion of all dermatitis
in construction workers, depending on the Cr(VI) content of the cement.
For example, examination of 1238 German and Austrian construction
workers in dermatitis units found about half those with occupational
dermatitis were skin sensitized to Cr(VI) (Ex. 40-10-10). Several other
epidemiological investigations conducted in the 1980s and 1990s also
reported that allergic contact dermatitis made up 50 percent or less of
all dermatitis cases in various groups of construction workers exposed
to wet cement (Ex. 46-74).
Cement is alkaline, abrasive, and hydroscopic (water-absorbing),
and it is likely that the irritant effect resulting from these
properties interferes with the skin's defenses, permitting penetration
and sensitization to take place more readily (Ex. 35-318, p. 624). Dry
cement is considered relatively innocuous because it is not as alkaline
as wet cement (Exs. 35-317, p. 147; 9-17). When water is mixed with
cement the water liberates calcium hydroxide, causing a rise in pH (Ex.
35-317, p. 147).
Flyvholm et al. (1996) noted a correlation between the Cr(VI)
concentration in the local cement and the frequency of allergic contact
dermatitis (Ex. 35-326, p. 278). Because the Cr(VI) content depends
partially upon the chromium concentration in raw materials, there is a
great variability in the Cr(VI) content in cement from different
geographical regions. In locations with low Cr(VI) content, the
prevalence of Cr(VI)-induced allergic contact dermatitis was reported
to be approximately one percent, while in regions with higher chromate
concentrations the prevalence was reported to rise to between 9 to 11%
of those exposed (Ex. 35-326, p. 278). For example, only one of 35 U.S.
construction workers with confirmed cement dermatitis was reported to
have a positive Cr(VI) patch test in a 1970 NIOSH study (Ex. 9-57).
However, the same study revealed a low Cr(VI) content in 42
representative cement samples from U.S. companies (e.g 80 percent of
the samples with C(VI) < 2 [mu]g/g).
The relationship between Cr(VI) content in cement and the
prevalence of Cr(VI)-induced allergic contact dermatitis is supported
by the findings of Avnstorp (1989) in a study of Danish workers who had
daily contact with wet cement during the manufacture of pre-fabricated
concrete products (Ex. 9-131). Beginning in September of 1981, low
concentrations of ferrous sulfate were added to all cement sold in
Denmark to reduce Cr(VI) to trivalent chromium. Two hundred and twenty
seven workers were examined in 1987 for Cr(VI)-related skin effects.
The findings from these examinations were compared to the results from
190 workers in the same plants who were examined in 1981. The
prevalence of hand eczema had declined from 11.7% to 4.4%, and the
prevalence of Cr(VI) sensitization had declined from 10.5% to 2.6%.
While the two-to four-fold drop in prevalence was statistically
significant, the magnitude of the reduction may be overstated because
the amount of exposure time was less in the 1987 than the 1981 group.
There is also the possibility that other factors, in addition to
ferrous sulfate, may have led to less dermal contact to Cr(VI), such as
greater automation or less construction work. However, the study found
no significant change in the frequency of irritant dermatitis.
Another study also found lower prevalence of allergic contact
dermatitis among Finish construction workers following the 1987
decision to reduce Cr(VI) content of cement used in Finland to less
than 2 ppm (Ex. 48-8). Ferrous sulfate was typically added to the
cement to meet this requirement. There was a significantly decreased
risk of allergic Cr(VI) contact dermatitis reported to the Finnish
Occupational Disease Registry post-1987 as compared to pre-1987
(OR=0.4, 95% CI: 0.2-0.7) indicating the occurrence of disease dropped
one-third after use of the low Cr(VI) content cement. On the other
hand, the occurrence of irritant dermatitis remained stable throughout
the study period. Time of exposure was not a significant explanatory
variable in the analysis. However, the findings may have been somewhat
confounded by changes in diagnostic procedure over time. The Finnish
study retested patients previously diagnosed with prior patch test
protocols and found several false positives (i.e. false diagnosis of
Cr(VI) allergy).
In 2003, the Norwegian National Institute of Occupational Health
sponsored an expert peer review of 24
key epidemiological investigations addressing; (1) whether exposure to
wet cement containing water soluble Cr(VI) caused allergic contact
dermatitis, and (2) whether there was a causal association between
reduction of Cr(VI) in cement and reduction in the prevalence of the
disease (Ex. 46-74). The panel of four experts concluded that, despite
the documented limitations of each individual study, the collective
evidence was consistent in supporting "fairly strong associations
between Cr(VI) content in cement and the occurrence of allergic
dermatitis * * * it seems unlikely that all these associations reported
in the reviewed papers are due to systematic errors only" (Ex. 46-74,
p. 42).
Even though the Norwegian panel felt that the available evidence
indicated a relationship between reduced Cr(VI) content of wet cement
and lower occurrence of allergic dermatitis, they stated that the
epidemiological literature was "not sufficient to conclude that there
is a causal association" (Ex. 46-74, p. 42). This is somewhat
different than the view expressed in a written June 2002 opinion by the
Scientific Committee on Toxicity, Ecotoxicity and the Environment
(CSTEE) to the European Commission, Directorate for General Health and
Consumer Protection (Ex. 40-10-7). In responding to the question of
whether it is scientifically justified to conclude that cement
containing less than 2 ppm Cr(VI) content could substantially reduce
the risk of skin sensitization, the CSTEE stated that "the available
information clearly demonstrates that reduction of chromium VI in
cement to less than 2 ppm * * * will reduce the prevalence of allergic
contact eczema in workers" (Ex. 40-10-7, p. 5)
b. Dermatitis Associated With Cr(VI) From Sources Other Than Cement
In 1953 the U.S. Public Health Service reported on hazards
associated with the chromium-producing industry in the United States
(Ex. 7-3). Workers were examined for skin effects from Cr(VI) exposure.
Workers' eyes were also examined for possible effects from splashes of
Cr(VI)-containing compounds that had been observed in the plants. Of
the 897 workers examined, 451 had skin ulcers or scars of ulcers.
Seventeen workers were reported to have skin lesions suggestive of
chrome dermatitis. The authors noted that most plants provided adequate
washing facilities, and had facilities for providing clean work
clothes. A statistically significant increase in congestion of the
conjunctiva was also reported in Cr(VI)-exposed workers when compared
with non-exposed workers (38.7% vs. 25.8%).
In the Baltimore, Maryland chromate production plant examined by
Gibb et al. (2000), a substantial number of workers were reported to
have experienced adverse skin effects (Ex. 31-22-12). The authors
identified a cohort of 2,357 workers first employed at the plant
between 1950 and 1974. Clinic and first aid records were examined to
identify findings of skin conditions. These clinical findings were
identified by a physician as a result of routine examinations or visits
to the medical clinic by members of the cohort. Percentages of the
cohort with various clinical findings were as follows:
Irritated skin: 15.1%
Dermatitis: 18.5%
Ulcerated skin: 31.6%
Conjunctivitis: 20.0%
A number of factors make these results difficult to interpret. The
reported findings are not specifically related to Cr(VI) exposure. They
may have been the result of other workplace exposures, or non-workplace
factors. The report also indicates the percentage of workers who were
diagnosed with a condition during their tenure at the plant; however,
no information is presented to indicate the expected incidence of these
conditions in a population that is not exposed to Cr(VI).
Measurements of Cr(VI) air concentrations by job title were used to
estimate worker exposures. Based on these estimates, the authors used a
proportional hazards model to find a statistically significant
correlation (p=0.004) between ulcerated skin and airborne Cr(VI)
exposure. Statistically significant correlations between year of hire
and findings of ulcerated skin and dermatitis were also reported.
Exposures to Cr(VI) in the plant had generally dropped over time.
Median exposure to Cr(VI) at the time of occurrence for most of the
findings was said to be about 10 [mu]g/m\3\ Cr(VI) (reported as 20
[mu]g/m\3\ CrO3). It is unclear, however, what contribution
airborne Cr(VI) exposures may have had to dermal effects. Direct dermal
contact with Cr(VI) compounds in the plant may have been a contributing
factor in the development of these conditions.
Mean and median times on the job prior to initial diagnosis were
also reported. The mean time prior to diagnosis of skin or eye effects
ranged from 373 days for ulcerated skin to 719 days for irritated skin.
Median times ranged from 110 days for ulcerated skin to 221 days for
conjunctivitis. These times are notable because many workers in the
plant stayed for only a short time. Over 40% worked for less than 90
days. Because these short-term workers did not remain in the workplace
for the length of time that was typically necessary for these effects
to occur, the results of this study may underestimate the incidence
that would occur with a more stable worker population.
Lee and Goh (1988) examined the skin condition of 37 workers who
maintained chrome plating baths and compared these workers with a group
of 37 control subjects who worked in the same factories but were not
exposed to Cr(VI) (Ex. 35-316). Mean duration of employment as a chrome
plater was 8.1 (SD7.9) years. Fourteen (38%) of the chrome
platers had some occupational skin condition; seven had chrome ulcers,
six had contact dermatitis and one had both. A further 16 (43%) of the
platers had scars suggestive of previous chrome ulcers. Among the
control group, no members had ulcers or scars of ulcers, and three had
dermatitis.
Where ulcers or dermatitis were noted, patch tests were
administered to determine sensitization to Cr(VI) and nickel. Of the
seven workers with chrome ulcers, one was allergic to Cr(VI). Of the
six workers with dermatitis, two were allergic to Cr(VI) and one to
nickel. The worker with ulceration and dermatitis was not sensitized to
either Cr(VI) or nickel. Although limited by a relatively small study
population, this report clearly indicates that Cr(VI)-exposed workers
face an increased risk of adverse skin effects. The fact that the
majority of workers with dermatitis were not sensitized to Cr(VI)
indicates that irritant factors play an important role in the
development of dermatitis in chrome plating operations.
Royle (1975) also investigated the occurrence of skin conditions
among workers involved in chrome plating (Ex. 7-50). A questionnaire
survey completed by 997 chrome platers revealed that 21.8% had
experienced skin ulcers, and 24.6% had suffered from dermatitis. No
information was presented to indicate the expected incidence in a
comparable population that was not exposed to Cr(VI). Of the 54 plants
involved in the study, 49 used nickel, another recognized cause of
allergic contact dermatitis.
The author examined the relationship between the incidence of these
conditions and length of exposure. The plater population was divided
into three groups: those with less than one year of Cr(VI) exposure,
those with one to five years of Cr(VI) exposure, and those with over
five years of Cr(VI) exposure. A statistically significant trend was
found between length of Cr(VI) exposure and incidence of skin ulcers. The
incidence of dermatitis, on the other hand, bore no relationship to
length of exposure.
In 1973, researchers from NIOSH reported on the results of a health
hazard investigation of a chrome plating establishment (Ex. 3-5). In
the plating area, airborne Cr(VI) concentrations ranged from less than
0.71 to 9.12 [mu]g/m\3\ (mean 3.24 [mu]g/m\3\; SD=2.48 [mu]g/m\3\). Of
the 37 exposed workers who received medical examinations, five were
reported to have chrome-induced lesions on their hands. Hygiene and
housekeeping practices in this facility were reportedly deficient, with
the majority of workers not wearing gloves, not washing their hands
before eating or leaving the plant, and consuming food and beverages in
work areas.
Gomes (1972) examined Cr(VI)-induced skin lesions among
electroplaters in Sao Paulo, Brazil (Ex. 9-31). A clinical examination
of 303 workers revealed 88 (28.8%) had skin lesions, while 175 (58.0%)
had skin and mucus membrane lesions. A substantial number of employers
(26.6%) also did not provide personal protective equipment to workers.
The author attributed the high incidence of skin ulcers on the hands
and arms to inadequate personal protective equipment, and lack of
training for employees regarding hygiene practices.
Fleeger and Deng (1990) reported on an outbreak of skin ulcerations
among workers in a facility where enamel paints containing chromium
were applied to kitchen range parts (Ex. 9-97). A ground coat of paint
was applied to the parts, which were then placed on hooks and
transported through a curing oven. In some cases, small parts were
places on hooks before paint application. Tiny holes in the oven coils
apparently resulted in improper curing of the paint, leaving sharp
edges and a Cr(VI)-containing residue on the hooks. Most of the workers
who handled the hooks reportedly did not wear gloves, because the
gloves were said to reduce dexterity and decrease productivity. As a
result, cuts from the sharp edges allowed the Cr(VI) to penetrate the
skin, leading to ulcerations (Ex. 9-97).
2. Prognosis of Dermal Effects
Cr(VI)-related dermatitis tends to become more severe and
persistent with continuing exposure. Once established, the condition
may persist even if occupational exposure ceases. Fregert followed up
on cases of occupational contact dermatitis diagnosed over a 10-year
period by a dermatology service in Sweden. Based on responses to
questionnaires completed two to three years after treatment, only 7% of
women and 10% of men with Cr(VI)-related allergic contact dermatitis
were reported to be healed (Ex. 35-322). Burrows reviewed the condition
of patients diagnosed with work-related dermatitis 10-13 years earlier.
Only two of the 25 cases (8%) caused by exposure to cement had cleared
(Ex. 35-323).
Hogan et al. reviewed the literature regarding the prognosis of
contact dermatitis, and reported that the majority of patients had
persistent dermatitis (Ex. 35-324). It was reported that job changes
did not usually lead to a significant improvement for most patients.
The authors surveyed contact dermatitis experts around the world to
explore their experience with the prognosis of patients suffering from
occupational contact dermatitis of the hands. Seventy-eight percent of
the 51 experts who responded to the survey indicated that chromate was
one of the allergens associated with the worst possible prognosis.
Halbert et al. reviewed the experience of 120 patients diagnosed
with occupational chromate dermatitis over a 10-year period (Ex. 35-
320). The time between initial diagnosis and the review ranged from a
minimum of six months to a maximum of nine years. Eighty-four (70%) of
patients were reviewed two or more years after initial diagnosis, and
40 (33%) after five years or more. In the majority of cases (78, or
65%), the dermatitis was attributed to work with cement. For the study
population as a whole, 76% had ongoing dermatitis at the time of the
review.
When the review was conducted, 62 (58%) patients were employed in
the same occupation as when initially diagnosed. Fifty-five (89%) of
these workers continued to suffer from dermatitis. Fifty-eight patients
(48%) changed occupations after their initial diagnosis. Each of these
individuals indicated that they had changed occupations because of
their dermatitis. In spite of the change, dermatitis persisted in 40
members of this group (69%).
Lips et al. found a somewhat more favorable outcome among 88
construction workers with occupational chromate dermatitis who were
removed from Cr(VI) exposure (Ex. 35-325). Follow-up one to five years
after removal indicated that 72% of the patients no longer had
dermatitis. The authors speculated that this result might be due to
strict avoidance of Cr(VI) contact. Nonetheless, the condition
persisted in a substantial portion of the affected population.
3. Thresholds for Dermal Effects
In a response to OSHA's RFI submitted on behalf of the Chrome
Coalition, Exponent indicated that the findings of Fowler et al. (1999)
and others provide evidence of a threshold for elicitation of allergic
contact dermatitis (Ex. 31-18-1, p. 27). Exponent also stated that
because chrome ulcers did not develop in the Fowler et al. study,
"more aggressive" exposures appear to be necessary for the
development of chrome ulcers.
The Fowler et al. study involved the dermal exposure of 26
individuals previously sensitized to Cr(VI) who were exposed to water
containing 25 to 29 mg/L Cr(VI) as potassium dichromate (pH 9.4) (Ex.
31-18-5). Subjects immersed one arm in the Cr(VI) solution, while the
other arm was immersed in an alkaline buffer solution as a control.
Exposure lasted for 30 minutes and was repeated on three consecutive
days. Based on examination of the skin, the authors concluded that the
skin response experienced by subjects was not consistent with either
irritant or allergic contact dermatitis.
The exposure scenario in the Fowler et al. study, however, does not
take into account certain skin conditions often encountered in the
workplace. While active dermatitis, scratches, and skin lesions served
as criteria for excluding both initial and continuing participation in
the study, it is reasonable to expect that individuals with these
conditions will often continue to work. Cr(VI)-containing mixtures and
compounds used in the workplace may also pose a greater challenge to
the integrity of the skin than the solution used by Fowler et al. Wet
cement, for example, may have a pH higher than 9.4, and may be capable
of abrading or otherwise damaging the skin. As damaged skin is liable
to make exposed workers more susceptible to Cr(VI)-induced skin
effects, the suggested threshold is likely to be invalid. The absence
of chrome ulcers in the Fowler et al. study is not unexpected, because
subjects with "fissures or lesions" on the skin were excluded from
the study (Ex. 31-18-5). As discussed earlier, chrome ulcers are not
believed to occur on intact skin.
4. Conclusions
OSHA believes that adverse dermal effects from exposure to Cr(VI),
including irritant contact dermatitis, allergic contact dermatitis, and
skin ulceration, have been firmly established. The available evidence
is not sufficient to relate these effects to any given Cr(VI) air
concentration. Rather, it appears that direct dermal contact with
Cr(VI) is the most relevant factor in the development of dermatitis and
ulcers. Based on the findings of Gibb et al. (Ex. 32-22-12) and U.S.
Public Health Service (Ex. 7-3), OSHA believes that conjunctivitis may
result from direct eye contact with Cr(VI).
OSHA does not believe that the available evidence is sufficient to
establish a threshold concentration of Cr(VI) below which dermal
effects will not occur in the occupational environment. This finding is
supported not only by the belief that the exposure scenario of Fowler
et al. is not consistent with occupational exposures, but by experience
in the workplace as well. As summarized by Flyvholm et al. (1996),
numerous reports have indicated that allergic contact dermatitis occurs
in cement workers exposed to Cr(VI) concentrations below the threshold
suggested by Fowler et al. (1999). OSHA considers the evidence of
Cr(VI)-induced allergic contact dermatitis in these workers to indicate
that the threshold for elicitation of response suggested by Fowler et
al. (1999) is not applicable to the occupational environment.
E. Other Health Effects
OSHA has examined the possibility of health effect outcomes
associated with Cr(VI) exposure in addition to such effects as lung
cancer, nasal ulcerations and perforations, occupational asthma, and
irritant and allergic contact dermatitis. Unlike the Cr(VI)-induced
toxicities cited above, the data on other health effects do not
definitively establish Cr(VI)-related impairments of health from
occupational exposure at or below the previous OSHA PEL.
There is some positive evidence that workplace inhalation of Cr(VI)
results in gastritis and gastrointestinal ulcers, especially at high
exposures (generally over OSHA's previous PEL) (Ex. 7-12). This is
supported by ulcerations in the gastrointestinal tract of mice
breathing high Cr(VI) concentration for long periods (Ex. 10-8). Other
studies reported positive effects but significant information was not
reported or the confounders made it difficult to draw positive
conclusions (Ex. 3-84; Sassi 1956 as cited in Ex. 35-41). Other studies
reported negative results (Exs. 7-14; 9-135).
Likewise, several studies reported increases in renal proteins in
the urine of chromate production workers and chrome platers (Exs. 35-
107; 5-45; 35-105; 5-57). The Cr(VI) air levels recorded in these
workers were usually below the previous OSHA PEL (Exs. 35-107; 5-45).
Workers with the highest urinary chromium levels tended to also have
the largest elevations in renal markers (Ex. 35-107). One study
reported no relationship between chromium in urine and renal function
parameters, no relationship with age or with duration of exposure, and
no relationship between the presence of chromium skin ulcers and
chromium levels in urine or renal function parameters (Ex. 5-57). In
most studies, the elevated renal protein levels were restricted to only
one or two proteins out of several examined per study, generally
exhibited small increases (Ex. 35-105) and the effects appeared to be
reversible (Ex. 5-45). In addition, it has been stated that low
molecular weight proteinuria can occur from other reasons and cannot by
itself be considered evidence of chronic renal disease (Ex. 35-195).
Other human inhalation studies reported no changes in renal markers
(Exs. 7-27; 35-104). Animal inhalation studies did not report kidney
damage (Exs. 9-135; 31-18-11; 10-11; 31-18-10; 10-10). Some studies
with Cr(VI) administered by drinking water or gavage were positive for
increases in renal markers as well as some cell and tissue damage (Exs.
9-143; 11-10). However, it is not clear how to extrapolate such
findings to workers exposed to Cr(VI) via inhalation. Well-designed
studies of effects in humans via ingestion were not found.
OSHA did not find information to clearly and sufficiently
demonstrate that exposures to Cr(VI) result in significant impairment
to the hepatic system. Two European studies, positive for an excess of
deaths from cirrhosis of the liver and hepatobiliarity disorders, were
not able to separate chromium exposures from exposures to the many
other substances present in the workplace. The authors also could not
rule out the role of alcohol use as a possible contributor to the
disorder (Ex. 7-92; Sassi as cited in Ex. 35-41). Other studies did not
report any hepatic abnormalities (Exs. 7-27; 10-11).
The reproductive studies showed mixed results. Some positive
reproductive effects occurred in some welding studies. However, it is
not clear that Cr(VI) is the causative agent in these studies (Exs. 35-
109; 35-110; 35-108; 35-202; 35-203). Other positive studies were
seriously lacking in information. Information was not given on
exposures, the nature of the reproductive complications, or the women's
tasks (Shmitova 1980, 1978 as cited in Ex. 35-41, p. 52). ATSDR states
that because these studies were generally of poor quality and the
results were poorly reported, no conclusions can be made on the
potential for chromium to produce adverse reproductive effects in
humans (Ex. 35-41, p. 52). In animal studies, where Cr(VI) was
administered through drinking water or diet, positive developmental
effects occurred in offspring (Exs. 9-142; 35-33; 35-34; 35-38).
However, the doses administered in drinking water or given in the diet
were high (i.e., 250, 500, and 750 ppm). Furthermore, strong studies
showing reproductive or developmental effects in other situations where
employees were working exclusively with Cr(VI) were not found. In fact,
the National Toxicology Program (NTP) (Exs. 35-40; 35-42; 35-44)
conducted an extensive multigenerational reproductive assessment by
continuous breeding where the chromate was administered in the diet.
The assessment yielded negative results (Exs. 35-40; 35-42; 35-44).
Animal inhalation studies were also negative (Exs. 35-199; 9-135; 10-
10; Glaser 1984 as cited in Ex. 31-22-33;). Thus, it cannot be
concluded that Cr(VI) is a reproductive toxin for normal working
situations.
VI. Quantitative Risk Assessment
A. Introduction
The Occupational Safety and Health (OSH) Act and some landmark
court cases have led OSHA to rely on quantitative risk assessment,
where possible, to support the risk determinations required to set a
permissible exposure limit (PEL) for a toxic substance in standards
under the OSH Act. Section 6(b)(5) of the Act states that "The
Secretary [of Labor], in promulgating standards dealing with toxic
materials or harmful agents under this subsection, shall set the
standard which most adequately assures, to the extent feasible, on the
basis of the best available evidence, that no employee will suffer
material impairment of health or functional capacity even if such
employee has regular exposure to the hazard dealt with by such standard
for the period of his working life." (29 U.S.C. 651 et seq.)
In a further interpretation of the risk requirements for OSHA
standard setting, the United States Supreme Court, in the 1980
"benzene" decision, (Industrial Union Department, AFL-CIO v. American
Petroleum Institute, 448 U.S. 607 (1980)) ruled that the OSH Act
requires that, prior to the issuance of a new standard, a determination
must be made that there is a significant risk of material impairment of
health at the existing PEL and that issuance of a new standard will
significantly reduce or eliminate that risk. The Court stated that
"before he can promulgate any
permanent health or safety standard, the Secretary is required to make
a threshold finding that a place of employment is unsafe in the sense
that significant risks are present and can be eliminated or lessened by
a change in practices" [448 U.S. 642]. The Court also stated "that
the Act does not limit the Secretary's power to require the elimination
of significant risks" [488 U.S. 644]. While the Court indicated that
the use of quantitative risk analysis was an appropriate means to
establish significant risk, they made clear that "OSHA is not required
to support its finding that a significant risk exists with anything
approaching scientific certainty."
The Court in the Cotton Dust case, (American Textile Manufacturers
Institute v. Donovan, 452 U.S. 490 (1981)) found that Section 6(b)(5)
of the OSH Act places benefits to worker health above all other
considerations except those making attainment of the health benefits
unachievable and, therefore, only feasibility analysis of OSHA health
standards is required and not cost-benefit analysis. It reaffirmed its
previous position in the "benzene" case, however, that a risk
assessment is not only appropriate but should be used to identify
significant health risk in workers and to determine if a proposed
standard will achieve a reduction in that risk. Although the Court did
not require OSHA to perform a quantitative risk assessment in every
case, the Court implied, and OSHA as a matter of policy agrees, that
assessments should be put into quantitative terms to the extent
possible.
The determining factor in the decision to perform a quantitative
risk assessment is the availability of suitable data for such an
assessment. As reviewed in section V.B. on Carcinogenic Effects, there
are a substantial number of occupational cohort studies that reported
excess lung cancer mortality in workers exposed to Cr(VI) in several
industrial operations. Many of these found that workers exposed to
higher levels of airborne Cr(VI) for a longer period of time had
greater standardized mortality ratios (SMRs) for lung cancer.
OSHA believes that two recently studied occupational cohorts by
Gibb et al. (Ex. 31-22-11) and Luippold et al. (Ex. 33-10) have the
strongest data sets on which to quantify lung cancer risk from
cumulative Cr(VI) exposure (i.e., air concentration x exposure
duration). A variety of exposure-response models were fit to these
data, including linear relative risk, quadratic relative risk, log-
linear relative risk, additive risk, and Cox proportional hazards
models. Using a linear relative risk model on these data to predict
excess lifetime risk, OSHA estimated that the lung cancer risk from a
45 year occupational exposure to Cr(VI) at an 8-hour TWA at the
previous PEL of 52 [mu]g/m\3\ is 101 to 351 excess deaths per 1000.
Quantitative lifetime risk estimates from a working lifetime exposure
at several lower alternative PELs under consideration by the Agency
were also estimated. The sections below discuss the selection of the
appropriate data sets and risk models, the estimation of lung cancer
risks based on the selected data sets and models, the uncertainty in
the risk estimates, and the key issues that were raised in comments
received during the public hearing process.
A preliminary quantitative risk assessment was previously published
in the Notice of Proposed Rulemaking (69 FR at 59306, 10/4/2004). This
was peer-reviewed by three outside experts in the fields of
occupational epidemiology and risk assessment. Their comments were
discussed in the NPRM (69 FR at 59385-59388). They commented on the
suitability of several occupational data sets for exposure-response
analysis, the choice of exposure metric and risk model, the
appropriateness of the risk estimates, and the characterization of key
issues and uncertainties. The reviewers agreed that the soluble
chromate production cohorts described by Gibb et al. and Luippold et
al. provided the strongest data sets for quantitative risk assessment.
They concurred that a linear model using cumulative exposure based on
time-weighted average Cr(VI) air concentrations by job title and
employment history was the most reasonable risk assessment approach.
The experts showed less enthusiasm for average monthly Cr(VI) air
concentrations as an appropriate exposure metric or for an exposure
threshold below which there is no lung cancer risk. They found the
range of excess lifetime lung cancer risks presented by OSHA to be
sound and reasonable. They offered suggestions regarding issues such as
the impact of cigarette smoking and the healthy worker effect on the
assessment of risk. OSHA revised the preliminary quantitative risk
assessment in several respects based on these peer review comments.
In contrast to the more extensive occupational cohort data on
Cr(VI) exposure-response, data from experimental animal studies are
less suitable for quantitative risk assessment of lung cancer. Besides
the obvious species difference, most of the animal studies administered
Cr(VI) to the respiratory tract by less relevant routes, such as
instillation or implantation. The few available inhalation studies in
animals were limited by a combination of inadequate exposure levels,
abbreviated durations, and small numbers of animals per dose group.
Despite these limitations, the animal data do provide semi-quantitative
information with regard to the relative carcinogenic potency of
different Cr(VI) compounds. A more detailed discussion can be found in
sections V.B.7 and V.B.9.
The data that relate non-cancer health impairments, such as damage
to the respiratory tract and skin, to Cr(VI) exposure are also not well
suited for quantitative assessment. There are some data from cross-
sectional studies and worker surveys that group the prevalence and
severity of nasal damage by contemporary time-weighted average (TWA)
Cr(VI) air measurements. However, there are no studies that track
either incidence or characterize exposure over time. Nasal damage is
also more likely influenced by shorter-term peak exposures that have
not been well characterized. While difficult to quantify, the data
indicate that the risk of damage to the nasal mucosa will be
significantly reduced by lowering the previous PEL, discussed further
in section VII on Significance of Risk.
There are even less suitable exposure-response data to assess risk
for other Cr(VI)-induced impairments (e.g., mild renal damage,
gastrointestinal ulceration). With the possible exception of
respiratory tract effects (e.g., nasal damage, occupational asthma),
the risk of non-cancer adverse effects that result from inhaling Cr(VI)
are expected to be very low, except as a result of long-term regular
airborne exposure around or above the previous PEL (52 [mu]g/m\3\).
Since the non-cancer effects occur at relatively high Cr(VI) air
concentrations, OSHA has concluded that lowering the PEL to reduce the
risk of developing lung cancer over a working lifetime will also
eliminate or reduce the risk of developing these other health
impairments. As discussed in section V.E., adverse effects to the skin
primarily result from dermal rather than airborne exposure.
B. Study Selection
The more than 40 occupational cohort studies reviewed in Section
VI.B on carcinogenic effects were evaluated to determine the adequacy
of the exposure-response information for the quantitative assessment of
lung cancer risk associated with Cr(VI) exposure. The key criteria were
data that allowed for estimation of input variables,
specifically levels of exposure and duration of exposure (e.g.,
cumulative exposure in mg/m\3\-yr); observed numbers of cancers (deaths
or incident cases) by exposure category; and expected (background)
numbers of cancer deaths by exposure category.
Additional criteria were applied to evaluate the strengths and
weaknesses of the available epidemiological data sets. Studies needed
to have well-defined cohorts with identifiable cases. Features such as
cohort size and length of follow-up affect the ability of the studies
to detect any possible effect of Cr(VI) exposure. Potential confounding
of the responses due to other exposures was considered. Study
evaluation also considered whether disease rates from an appropriate
reference population were used to derive expected numbers of lung
cancers. One of the most important factors in study evaluation was the
ascertainment and use of exposure information (i.e., well-documented
historical exposure data). Both level and duration of exposure are
important in determining cumulative dose, and studies are often
deficient with respect to the availability or use of such information.
Two recently studied cohorts of chromate production workers, the
Gibb cohort and the Luippold cohort, were found to be the strongest
data sets for quantitative assessment (Exs. 31-22-11; 33-10). Of the
various studies, these two had the most extensive and best documented
Cr(VI) exposures spanning three or four decades. Both cohort studies
characterized observed and expected lung cancer mortality and reported
a statistically significant positive association between lung cancer
risk and cumulative Cr(VI) exposure. For the remainder of this preamble
the Gibb and Luippold cohorts are referred to as the "preferred
cohorts", denoting that they are the cohorts used to derive OSHA's
model of lung cancer risk from exposure to Cr(VI).
Four other cohorts (Mancuso, Hayes et al., Gerin et al., and
Alexander et al.) had less satisfactory data for quantitative
assessments of lung cancer risk (Exs. 7-11; 23; 7-14; 7-120; 31-16-3).
These cohorts include chromate production workers, stainless steel
welders, and aerospace manufacturing workers. While the lung cancer
response in these cohorts was stratified across multiple exposure
groups, there were limitations to these data that affected their
reliability for quantitative risk assessment. OSHA therefore did not
consider them to be preferred cohorts (i.e., they were not used to
derive OSHA's model of lung cancer risk from exposure to Cr(VI)).
However, OSHA believes that quantitative analysis of these cohorts
provides valuable information to the risk assessment, especially for
the purpose of comparison with OSHA's risk model based on the preferred
Gibb and Luippold cohorts. Analyses based on the Mancuso, Hayes et al.,
Gerin et al., and Alexander et al. cohorts, referred to as "additional
cohorts" for the remainder of this preamble, were compared with the
assessments based on the Gibb and Luippold cohorts. The strengths and
weaknesses of all six cohorts as a basis for exposure-response analysis
are discussed in more detail below.
1. Gibb Cohort
The Gibb et al. study was a particularly strong study for
quantitative risk assessment, especially in terms of cohort size and
historical exposure data (Exs. 31-22-11; 33-11). Gibb et al. studied an
updated cohort from the same Baltimore chromate production plant
previously studied by Hayes et al. (see section VI.B.4). The cohort
included 2357 male workers (white and non-white) first employed between
1950 and 1974. Follow-up was through the end of 1992 for a total of
70,736 person-years and an average length of 30 years per cohort
member. Smoking status and amount smoked in packs per day at the start
of employment was available for the majority of the cohort members.
A significant advantage of the Gibb data was the availability of a
large number of personal and area sampling measurements from a variety
of locations and job titles which were collected over the years during
which the cohort members were exposed (from 1950 to 1985, when the
plant closed). Using these concentration estimates, a job exposure
matrix was constructed giving annual average exposures by job title.
Based on the job exposure matrix and work histories for the cohort
members, Gibb et al. computed the person-years of observation, the
observed numbers of lung cancer deaths, and the expected numbers of
lung cancer deaths categorized by cumulative Cr(VI) exposure and age of
death. They found that cumulative Cr(VI) exposure was a significant
predictor of lung cancer risk over the exposure range of 0 to 2.76
(meanSD = 0.702.75) mg/m\3\-yr. This included a
greater than expected number of lung cancer deaths among relatively
young workers. For example, chromate production workers between 40 and
50 years of age with mean cumulative Cr(VI) exposure of 0.41 mg
CrO3/m\3\-yr (equivalent to 0.21 mg Cr(VI)/m\3\-yr) were
about four times more likely to die of lung cancer than a State of
Maryland resident of similar age (Ex. 31-22-11, Table V).
The data file containing the demographic, exposure, smoking, and
mortality data for the individual cohort members was made available to
OSHA (Ex. 295). These data were used in several reanalyses to produce
several different statistical exposure-response models and to explore
various issues raised in comments to OSHA, such as the use of linear
and nonlinear exposure-response models, the difference between modern
and historical levels of Cr(VI) exposure, and the impact of including
or excluding short-term workers from the exposure-response analysis.
The Agency's access to the dataset and to reanalyses of it performed by
several different analysts has been a tremendous advantage in its
consideration of these and other issues in the development of the final
risk assessment.
2. Luippold Cohort
The other well-documented exposure-response data set comes from a
second cohort of chromate production workers. Luippold et al. studied a
cohort of 482 predominantly white, male employees who started work
between 1940 and 1972 at the same Painesville, Ohio plant studied
earlier by Mancuso (Ex. 33-10) (see subsection VI.B.3). Mortality
status was followed through 1997 for a total of 14,048 person-years.
The average worker had 30 years of follow-up. Cr(VI) exposures for the
Luippold cohort were based on 21 industrial hygiene surveys conducted
at the plant between 1943 and 1971, yielding a total of more than 800
area samples (Ex. 35-61). A job exposure matrix was computed for 22
exposure areas for each month of plant operation starting in 1940 and,
coupled with detailed work histories available for the cohort members,
cumulative exposures were calculated for each person-year of
observation. Luippold et al. found significant dose-related trends for
lung cancer SMRs as a function of year of hire, duration of employment,
and cumulative Cr(VI) exposure. Risk assessments on the Luippold et al.
study data performed by Crump et al. had access to the individual data
and, therefore, had the best basis for analysis of this cohort (Exs.
31-18-1; 35-205; 35-58).
While the Luippold cohort was smaller and less racially diverse
than the Gibb cohort, the workforce contained fewer transient, short-
term employees. The Luippold cohort consisted entirely of workers
employed over one year. Fifty-five percent worked
for more than five years. In comparison, 65 percent of the Gibb cohort
worked for less than a year and 15 percent for more than five years at
the Baltimore plant. There was less information about the smoking
behavior (smoking status available for only 35 percent of members) of
the Luippold cohort than the Gibb cohort.
One aspect that the Luippold cohort had in common with the Gibb
cohort was extensive and well-documented air monitoring of Cr(VI). The
quality of exposure information for both the Gibb and Luippold cohorts
was considerably better than that for the Mancuso, Hayes et al., Gerin
et al., and Alexander et al. cohorts. The cumulative Cr(VI) exposures
for the Luippold cohort, which ranged from 0.003 to 23 (mean< plus-
minus>SD = 1.582.50) mg Cr(VI)/m\3\-yr, were generally
higher but overlapped those of the Gibb cohort. The use of individual
work histories to define exposure categories and presentation of mean
cumulative doses in the exposure groups provided a strong basis for a
quantitative risk assessment. The higher cumulative exposure range and
the longer work duration of the Luippold cohort serve to complement
quantitative data available on the Gibb cohort.
3. Mancuso Cohort
Mancuso (Ex. 7-11) studied the lung cancer incidence of an earlier
cohort of 332 white male employees drawn from the same plant in
Painesville, Ohio that was evaluated by the Luippold group. The Mancuso
cohort was first employed at the facility between 1931 and 1937 and
followed up through 1972, when the plant closed. Mancuso (Ex. 23) later
extended the follow-up period through 1993, yielding a total of 12,881
person-years of observation for an average length of 38.8 years and a
total of 66 lung cancer deaths. Since the Mancuso workers were first
employed in the 1930s and the Luippold workers were first employed
after 1940, the two cohorts are completely different sets of
individuals.
A major limitation of the Mancuso study is the uncertainty of the
exposure data. Mancuso relied exclusively on the air monitoring
reported by Bourne and Yee (Ex. 7-98) conducted over a single short
period of time during 1949. Bourne and Yee presented monitoring data as
airborne insoluble chromium, airborne soluble chromium, and total
airborne chromium by production department at the Painesville plant.
The insoluble chromium was probably Cr(III) compounds with some
slightly water-soluble and insoluble chromates. The soluble chromium
was probably highly water-soluble Cr(VI). Mancuso (Exs. 7-11; 23)
calculated cumulative exposures (mg/m\3\-yr) for each cohort member
based on the 1949 mean chromium concentrations, by production
department, under the assumption that those levels reflect exposures
during the entire duration of employment for each cohort member, even
though employment may have begun as early as 1931 and may have extended
to 1972. Due to the lack of air measurements spanning the full period
of worker exposure and the lack of adequate methodology to distinguish
chromium valence states (i.e., Cr(VI) vs. Cr(III)), the exposure data
associated with the Mancuso cohort were not as well characterized as
data from the Luippold or Gibb cohorts.
Mancuso (Exs. 7-11; 23)reported cumulative exposure-related
increases in age-adjusted lung cancer death rates for soluble,
insoluble, or total chromium. Within a particular range of exposures to
insoluble chromium, lung cancer death rates also tended to increase
with increasing total cumulative chromium. However, the study did not
report whether these tendencies were statistically significant, nor did
it report the extent to which exposures to soluble and insoluble
chromium were correlated. Thus, it is possible that the apparent
relationship between insoluble chromium (e.g., primarily Cr(III)) and
lung cancer may have arisen because both insoluble chromium
concentrations and lung cancer death rates were positively correlated
with Cr(VI) concentrations. Further discussion with respect to
quantitative risk estimation from the Mancuso cohort is provided in
section VI.E.1 on additional risk assessments.
4. Hayes Cohort
Hayes et al. (Ex. 7-14) studied a cohort of employees at the same
chromate production site in Baltimore examined by Gibb et al. The Hayes
cohort consisted of 2101 male workers who were first hired between 1945
and 1974, excluding those employed for less than 90 days. The Gibb
cohort had different but partially overlapping date criteria for first
employment (1950-1974) and no 90 day exclusion. Hayes et al. reported
SMRs for respiratory tract cancer based on workers grouped by time of
hire, employment duration, and high or low exposure groups. Workers who
had ever worked at an older plant facility and workers whose location
of employment could not be determined were combined into a single
exposure group referred to as "high or questionable" exposure.
Workers known to have been employed exclusively at a newer renovated
facility built in 1950 and 1951 were considered to have had "low"
exposure. A dose-response was observed in the sense that higher SMRs
for respiratory cancer were observed among long-term workers (workers
who had worked for three or more years) than among short-term workers.
Hayes et al. did not quantify occupational exposure to Cr(VI) at
the time the cohort was studied, but Braver et al. (Ex. 7-17) later
estimated average cumulative soluble chromium (presumed by the authors
to be Cr(VI)) exposures for four subgroups of the Hayes cohort first
employed between 1945 and 1959. The TWA Cr(VI) concentrations were
determined from a total of 555 midget impinger air measurements that
were collected at the older plant from 1945 to 1950. The cumulative
exposures for the subgroups were estimated from the yearly average
Cr(VI) exposure for the entire plant and the subgroups' average
duration of employment rather than job-specific Cr(VI) concentrations
and individual work histories. Such "group level" estimation of
cumulative exposure is less appropriate than the estimation based on
individual experiences as was done for the Gibb and Luippold cohorts.
A more severe limitation of this study is that exposures attributed
to many workers in the newly renovated facility at the Baltimore site
throughout the 1950s were based on chromium measurements from an
earlier period (i.e., 1949-1950) at an older facility. Samples
collected at the new facility and reviewed by Gibb et al. (Exs. 25, 31-
22-12) show that the exposures in the new facility were substantially
lower than assumed by Braver et al. Braver et al. (Ex. 7-17) discussed
a number of other potential sources of uncertainty in the Cr(VI)
exposure estimates, such as the possible conversion to Cr(III) during
sample collection and the likelihood that samples may have been
collected mainly in potential problem areas.
5. Gerin Cohort
Gerin et al. (Ex. 7-120) developed a job exposure matrix that was
used to estimate cumulative Cr(VI) exposures for male stainless steel
welders who were part of the International Agency for Research on
Cancer's (IARC) multi-center historical cohort study (Ex. 7-114). The
IARC cohort included 11,092 welders. However, the number of cohort
members who were stainless steel welders, for which Cr(VI) exposures
were estimated, could not be determined from their report. Gerin et al.
used occupational hygiene surveys reported in the published literature,
including a limited amount of data collected from 8 of the 135
companies that employed welders in the cohort, to estimate typical eight-hour
TWA Cr(VI) breathing zone concentrations for various combinations of
welding processes and base metal. The resulting exposure matrix was
then combined with information about individual work history, including
time and length of employment, type of welding, base metal welded, and
information on typical ventilation status for each company (e.g.,
confined area, use of local exhaust ventilation, etc.) to estimate the
cumulative Cr(VI) exposure. Individual work histories were not
available for about 25 percent of the stainless steel welders. In these
cases, information was assumed based on the average distribution of
welding practices within the company. The lack of Cr(VI) air
measurements from most of the companies in the study and the
limitations in individual work practice information for this cohort
raise questions concerning the accuracy of the exposure estimates.
Gerin et al. reported no upward trend in lung cancer mortality
across four cumulative Cr(VI) exposure categories for stainless steel
welders, each accumulating between 7,000 and 10,000 person-years of
observation. The welders were also known to be exposed to nickel,
another potential lung carcinogen. Co-exposure to nickel may obscure or
confound the Cr(VI) exposure-response relationship. As discussed
further in Sections VI.E.3 and VI.G.4, exposure misclassification in
this cohort may obscure an exposure-response relationship. This is the
primary reason that the Gerin et al. cohort was not considered a
preferred cohort (i.e., it was not used to derive OSHA's quantitative
risk estimates), although a quantitative analysis of this cohort was
performed for comparison with the preferred cohorts.
6. Alexander Cohort
Alexander et al. (Ex. 31-16-3) conducted a retrospective cohort
study of 2429 aerospace workers employed in jobs entailing chromate
exposure (e.g., spray painting, sanding/polishing, chrome plating,
etc.) between 1974 and 1994. The cohort included workers employed as
early as 1940. Follow-up time was short, averaging 8.9 years per cohort
member; in contrast, the Gibb and Luippold cohorts accumulated an
average 30 or more years of follow-up. Long-term follow-up of cohort
members is particularly important for determining the risk of lung
cancer, which typically has an extended latency period of twenty years
or more.
Industrial hygiene data collected between 1974 and 1994 were used
to classify jobs in categories of "high" exposure, "moderate"
exposure, or "low" exposure to Cr(VI). The use of respiratory
protection was accounted for when setting up the job exposure matrix.
These exposure categories were assigned summary TWA concentrations and
combined with individual job history records to estimate cumulative
exposures for cohort members over time. As further discussed in section
VI.E.4, it was not clear from the study whether exposures are expressed
in units of Cr(VI) or chromate (CrO3). Exposures occurring
before 1974 were assumed to be at TWA levels assigned to the interval
from 1974 to 1985.
Alexander et al. presented lung cancer incidence data for four
cumulative chromate exposure categories based on worker duration and
the three (high, moderate, low) exposure levels. Lung cancer incidence
rates were determined using a local cancer registry, part of the
National Cancer Institute (NCI) Surveillance Epidemiology and End
Results (SEER) program. The authors reported no positive trend in lung
cancer incidence with increasing Cr(VI) exposure. Limitations of this
cohort study include the young age of the cohort members (median = 42)
and lack of information on smoking. As discussed above, the follow-up
time (average < 9 years) was probably too short to capture lung cancers
resulting from Cr(VI) exposure. Finally, the available Cr(VI) air
measurement data did not span the entire employment period of the
cohort (e.g., no data for 1940 to 1974) and was heavily grouped into a
relatively small number of "summary" TWA concentrations that may not
have fully captured individual differences in workplace exposures to
Cr(VI). For the above reasons, in particular the insufficient follow-up
time for most cohort members, the Alexander cohort was not considered a
preferred dataset for OSHA's quantitative risk analysis. However, a
quantitative analysis of this cohort was performed for comparison with
the preferred cohorts.
7. Studies Selected for the Quantitative Risk Assessment
The epidemiologic database is quite extensive and contains several
studies with exposure and response data that could potentially be used
for quantitative risk assessment. OSHA considers certain studies to be
better suited for quantitative assessment than others. The Gibb and
Luippold cohorts are the preferred sources for quantitative risk
assessment because they are large, have extensive follow-up, and have
documentation of historical Cr(VI) exposure levels superior to the
Mancuso, Hayes, Gerin and Alexander cohorts. In addition, analysts have
had access to the individual job histories of cohort members and
associated exposure matrices. OSHA's selection of the Gibb and Luippold
cohorts as the best basis of exposure-response analysis for lung cancer
associated with Cr(VI) exposure was supported by a variety of
commenters, including for example NIOSH (Tr. 314; Ex. 40-10-2, p. 4),
EPRI (Ex. 38-8, p.6), and Exponent (Ex. 38-215-2, p. 15). It was also
supported by the three external peer reviewers who reviewed OSHA's
preliminary risk assessment, Dr. Gaylor (Ex. 36-1-4-1, p. 24), Dr.
Smith (Ex. 36-1-4-2 p. 28), and Dr. Hertz-Picciotto (Ex. 36-1-4-4, pp.
41-42).
The Mancuso cohort and the Hayes cohort were derived from workers
at the same plants as Luippold and Gibb, respectively, but have
limitations associated with the reporting of quantitative information
and exposure estimates that make them less suitable for risk
assessment. Similarly, the Gerin and Alexander cohorts are less
suitable, due to limitations in exposure estimation and short follow-
up, respectively. For these reasons, OSHA did not rely upon the
Mancuso, Hayes, Gerin, and Alexander cohorts to derive its exposure-
response model for the risk of lung cancer from Cr(VI).
Although the Agency did not rely on the Mancuso, Hayes, Gerin, and
Alexander studies to develop its exposure-response model, OSHA believes
that evaluating risk among several different worker cohorts and
examining similarities and differences between them adds to the overall
completeness and quality of the assessment. The Agency therefore
analyzed these datasets and compared the results with the preferred
Gibb and Luippold cohorts. This comparative analysis is discussed in
Section VI.E. In light of the extensive worker exposure-response data,
there is little additional value in deriving quantitative risk
estimates from tumor incidence results in rodents, especially
considering the concerns with regard to route of exposure and study
design.
OSHA received a variety of public comments regarding the overall
quality of the Gibb and Luippold cohorts and their suitability as the
preferred cohorts in OSHA's quantitative risk analysis. Some commenters
raised concerns about the possible impact of short-term workers in the
Gibb cohort on the risk assessment (Tr. 123; Exs. 38-106, p. 10, 21;
40-12-5, p. 9). The Gibb cohort's inclusion of many workers employed
for short periods of time was cited as a
"serious flaw" by one commenter, who suggested that many lung cancers
among short-term workers in the study were caused by unspecified other
factors (Ex. 38-106, p. 10, p. 21). Another commenter stated that the
Davies cohort of British chromate production workers "gives greater
credence to the Painesville cohort as it showed that brief exposures
(as seen in a large portion of the Baltimore cohort) did not have an
increased risk of lung cancer" (Ex. 39-43, p. 1). However, separate
analyses of the short-term (< 1 year employment) and longer-term ( 1
year) Gibb cohort members indicated that restriction of the cohort to
workers with tenures of at least one year did not substantially impact
estimates of excess lung cancer mortality (Ex. 31-18-15-1 , p. 29). At
the public hearing, Ms. Deborah Proctor of Exponent, Inc. stated that
"the short term workers did not affect the results of the study" (Tr.
1848). OSHA agrees with Ms. Proctor's conclusion, and does not believe
that the inclusion of short term workers in the Gibb cohort is a source
of substantial uncertainty in the Agency's risk estimates.
Some commenters expressed concern that the Gibb study did not
control for smoking (Exs. 38-218, pp. 20-21; 38-265, p. 28; 39-74, p.
3). However, smoking status at the time of employment was ascertained
for approximately 90% of the cohort (Ex. 35-435) and was used in
statistical analyses by Gibb et al., Environ Inc., and Exponent Inc. to
adjust for the effect of smoking on lung cancer in the cohort (Exs. 25;
31-18-15-1; 35-435). NIOSH performed similar analyses using more
detailed information on smoking level (packs per day) that was
available for 70% of the cohort (Ex. 35-435, p.1100). OSHA believes
that these analyses appropriately addressed the potential confounding
effect of smoking in the Gibb cohort. Issues and analyses related to
smoking are further discussed in Section VI.G.3.
Other issues and uncertainties raised about the Gibb and Luippold
cohorts include a lack of information necessary to estimate deposited
dose of Cr(VI) for workers in either cohort and a concern that the
Luippold exposure data were based on exposures to "airborne total
soluble and insoluble chromium* * * rather than exposures to Cr(VI)"
(Ex. 38-218, pp. 20-21). However, the exposure estimates for the
Luippold (2003) cohort were recently developed by Proctor et al. using
measurements of airborne Cr(VI), not the total chromium measurements
used previously in Mancuso et al.'s analysis (Exs. 35-58, p. 1149; 35-
61). And, while it is true that the Gibb and Luippold (2003) datasets
do not lend themselves to construction of deposited dose measures, the
extensive Cr(VI) air monitoring data available on these cohorts are
more than adequate for quantitative risk assessment. In the case of the
Gibb cohort, the exposure dataset is extraordinarily comprehensive and
well-documented (Tr. 709-710; Ex. 44-4, p.2), even "exquisite"
according to one NIOSH expert (Tr. 312). Further discussion of the
quality and reliability of the Gibb and Luippold (2003) exposure data
and related comments appears in Section VI.G.1.
OSHA received several comments regarding a new epidemiological
study conducted by Environ, Inc. for the Industrial Health Foundation,
Inc. of workers hired after the institution of process changes and
industrial hygiene practices designed to limit exposure to Cr(VI) in
two chromate production plants in the United States and two plants in
Germany (Exs. 47-24-1; 47-27, pp. 15-16; 47-35-1, pp. 7-8). These
commenters suggested that OSHA should use these cohorts to model risk
of lung cancer from low exposures to Cr(VI). Unfortunately, the public
did not have a chance to comment on this study because documents
related to it were submitted to the docket after the time period when
new information should have been submitted. However, OSHA reviewed the
study and comments that were submitted to the docket. Based on the
information submitted, the Agency does not believe that quantitative
analysis of these studies would provide additional information on risk
from low exposures to Cr(VI).
A cohort analysis based on the U.S. plants is presented in an April
2005 publication by Luippold et al. (Ex. 47-24-2). Luippold et al.
studied a total of 617 workers with at least one year of employment,
including 430 at a plant built in the early 1970s ("Plant 1") and 187
hired after the 1980 institution of exposure-reducing process and work
practice changes in a second plant ("Plant 2"). Workers were followed
through 1998. Personal air-monitoring measures available from 1974 to
1988 for the first plant and from 1981 to 1998 for the second plant
indicated that exposure levels at both plants were low, with overall
geometric mean concentrations below 1.5 [mu]g/m3 and area-
specific average personal air sampling values not exceeding 10 [mu]g/
m3 for most years (Ex. 47-24-2, p. 383). By the end of
follow-up, which lasted an average of 20.1 years for workers at Plant 1
and 10.1 years at Plant 2, 27 cohort members (4%) were deceased. There
was a 41% deficit in all-cause mortality when compared to all-cause
mortality from age-specific state reference rates, suggesting a strong
healthy worker effect. Lung cancer was 16% lower than expected based on
three observed vs. 3.59 expected cases, also using age-specific state
reference rates (Ex. 47-24-2, p. 383). The authors concluded that
"[t]he absence of an elevated lung cancer risk may be a favorable
reflection of the postchange environment. However, longer follow-up
allowing an appropriate latency for the entire cohort will be needed to
confirm this conclusion" (Ex. 47-24-2, p. 381).
OSHA agrees with the study authors that the follow-up in this study
was not sufficiently long to allow potential Cr(VI)-related lung cancer
deaths to occur among many cohort members. The mean times since first
exposure of 10 and 20 years for Plant 1 and Plant 2 employees,
respectively, suggest that most workers in the cohort may not have
completed the " * * * typical latency period of 20 years or more"
that Luippold et al. suggest is required for occupational lung cancer
to emerge (Ex. 47-24-2, p. 384). Other important limitations of this
study include the striking healthy worker effect on the SMR analysis,
and the relatively young age of most workers at the end of follow-up
(approximately 90% < 60 years old) (Ex. 47-24-2, p. 383). OSHA also
agrees with the study authors' statements that " * * * the few lung
cancer deaths in this cohort precluded * * * [analyses to] evaluate
exposure-response relationships * * * " (Ex. 47-24-2, p. 384).
Although OSHA's model predicts high excess lung cancer risk for
highly exposed individuals (e.g., workers exposed for 45 years at the
previous PEL of 52 [mu]g/m3), the model would predict much
lower risks for workers with low exposures, as in the Luippold (2005)
cohorts. To provide a point of comparison between the results of the
Luippold et al. (2005) 'post-change' study and OSHA's risk model, the
Agency used its risk model to generate an estimate of lung cancer risk
for a population with exposure characteristics approximately similar to
the 'post-change' cohorts described in Luippold et al. (2005). It
should be noted that since this comparative analysis used year 2000
U.S. reference rates were rather than the state-, race-, and gender-
specific historical reference mortality rates used by Luippold et al.
(2005), this risk calculation provides only a rough estimate of
expected excess lung cancer risk for the cohort. The derivation of
OSHA's risk model (based on the preferred Gibb and Luippold
(2003) cohorts) is described in Sections VI.C.1 and VI.C.2.
It is difficult to tell from the publication what the average level
or duration of exposure was for the cohort. However, personal sampling
data reported by Luippold et al. (2005) had annual geometric mean 8-
hour TWA concentrations "much less" than 1.5 [mu]g/m3 in
most years (Ex. 47-24-2, p. 383). Most workers also probably had less
than 20 years of exposure, given the average follow-up periods of 20
and 10 years reported for the Luippold (2005) Plant 1 and Plant 2,
respectively. OSHA assumed that workers had TWA exposures of 1.5 [mu]g/
m3 for 20 years, with the understanding that this assumption
would lead to somewhat higher estimates of risk than OSHA s model would
predict if the average exposure of the cohort was known. Using these
assumptions, OSHA's model predicts a 2-9% excess lung cancer risk due
to Cr(VI) exposure, or less than four cancers in the population the
size and age of the Luippold 2005 cohort.
Since this analysis used year 2000 U.S. reference rates rather than
the state-, race-, and gender-specific historical reference mortality
rates used by Luippold et al. (2005), this risk calculation provides
only a rough estimate of the lung cancer risk that OSHA's model would
predict for the cohort. Nevertheless, it illustrates that for a
relatively young population with low exposures, OSHA's risk model
(derived from the preferred Gibb and Luippold 2003 cohorts) predicts
lung cancer risk similar to that observed in the low-exposure Luippold
2005 cohort. The small number of lung cancer deaths observed in
Luippold 2005 should not be considered inconsistent with the risk
estimates derived using models developed by OSHA based on the Gibb and
Luippold (2003) cohorts (Ex. 47-24-2, p. 383).
Some commenters believed that analysis of the unpublished German
cohorts would demonstrate that lung cancer risk was only increased at
the highest Cr(VI) levels and, therefore, could form the basis for an
exposure threshold (Exs. 47-24-1; 47-35-1). Although no data were
provided to corroborate their comments, the Society of the Plastics
Industry requested that OSHA obtain and evaluate the German study as
"new and available evidence which may suggest a higher PEL than
proposed" (Ex. 47-24-1, p. 4).
Following the close of the comment period, OSHA gained access to a
2002 final contract report by Applied Epidemiology Inc. prepared for
the Industrial Health Foundation (Ex. 48-1-1; 48-1-2) and a 2005
prepublication by ENVIRON Germany (Ex. 48-4). The 2002 report contained
detailed cohort descriptions, exposure assessments, and mortality
analyses of 'post-change' workers from the two German chromate
production plants referred to above and two U.S. chromate production
plants, one of which is plant 1 discussed in the 2005 study by Luippold
et al. The mortality and multivariate analyses were performed on a
single combined cohort from all four plants. The 2005 prepublication
contained a more abbreviated description and analysis of a smaller
cohort restricted to the two German plants only. The cohorts are
referred to as 'post-change' because the study only selected workers
employed after the participating plants switched from a high-lime to a
no-lime (or very low lime facility, in the case of U.S. plant 1)
chromate production process and implemented industrial hygiene
improvements that considerably reduced Cr(VI) air levels in the
workplace.
The German cohort consisted of 901 post-change male workers from
two chromate production plants employed for at least one year.
Mortality experience of the cohort was evaluated through 1998. The
study found elevated lung cancer mortality (SMR=1.48 95% CI: 0.93-2.25)
when compared to the age- and calendar year-adjusted German national
population rates (Ex. 48-4). The cohort lacked sufficient job history
information and air monitoring data to develop an adequate job-exposure
matrix required to estimate individual airborne exposures (Ex. 48-1-2).
Instead, the researchers used the large amount of urinary chromium data
from routine biomonitoring of plant employees to analyze lung cancer
mortality using cumulative urinary chromium as an exposure surrogate,
rather than the conventional cumulative Cr(VI) air concentrations. The
study reported a statistically significant two-fold excess lung cancer
mortality (SMR=2.09; 95% CI: 1.08-3.65; 12 observed lung cancer deaths)
among workers in the highest cumulative exposure grouping (i.e. >200
[mu]g Cr/L--yr). There was no increase in lung cancer mortality in the
lower exposure groups, but the number of lung cancer deaths was small
(i.e. < 5 deaths) and the confidence intervals were wide. Logistic
regression modeling in the multi-plant cohort (i.e. German and U.S.
plants combined) showed an increased risk of lung cancer in the high
(OR=20.2; 95% CI: 6.2-65.4; 10 observed deaths) and intermediate
(OR=4.9; 95% CI: 1.5-16.0; 9 deaths) cumulative exposure groups when
compared to the low exposure group (Ex. 48-1-2, Table 18). The lung
cancer risks remained unchanged when smoking status was controlled for
in the model, indicating that the elevated risks were unlikely to be
confounded by smoking in this study.
OSHA does not believe that the results of the German study provide
a basis on which to establish a threshold exposure below which no lung
cancer risk exists. Like the U.S. post-change cohort (i.e., Luippold
(2005) cohort) discussed above, small cohort size, few lung cancer
cases (e.g., 10 deaths in the three lowest exposure groups combined)
and limited follow-up (average 17 years) severely limit the power to
detect small increases in risk that may be present with low cumulative
exposures. The limited power of the study is reflected in the wide
confidence intervals associated with the SMRs. For example, there is no
apparent evidence of excess lung cancer (SMR=0.95; 95% CI: 0.26-2.44)
in workers exposed to low cumulative urine chromium levels between 40-
100 [mu]g Cr/L--yr. However, the lack of precision in this estimate is
such that a two-fold increase in lung cancer mortality can not be ruled
out with a high degree of confidence. Although the study authors state
that the data suggest a possible threshold effect, they acknowledge
that "demonstrating a clear (and statistically significant) threshold
response in epidemiological studies is difficult especially [where], as
in this study, the number of available cases is relatively small, and
the precise estimation of small risks requires large numbers" (Ex. 48-
4, p. 8). OSHA agrees that the number of lung cancer cases in the study
is too small to clearly demonstrate a threshold response or precisely
estimate small risks.
OSHA has relied upon a larger, more robust cohort study for its
risk assessment than the German cohort. In comparison, the Gibb cohort
has about five times the person-years of observation (70736 vs. 14684)
and number of lung cancer cases (122 vs. 22). The workers, on average,
were followed longer (30 vs. 17 years) and a greater proportion of the
cohort is deceased (36% vs. 14%). Limited air monitoring from the
German plants indicate that average plant-wide airborne Cr(VI) roughly
declined from about 35 [mu]g Cr(VI)/m\3\ in the mid 1970s to 5 [mu]g
Cr(VI)/m\3\ in the 1990s (2002 report; Ex. 7-91). This overlaps the
Cr(VI) air levels in the Baltimore plant studied by Gibb et al. (Ex.
47-8). Furthermore, cumulative exposure estimates for members of the
Gibb cohort were individually reconstructed
from job histories and Cr(VI) air monitoring data. These airborne
Cr(VI) exposures are better suited than urinary chromium for evaluating
occupational risk at the permissible exposure limits under
consideration by OSHA. An appropriate conversion procedure that
credibly predicts time-weighted average Cr(VI) air concentrations in
the workplace from urinary chromium measurements is not evident and,
thus, would undoubtedly generate additional uncertainty in the risk
estimates. For the above reasons, OSHA believes the Gibb cohort
provides a stronger dataset than the German cohort on which to assess
the existence of a threshold exposure. This and other issues pertaining
to the relationship between the cumulative exposure and lung cancer
risk are further discussed in section VI.G.1.a.
C. Quantitative Risk Assessments Based on the Gibb Cohort
Quantitative risk assessments were performed on the exposure-
response data from the Gibb cohort by three groups: Environ
International (Exs. 33-15; 33-12) under contract with OSHA; the
National Institute for Occupational Safety and Health (Ex. 33-13); and
Exponent (Ex. 31-18-15-1) for the Chrome Coalition. All reported
similar risks for Cr(VI) exposure over a working lifetime despite using
somewhat different modeling approaches. The exposure-response data,
risk models, statistical evaluation, and risk estimates reported by
each group are discussed below.
1. Environ Risk Assessments
In 2002, Environ International (Environ) prepared a quantitative
analysis of the association between Cr(VI) exposure and lung cancer
(Ex. 33-15) , which was described in detail in the Preamble to the
Proposed Rule (69 FR at 59364-59365). After the completion of the 2002
Environ analysis, individual data for the 2357 men in the Gibb et al.
cohort became available. The new data included cumulative Cr(VI)
exposure estimates, smoking information, date of birth, race, date of
hire, date of termination, cause of death, and date of the end of
follow-up for each individual (Ex. 35-295). The individual data allowed
Environ to do quantitative risk assessments based on (1) redefined
exposure categories, (2) alternate background reference rates for lung
cancer mortality, and (3) Cox proportional hazards modeling (Ex. 33-
12). These are discussed below and in the 2003 Environ analysis (Ex.
33-12).
The 2003 Environ analysis presented two alternate groupings with
ten cumulative Cr(VI) exposure groups each, six more than reported by
Gibb et al. and used in the 2002 analysis. One alternative grouping was
designed to divide the person-years of follow-up fairly evenly across
groups. The other alternative allocated roughly the same number of
observed lung cancers to each group. These two alternatives were
designed to remedy the uneven distribution of observed and expected
cases in the Gibb et al. categories, which may have caused parameter
estimation problems due to the small number of cases in some groups.
The new groupings assigned adequate numbers of observed and expected
lung cancer cases to all groups and are presented in Table VI-1.
Environ used a five-year lag to calculate cumulative exposure for
both groupings. This means that at any point in time after exposure
began, an individual's cumulative exposure would equal the product of
chromate concentration and duration of exposure, summed over all jobs
held up to five years prior to that point in time. An exposure lag is
commonly used in exposure-response analysis for lung cancer since there
is a long latency period between first exposure and the development of
disease. Gibb et al. found that models using five- and ten-year lags
provided better fit to the mortality data than lags of zero, two and
twenty years (Ex. 31-22-11).
BILLING CODE 4510-26-P
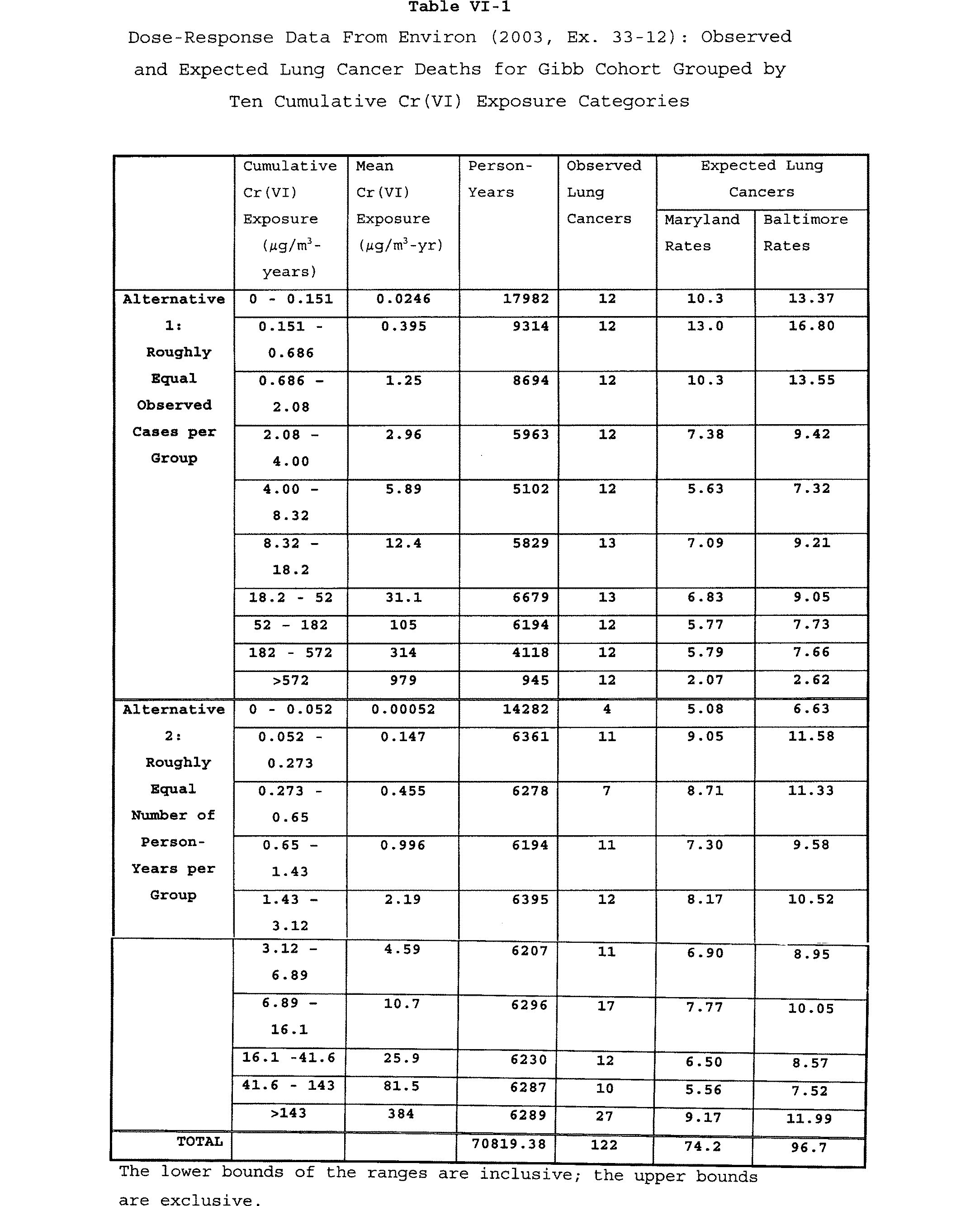
BILLING CODE 4510-26-C
The 2003 Environ analysis also derived expected cases using lung
cancer rates from alternative reference populations. In addition to the
State of Maryland lung cancer rates that were used by Gibb et al.,
Environ used age- and race-specific rates from the city of Baltimore,
where the plant was located. Baltimore may represent a more appropriate
reference population because most of the cohort members
resided in Baltimore and Baltimore residents may be more similar to the
cohort members than the Maryland or U.S. populations in their co-
exposures and lifestyle characteristics, especially smoking habits and
urban-related risk factors. On the other hand, Baltimore may not be the
more appropriate reference population if the higher lung cancer rates
in the Baltimore population primarily reflect extensive exposure to
industrial carcinogens. This could lead to underestimation of risk
attributable to Cr(VI) exposure.
The 2003 analysis used two externally standardized models, a
relative risk model (model E1 below) and an additive risk model (model
E2) defined as follows:
E1. Ni = C0 * Ei * (1 + C1Di + C2Di\2\)
E2. Ni = C0 * Ei + PYi * (C1Di + C2Di\2\)
where Ni is the predicted number of lung cancers in the i
th group; PYi is the number of person-years for
group i; Ei is the expected number of lung cancers in that
group, based on the reference population; Di is the mean
cumulative dose for that group; and C0, C1, and
C2 are parameters to be estimated. Both models initially
included quadratic exposure terms (C2Di\2\ ) as
one way to test for nonlinearity in the exposure-response. Model E1 is
a relative risk model, whereas Model E2 is an additive risk model. In
the case of additive risk models, the exposure-related estimate of
excess risk is the same regardless of the age- and race-specific
background rate of lung cancer. For relative risk models, a dose term
is multiplied by the appropriate background rate of lung cancer to
derive an exposure-related estimate of risk, so that excess risk always
depends on the background.
Maximum likelihood techniques were used to estimate the parameters
C0, C1, and C2. Likelihood ratio tests
were used to determine which of the model parameters contributed
significantly to the fit of the model. Parameters were sequentially
added to the model, starting with C1, when they contributed
significantly (p < 0.05) to improving the fit. Parameters that did not
contribute significantly, including the quadratic exposure terms
(C2Di\2\ ), were removed from the models.
Two Cox proportional hazards models were also fit to the individual
exposure-response data. The model forms were:
C1. h(t;z;D) = h0(t)*exp([beta]1z +
[beta]2D)
C2. h(t;z;D) = h0(t)*[exp([beta]1z)][1 +
[beta]2D]
where h is the hazard function, which expresses the age-specific rate
of lung cancer among workers, as estimated by the model. In addition, t
is age, z is a vector of possible explanatory variables other than
cumulative dose, D is cumulative dose, h0(t) is the baseline
hazard function (a function of age only), [beta]2 is the
cumulative dose coefficient, and [beta]1 is a vector of
coefficients for other possible explanatory variables--here, cigarette
smoking status, race, and calendar year of death (Ex. 35-57). Cox
modeling is an approach that uses the experience of the cohort to
estimate an exposure-related effect, irrespective of an external
reference population or exposure categorization. Because they are
internally standardized, Cox models can sometimes eliminate concerns
about choosing an appropriate reference population and may be
advantageous when the characteristics of the cohort under study are not
well matched against reference populations for which age-related
background rates have been tabulated. Model C1 assumes the lung cancer
response is nonlinear with cumulative Cr(VI) exposure, whereas C2
assumes a linear lung cancer response with Cr(VI) exposure. For the Cox
proportional hazards models, C1 and C2, the other possible explanatory
variables considered were cigarette smoking status, race, and calendar
year of death.
The externally standardized models E1 and E2 provided a good fit to
the data (p>=0.40). The choice of exposure grouping had little effect
on the parameter estimates of either model E1 or E2. However, the
choice of reference rates had some effect, notably on the
"background" parameter, C0, which was included as a fitted
parameter in the models to adjust for differences in background lung
cancer rates between cohort members and the reference populations. For
example, values of C0 greater than one "inflate" the base
reference rates, reducing the magnitude of excess risks in the model.
Such an adjustment was necessary for the Maryland reference population
(the maximum likelihood estimate of C0 was significantly
higher than one), but not for the Baltimore city reference population
(C0 was not significantly different from one). This result
suggests that the Maryland lung cancer rates may be lower than the
cohort's background lung cancer rates, but the Baltimore city rates may
adequately reflect the cohort background rates. The inclusion of the
C0 parameter yielded a cumulative dose coefficient that
reflected the effect of exposure and not the effect of differences in
background rates, and was appropriate.
The model results indicated a relatively consistent cumulative dose
coefficient, regardless of reference population. The coefficient for
cumulative dose in the models ranged from 2.87 to 3.48 per mg/m\3\-yr
for the relative risk model, E1, and from 0.0061 to 0.0071 per mg/m\3\-
person-yr for the additive risk model, E2. These coefficients determine
the slope of the linear cumulative Cr(VI) exposure-lung cancer response
relationship. In no case did a quadratic model fit the data better than
a linear model.
Based on comparison of the models' AIC values, Environ indicated
that the linear relative risk model E1 was preferred over the additive
risk model E2. OSHA agrees with Environ's conclusion. The relative risk
model is also preferred over an additive risk model because the
background rate of lung cancer varies with age. It may not be
appropriate to assume, as an additive model does, that increased lung
cancer risk at age 25, where background risk is relatively low, would
be the same (for the same cumulative dose) as at age 65, where
background rates are much higher.
The Cox proportional hazards models, C1 and C2, also fit the data
well (although the fit was slightly better for model C2 than C1).
Recall that for the Cox proportional hazards models, C1 and C2, the
other possible explanatory variables considered were cigarette smoking
status, race, and calendar year of death. For both models, addition of
a term for smoking status significantly improved the fit of the models
to the data (p< 0.00001). The experience with model C1 indicated that
race (p=0.15) and year of death (p=0.4) were not significant
contributors when cumulative dose and smoking status were included in
the model. Based on results for model C1, race and year of death were
not considered by Environ in the linear model C2. The cumulative dose
coefficient, [beta]2, was 1.00 for model C1 and 2.68 for
model C2. A more complete description of the models and variables can
be found in the 2003 Environ analysis (Ex. 33-12, p. 10).
Lifetable calculations were made of the number of extra lung
cancers per 1000 workers exposed to Cr(VI) based on models E1, E2, C1,
and C2, assuming a constant exposure from age 20 through a maximum of
age 65. The lifetable accounted for both lung cancer risk and competing
mortality through age 100. Rates of lung cancer and other mortality for
the lifetable calculations were based, respectively, on 2000 U.S. lung
cancer and all-cause mortality rates for both sexes and all races. In
addition to the maximum likelihood estimates, 95% confidence intervals
for the excess lifetime risk were derived. Details about the procedures
used to estimate parameters, model fit, lifetable calculations, and
confidence intervals are described in the 2003 Environ report (Ex. 33-12, p. 8-9).
BILLING CODE 4510-26-P

BILLING CODE 4510-26-C
Table VI-2 shows each model's predictions of excess lifetime lung
cancer risk from a working lifetime of exposure to various Cr(VI) air
levels. The estimates are very consistent regardless of model, exposure
grouping, or reference population. The model that appears to generate
results least similar to the others is C1, which yielded one of the
higher risk estimates at 52 [mu]g/m3, but estimated the
lowest risks for exposure levels of 10 [mu]g/m3 or lower.
The change in magnitude, relative to the other models, is a result of
the nonlinearity of this model. Confidence limits for all models,
including C1, tend to overlap, suggesting a fair degree of statistical
consistency.
2. National Institute for Occupational Safety and Health (NIOSH) Risk
Assessment
NIOSH (Ex. 33-13) developed a risk assessment from the Gibb cohort.
The NIOSH analysis, like the 2003 Environ assessment, used the cohort
individual data files to compute cumulative Cr(VI) exposure. However,
NIOSH also explored some other exposure-related assumptions. For
example, they performed the dose-response analysis with lag times in
addition to the 5-year lag used by Environ. NIOSH also analyzed dose-
response using as many as 50 exposure categories, although their report
presents data in five cumulative Cr(VI) exposure groupings.
NIOSH incorporated information on the cohort smoking behavior in
their quantitative assessments. They estimated (packs/day)-years of
cumulative smoking for each individual in the cohort, using information
from a questionnaire that was administered at the time of each cohort
member's date of hire. To estimate cumulative smoking, NIOSH assumed
that the cohort members maintained the level of smoking reported in the
questionnaire from the age of 18 through the end of follow-up.
Individuals with unknown smoking status were assigned a value equal to
the average smoking level among all individuals with known smoking
levels (presumably including non-smokers). Individuals who were known
to smoke but for whom the amount was unknown were assigned a smoking
level equal to the average of all smokers.
NIOSH considered six different relative risk models, fit to the
Gibb cohort data by Poisson regression methods. They did not consider
additive risk models. The six relative risk models were externally
standardized using age- and race-specific U.S. lung cancer rates. Their
background coefficients, C0, explicitly included smoking,
race, and age terms to adjust for differences between the cohort and
the reference population. These models are described as follows:
NIOSH1a: Ni = C0 * Ei * exp(C1Di)
NIOSH1b: Ni = C0 * Ei * exp(C1Di\1/2\\)
NIOSH1c: Ni = C0 * Ei * exp(1 + C1Di + C2Di2)
NIOSH1d: Ni = C0 * Ei * (1 + Di)[alpha]
NIOSH1e: Ni = C0 * Ei * (1 + C1Di)
NIOSH1f: Ni = C0 * Ei * (1 + C1Di[alpha])
where the form of the equation has been modified to match the format
used in the Environ reports. In addition, NIOSH fit Cox proportional
hazard models (not presented) to the lung cancer mortality data using
the individual cumulative Cr(VI) exposure estimates.
NIOSH reported that the linear relative risk model 1e generally
provided a superior fit to the exposure-response data when compared to
the various log linear models, 1a-d. Allowing some non-linearity (e.g.,
model 1f) did not significantly improve the goodness-of-fit, therefore,
they considered the linear relative risk model form 1e (analogous to
the Environ model E1) to be the most appropriate for determining their
lifetime risk calculations. A similar fit could be achieved with a log-
linear power model (model 1d) using log-transformed cumulative Cr(VI)
and a piece-wise linear specification for the cumulative smoking term.
The dose coefficient (C1) for the linear relative risk
model 1e was estimated by NIOSH to be 1.444 per [mu]g CrO3/
m3-yr (Ex. 33-13, Table 4). If the exposures were converted
to units of [mu]g Cr(VI)/m3-yr, the estimated cumulative
dose coefficient would be 2.78 (95% CI: 1.04 to 5.44) per [mu]g/
m3-yr. This value is very close to the estimates derived in
the Environ 2003 analysis (maximum likelihood estimates ranging from
2.87 to 3.48 for model E1, depending on the exposure grouping and the
reference population). Lifetime risk estimates based on the NIOSH-
estimated dose coefficient and the Environ lifetable method using 2000
U.S. rates for lung cancer and all cause mortality are shown in Table
VI-3. The values are very similar to the estimates predicted by the
Environ 2003 analysis (Table VI-3). The small difference may be due to
the NIOSH adjustment for smoking in the background coefficient. NIOSH
found that excess lifetime risks for a 45-year occupational exposure to
Cr(VI) predicted by the best-fitting power model gave very similar
risks to the preferred linear relative risk model at TWA Cr(VI)
concentrations between 0.52 and 52 [mu]g/m3 (Ex. 33-13,
Table 5). Although NIOSH did not report the results, they stated that
Cox modeling produced risk estimates similar to the Poisson regression.
The consistency between Cox and Poisson regression modeling is
discussed further in section VI.C.4.
BILLING CODE 4510-26-P

BILLING CODE 4510-26-C
NIOSH reported a significantly higher dose-response coefficient for
nonwhite workers than for white workers. That is, nonwhite workers in
the Gibb cohort are estimated to have a higher excess risk of lung
cancer than white workers, given equal cumulative exposure to Cr(VI).
In contrast, no significant race difference was found in the Cox
proportional hazards analysis reported by 2003 Environ.
3. Exponent Risk Assessment
In response to OSHA's Request For Information, Exponent prepared an
analysis of lung cancer mortality from the Gibb cohort. Like the 2003
Environ and NIOSH analyses, the Exponent analysis relied on the
individual worker data. Exponent performed their dose-response analyses
based on three different sets of exposure categories using two
reference populations and 70,808 person-years of follow-up. A total of
four analyses were completed, using (1) Maryland reference rates and
the four Gibb et al. exposure categories; (2) Baltimore reference rates
and the four Gibb et al. exposure categories; (3) Baltimore reference
rates and six exposure groups defined by Exponent; and (4) Baltimore
City reference rates and five exposure categories, obtained by removing
the highest of the six groups defined by Exponent from the dose-
response analysis. A linear relative risk model without a background
correction term (the term C0 used by Environ and NIOSH) was
applied in all of these cases and cumulative exposures were lagged five
years (as done by Environ and NIOSH). The analyses showed excess
lifetime risk between 6 and 14 per 1000 for workers exposed to 1 [mu]g/
m\3\ Cr(VI) for 45 years.
The analysis using Maryland reference lung cancer rates and the
Gibb et al. four-category exposure grouping yielded an excess lifetime
risk of 14 per 1000. This risk, which is higher than the excess
lifetime risk estimates by Environ and NIOSH for the same occupational
exposure, probably results from the absence of a background rate
coefficient (C0) in Exponent's model. As reported in the
Environ 2002 and 2003 analyses, the Maryland reference lung cancer
rates require a background rate coefficient greater than 1 to achieve
the best fit to the exposure-response data. The unadjusted Maryland
rates probably underestimate the cohort's background lung cancer rate,
leading to overestimation of the risk attributable to cumulative Cr(VI)
exposure.
The two analyses that used Baltimore reference rates and either
Exponent's six-category exposure grouping or the Gibb et al. four-
category grouping both resulted in an excess lifetime unit risk of 9
per 1000 for workers exposed to 1 [mu]g/m\3\ Cr(VI) for 45 years (Ex.
31-18-15-1, p. 41). This risk is close to estimates reported by Environ
using their relative risk model (E1) and Baltimore reference rates for
the same occupational exposure (Table VI-2). The Environ analysis
showed that, unlike the Maryland-standardized model discussed above,
the Baltimore-standardized models had background rate coefficients very
close to 1, the "default" value assumed by the Exponent relative risk
model. This suggests that the Baltimore reference rates may represent
the background lung cancer rate for this cohort more accurately than
the Maryland reference rates.
The lowest excess lifetime unit risk for workers exposed to 1
[mu]g/m\3\ Cr(VI) for 45 years reported by Exponent, at 6 per 1000, was
derived from the analysis that excluded the highest of Exponent's six
exposure groups. While this risk value is close to the Environ and
NIOSH unit risk estimates, the analysis merits some concern. Exponent
eliminated the highest exposure group on the basis that most cumulative
exposures in this group were higher than exposures usually found in
current workplace conditions. However, eliminating this group could
exclude possible long-term exposures (e.g., >15 years) below the
previous OSHA PEL (52 [mu]g/m\3\ ) from the risk analysis. Moreover, no
matter what current exposures might be, data on higher cumulative
exposures are relevant for understanding the dose-response
relationships.
In addition, the Exponent six category cumulative exposure grouping
may have led to an underestimate of the dose effect. The definition of
Exponent's six exposure groups was not related to the distribution of
cumulative exposure associated with individual person-years, but rather
to the distribution of cumulative exposure among the workers at the end
of their employment. This division does not result in either a uniform
distribution of person-years or observed lung cancer cases among
exposure categories. In fact, the six category exposure groupings of
both person-years and observed lung cancers were very uneven, with a
preponderance of both allocated to the lowest exposure group. This
skewed distribution of person-years and observed cases puts most of the
power for detecting significant differences from background cancer
rates at low exposure levels, where these differences are expected to
be small, and reduces the power to detect any significant differences
from background at higher exposure concentrations.
4. Summary of Risk Assessments Based on the Gibb Cohort
OSHA finds remarkable consistency among the risk estimates from the
various quantitative analyses of the Gibb cohort. Both Environ and
NIOSH determined that linear relative risk models generally provided a
superior fit to the data when compared to other relative risk models,
although the confidence intervals in the non-linear Cox model reported
by Environ overlapped with the confidence intervals in their linear
models. The Environ 2003 analysis further suggested that a linear
additive risk model could adequately describe the observed dose-
response data. The risk estimates for NIOSH and Environ's best-fitting
models were statistically consistent (compare Tables VI-2 and VI-3).
The choice of reference population had little impact on the risk
estimates. NIOSH used the entire U.S. population as the reference, but
included adjustment terms for smoking, age and race in its models. The
Environ 2003 analysis used both Maryland and Baltimore reference lung
cancer rates, and included a generic background coefficient
C0 to adjust for potential differences in background risk
between the reference population and the worker cohort. This term was
significant in the fitted model when Maryland rates were used for
external standardization, but not when Baltimore rates were used. Since
no adjustment in the model background term was required to better fit
the exposure-response data using Baltimore City lung cancer rates, they
may best represent the cohort's true background lung cancer incidence.
OSHA considers the inclusion of such adjustment factors, whether
specific to smoking, race, and age (as defined by NIOSH), or generic
(as defined by Environ), to be appropriate and believes they contribute
to accurate risk estimation by helping to correct for confounding risk
factors. The Cox proportional hazard models, especially the linear Cox
model, yielded risk estimates that were generally consistent with the
externally standardized models.
Finally, the number of exposure categories used in the analysis had
little impact on the risk estimates. When an appropriate adjustment to
the background rates was included, the four exposure groups originally
defined by Gibb et al. and analyzed in the 2002 Environ report, the six
exposure groups defined by Exponent, the two alternate sets of ten
exposure categories as defined in the 2003 Environ analysis, and the
fifty groups defined and aggregated by NIOSH all gave essentially the
same risk estimates. The robustness of the results to various
categorizations of cumulative exposure adds credence to the risk
projections.
Having reviewed the analyses described in this section, OSHA finds
that the best estimates of excess lung cancer risk to workers exposed
to the previous PEL (52 [mu]g Cr(VI)/m3) for a
working lifetime are about 300 to 400 per thousand based on data from
the Gibb cohort. The best estimates of excess lung cancer risks to
workers exposed to other TWA exposure concentrations are presented in
Table VI-2. These estimates are consistent with predictions from
Environ, NIOSH and Exponent models that applied linear relative and
additive risk models based on the full range of cumulative Cr(VI)
exposures experienced by the Gibb cohort and used appropriate
adjustment terms for the background lung cancer mortality rates.
D. Quantitative Risk Assessments Based on the Luippold Cohort
As discussed earlier, Luippold et al. (Exs. 35-204; 33-10) provided
information about the cohort of workers employed in a chromate
production plant in Painesville, Ohio. Follow-up for the 482 members of
the Luippold cohort started in 1940 and lasted through 1997, with
accumulation of person-years for any individual starting one year after
the beginning of his first exposure. There were 14,048 total person-
years of follow-up for the cohort. The person-years were then divided
into five exposure groups that had approximately equal numbers of
expected lung cancers in each group. Ohio reference rates were used to
compute expected numbers of deaths. White male rates were used because
the number of women was small (4 out of 482) and race was known to be
white for 241 of 257 members of the cohort who died and for whom death
certificates were available. The 1960-64 Ohio rates (the earliest
available) were assumed to hold for the time period from 1940 to 1960.
Rates from 1990-94 were assumed to hold for the period after 1994. For
years between 1960 and 1990, rates from the corresponding five-year
summary were used. There were significant trends for lung cancer SMR as
a function of year of hire, duration of employment, and cumulative
Cr(VI) exposure. The cohort had a significantly increased SMR for lung
cancer deaths of 241 (95% C.I. 180 to 317).

Environ conducted a risk assessment based on the cumulative Cr(VI)
exposure-lung cancer mortality data from Luippold et al. and presented
in Table VI-4 (Ex. 33-15). Cumulative Cr(VI) exposures were categorized
into five groups with about four expected lung cancer deaths in each
group. In the absence of information to the contrary, Environ assumed
Luippold et al. did not employ any lag time in determining the
cumulative exposures. The calculated
and expected numbers of lung cancers were derived from Ohio reference
rates. Environ applied the relative and additive risk models, E1 and
E2, to the data in Table VI-4.
Linear relative and additive risk models fit the Luippold cohort
data adequately (p>=0.25). The final models did not include the
quadratic exposure coefficient, C2, or the background rate
parameter, C0, as they did not significantly improve the fit
of the models. The maximum likelihood estimates for the Cr(VI)
exposure-related parameter, C1, of the linear relative and
additive risk models were 0.88 per mg/m3-yr and 0.0014 per
mg/m3-person-yr, respectively. The C1 estimates
based on the Luippold cohort data were about 2.5-fold lower than the
parameter estimates based on the Gibb cohort data. The excess lifetime
risk estimate calculated by Environ for a 45-year working-lifetime
exposure to 1 [mu]g Cr(VI)/m3 (e.g., the unit risk) for both
models was 2.2 per 1000 workers (95% confidence intervals from 1.3 to
3.5 per 1000 for the relative risk model and 1.2 to 3.4 per 1000 for
the additive risk model) using a lifetable analysis with 1998 U.S.
mortality reference rates. These risks were 2.5 to 3-fold lower than
the projected unit risks based on the Gibb data set for equivalent
cumulative Cr(VI) exposures.
Crump et al. (Exs. 33-15; 35-58; 31-18) also performed an exposure-
response analysis from the Painesville data. In a Poisson regression
analysis, cumulative exposures were grouped into ten exposure
categories with approximately two expected lung cancer deaths in each
group. The observed and expected lung cancer deaths by Cr(VI) exposure
category are shown in Table VI-5. Ohio reference rates were used in
calculating the expected lung cancer deaths and cumulative exposures
were lagged five years.

The Crump et al. analysis used the same linear relative risk and
additive risk models as Environ on the individual data categorized into
the ten cumulative exposure groups (Ex. 35-58). Tests for systematic
departure from
linearity were non-significant for both models (p>=0.11). The
cumulative dose coefficient determined by the maximum likelihood method
was 0.79 (95% CI: 0.47 to 1.19) per mg/m3-yr for the
relative risk model and 0.0016 (95% CI: 0.00098 to 0.0024) per mg/
m3-person-yr for the additive risk model, respectively. The
authors noted that application of the linear models to five and seven
exposure groups resulted in no significant difference in dose
coefficients, although the results were not presented. The exposure
coefficients reported by Crump et al. were very similar to those
obtained by Environ above, although different exposure groups were used
and Crump et al. used a five-year lag for the cumulative exposure
calculation. The authors noted that the linear models did not fit the
exposure data grouped into ten categories very well (goodness-of-fit
p< =0.01) but fit the data much better with seven exposure groups
(p>0.3), replacing the many lower exposure categories where there were
few observed and expected cancers with more stable exposure groupings
with greater numbers of cancers. The reduction in number of exposure
groups did not substantially change the fitted exposure coefficients.
The maximum likelihood estimate for the cumulative exposure
coefficient using the linear Cox regression model C2 was 0.66 (90% CI:
0.11 to 1.21), which was similar to the linear [Poisson regression]
relative risk model. When the Cox analysis was restricted to the 197
workers with known smoking status and a smoking variable in the model,
the dose coefficient for Cr(VI) was nearly identical to the estimate
without controlling for smoking. This led the authors to conclude that
"the available smoking data did not suggest that exposure to Cr(VI)
was confounded with smoking in this cohort, or that failure to control
for smoking had an appreciable effect upon the estimated carcinogenic
potency of Cr(VI)" (Ex. 35-58, p. 1156).
Given the similarity in results, OSHA believes it is reasonable to
use the exposure coefficients reported by Crump et al. based on their
groupings of the individual cumulative exposure data to estimate excess
lifetime risk from the Luippold cohort. Table VI-6 presents the excess
risk for a working lifetime exposure to various TWA Cr(VI) levels as
predicted by Crump et al.'s relative and additive risk models using a
lifetable analysis with 2000 U.S. rates for all causes and lung cancer
mortality. The resulting maximum likelihood estimates indicate that
working lifetime exposures to the previous Cr(VI) PEL would result in
excess lifetime lung cancer risks around 100 per 1000 (95% C.I. approx.
60-150). The risk estimates based on the Luippold cohort are lower than
the risk estimates based on the Gibb cohort, as discussed further in
section VI.F.
BILLING CODE 4510-26-P
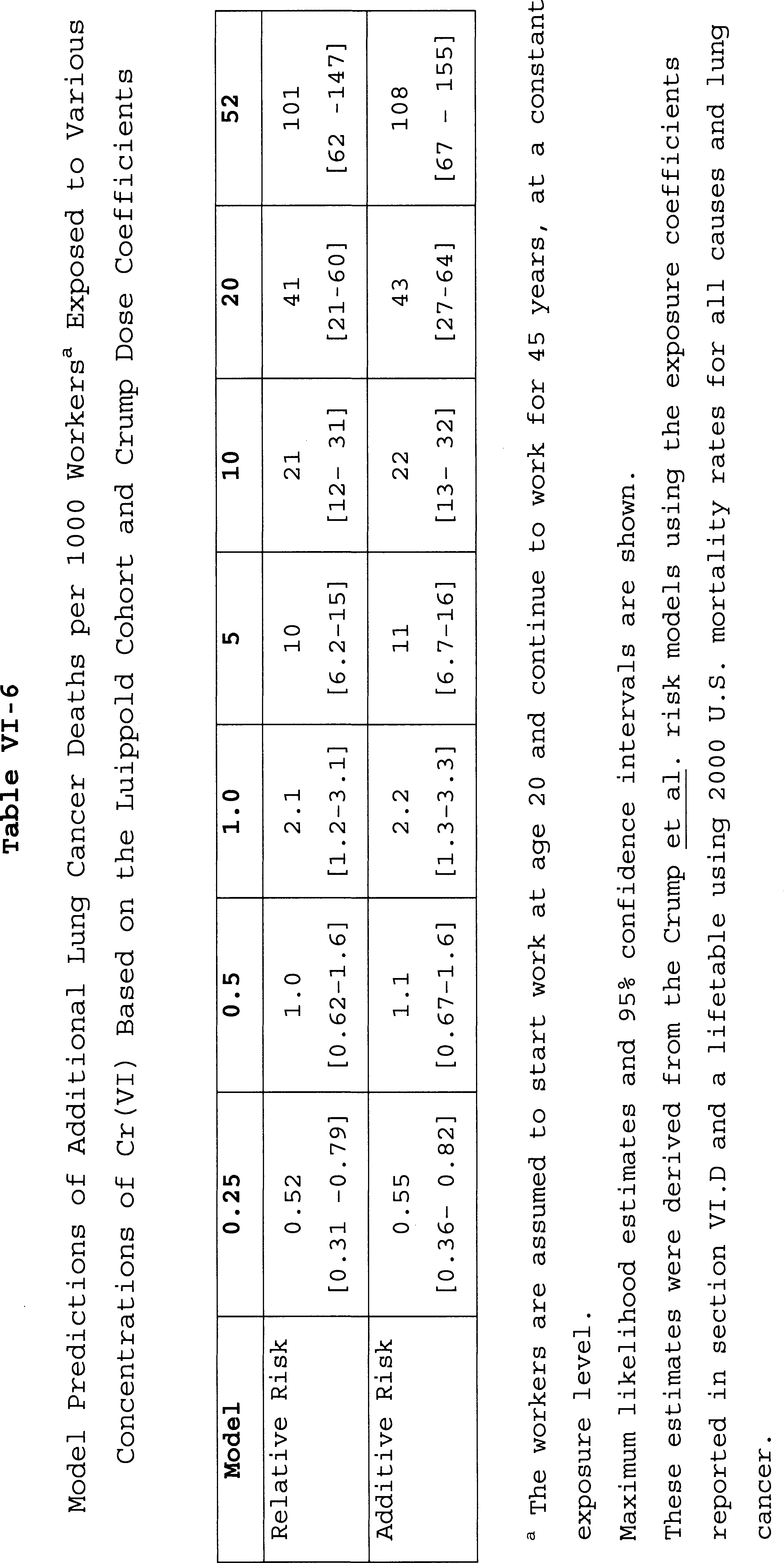
BILLING CODE 4510-26-C
E. Quantitative Risk Assessments Based on the Mancuso, Hayes, Gerin,
and Alexander Cohorts
In addition to the preferred data sets analyzed above, there are
four other cohorts with available data sets for estimation of
additional lifetime risk of lung cancer. These are the Mancuso cohort,
the Hayes cohort, the Gerin cohort, and the Alexander cohort. Environ
did exposure-response analysis for all but the Hayes cohort (Ex. 33-
15). Several years earlier, the K.S. Crump Division did quantitative
assessments on data from the Mancuso and Hayes cohort, under contract
with OSHA (Ex.13-5). The U.S. EPA developed quantitative risk
assessments from the Mancuso cohort data for its Integrated Risk
Information System (Exs. 19-1; 35-52). The California EPA (Ex. 35-54),
Public Citizen Health Research Group (Ex. 1), and the U.S. Air Force
Armstrong Laboratory (AFAL) for the Department of Defense (Ex. 35-51)
performed assessments from the Mancuso data using the 1984 U.S. EPA
risk estimates as their starting point. The U.S. EPA also published a
risk assessment based on the Hayes cohort data (Ex. 7-102). Until the
cohort studies of Gibb et al. and Luippold et al. became available,
these earlier assessments provided the most current projected cancer
risks from airborne exposure to Cr(VI). The previous risk assessments
were extensively described in the NPRM sections VI.E.1 and VI.E.2 (69
FR at 59375-59378). While the risk estimates from Mancuso, Hayes,
Gerin, and Alexander data sets are associated with a greater degree of
uncertainty, it is nevertheless valuable to compare them to the risk
estimates from the higher quality Gibb and Luippold data sets in order
to determine if serious discrepancies exist between them. OSHA believes
evaluating consistency in risk among several worker cohorts adds to the
overall quality of the assessment.
The Mancuso and Luippold cohorts each worked at the Painesville
plant but the worker populations did not overlap due to different
selection criteria. Exposure estimates were also based on different
industrial hygiene surveys. The Hayes and Gibb cohorts both worked at
the Baltimore plant. Even though Cr(VI) exposures were reconstructed
from monitoring data measured at different facilities resulting in
significantly different exposure-response functions (see section VI.F),
there was some overlap in the two study populations. As a result, the
projected risks from these data sets can not strictly be viewed as
independent estimates. The Gerin and Alexander cohorts were not
chromate production workers and are completely independent from the
Gibb and Luippold data sets. The quantitative assessment of the four
data sets and comparison with the risk assessments based on the Gibb
and Luippold cohorts are discussed below.
1. Mancuso Cohort
As described in subsection VII.B.3, the Mancuso cohort was
initially defined in 1975 and updated in 1997. The cohort members were
hired between 1931 and 1937 and worked at the same Painesville facility
as the Luippold cohort workers. However, there was no overlap between
the two cohorts since all Luippold cohort workers were hired after
1939. The quantitative risk assessment by Environ used data reported in
the 1997 update (Ex. 23, Table XII) in which lung cancer deaths and
person-years of follow-up were classified into four groups of
cumulative exposure to soluble chromium, assumed to represent Cr(VI)
(Ex. 33-15). The mortality data and person-years were further broken
down by age of death in five year increments starting with age interval
40 to 44 years and going up to >75 years. No expected numbers of lung
cancers were computed, either for the cohort as a whole or for specific
groups of person-years. Environ applied an indirect method based on the
recorded median age and year of entry into the cohort to estimate age
information necessary to derive expected numbers of age- and calendar
year-adjusted lung cancers deaths required to complete the risk
assessment.
Observed and expected lung cancer deaths by age and cumulative
exposure (mg/m\3\-yr) are presented in Table 3 of the 2002 Environ
report (Ex. 33-15, p. 39). The mean cumulative exposures to soluble
Cr(VI) were assumed to be equal to the midpoints of the tabulated
ranges. No lag was used for calculating the cumulative exposures.
Environ applied externally standardized risk models to these data,
similar to those described in section VI.C.1 but using an age-related
parameter, as discussed in the 2002 report (Ex. 33-15, p. 39). The
externally-standardized linear relative risk model with an age-
dependent exposure term provided a superior fit over the other models.
The predicted excess risk of lung cancer from a 45-year working
lifetime of exposure to Cr(VI) at the previous OSHA PEL using the best-
fitting linear relative risk model is 293 per 1000 workers (95% C.I.
188 to 403). The maximum likelihood estimate from working lifetime
exposure to new PEL of 5.0 [mu]g/m\3\ Cr(VI) is 34 per 1000 workers
(95% C.I. 20 to 52 per 1000). These estimates are close to those
predicted from the Gibb cohort but are higher than predicted from the
Luippold cohort.
There are uncertainties associated with both the exposure estimates
and the estimates of expected numbers of lung cancer deaths for the
1997 Mancuso data set. The estimates of exposure were derived from a
single set of measurements obtained in 1949 (Ex. 7-98). Although little
prior air monitoring data were available, it is thought that the 1949
air levels probably understate the Cr(VI) concentrations in the plant
during some of the 1930s and much of the 1940s when chromate production
was high to support the war. The sampling methodology used by Bourne
and Yee only measured soluble Cr(VI), but it is believed that the
chromate production process employed at the Painesville plant in these
early years yielded slightly soluble and insoluble Cr(VI) compounds
that would not be fully accounted for in the sampling results (Ex. 35-
61). This would imply that risks would be overestimated by use of
concentration estimates that were biased low. However, it is possible
that the 1949 measurements did not underestimate the Cr(VI) air levels
in the early 1930s prior to the high production years. Some older
cohort members were also undoubtedly exposed to less Cr(VI) in the
1950s than measured in 1949 survey.
Another uncertainty in the risk assessment for the Mancuso cohort
is associated with the post-hoc estimation of expected numbers of lung
cancer deaths. The expected lung cancers were derived based on
approximate summaries of the ages and assumed start times of the cohort
members. Several assumptions were dictated by reliance on the published
groupings of results (e.g., ages at entry, calendar year of entry, age
at end of follow-up, etc.) as well as by the particular choices for
reference mortality rates (e.g., U.S. rates, in particular years close
to the approximated time at which the person-years were accrued). Since
the validity of these assumptions could not be tested, the estimates of
expected numbers of lung cancer deaths are uncertain.
There is also a potential healthy worker survivor effect in the
Mancuso cohort. The cohort was identified as workers first hired in the
1930s based on employment records surveyed in the late 1940s (Ex. 2-
16). The historical company files in this time period were
believed to be sparse and more likely to only identify employees still
working at the plant in the 1940s (Ex. 33-10). If there was a sizable
number of unidentified short-term workers who were hired but left the
plant in the 1930s or who died before 1940 (i.e. prior to systematic
death registration), then there may have been a selection bias (i.e.,
healthy worker survivor effect) toward longer-term, healthier
individuals (Ex. 35-60). Since the mortality of these long-term
"survivors" is often more strongly represented in the higher
cumulative exposures, it can negatively confound the exposure-response
and lead to an underestimation of risk, particularly to shorter-term
workers (Ex. 35-63). This may be an issue with the Mancuso cohort,
although the magnitude of the potential underestimation is unclear.
Earlier quantitative risk assessments by the K.S. Crump Division,
EPA, and others were done on cohort data presented in the 1975 Mancuso
report (Ex. 7-11). These assessments did not have access to the 20
additional years of follow-up nor did they have age-grouped lung cancer
mortality stratified by cumulative soluble chromium (presumed Cr(VI))
exposure), which was presented later in the 1997 update. Instead, age-
grouped lung cancer mortality was stratified by cumulative exposure to
total chromium that included not only carcinogenic Cr(VI) but
substantial amounts of non-carcinogenic Cr(III). OSHA believes that the
Environ quantitative risk assessment is the most credible analysis from
the Mancuso cohort. It relied on the updated cohort mortality data and
cumulative exposure estimates derived directly from air measurements of
soluble chromium.
2. Hayes Cohort
The K.S. Crump Division (Ex. 13-5) assessed risk based on the
exposure-response data reported in Table IV by Braver et al. (Ex. 7-17)
for the cohort studied by Hayes et al. (Ex. 7-14). The Hayes cohort
overlapped with the Gibb cohort. The Hayes cohort included 734 members,
not part of the Gibb cohort, who worked at an older facility from 1945
to 1950 but did not work at the newer production facility built in
August 1950. The Hayes cohort excluded 990 members of the Gibb cohort
who worked less than 90 days in the new production facility after
August 1950. As noted in section VI.B.4, Braver et al. derived a single
cumulative soluble Cr(VI) exposure estimate for each of four subcohorts
of chromate production workers categorized by duration of employment
and year of hire by Hayes et al. Thus, exposures were not determined
for individual workers using a more comprehensive job exposure matrix
procedure, as was done for the Gibb and Luippold cohorts. In addition,
the exposures were estimated from air monitoring conducted only during
the first five of the fifteen years the plant was in operation. Unlike
the Mancuso cohort, Hayes et al. did not stratify the observed lung
cancer deaths by age group. The expected number of lung cancer deaths
for each subcohort was based on the mortality statistics from
Baltimore.
The K.S. Crump Division applied the externally standardized linear
relative risk approach to fit the exposure-response data (Ex. 13-5).
The maximum likelihood estimate for the dose coefficient (e.g.,
projected linear slope of the Cr(VI) exposure-response curve) was 0.75
per mg Cr(VI)/m\3\-yr with a 90% confidence bound of between 0.45 and
1.1 per mg Cr(VI)/m\3\-yr. These confidence bounds are consistent with
the dose coefficient estimate obtained from modeling the Luippold
cohort data (0.83, 95% CI: 0.55 to 1.2) but lower than that from the
Gibb cohort data (3.5, 95% CI: 1.5 to 6.0). The linear relative risk
model fit the Hayes cohort data well (p=0.50). The K.S. Crump Division
predicted the excess risk from occupational exposure to Cr(VI) for a 45
year working lifetime at the previous OSHA PEL (52 [mu]g/m\3\) to be 88
lung cancer cases per 1000 workers (95% CI: 61 to 141). Predicted
excess risk at the new PEL of 5 [mu]g/m\3\ is about 9 excess lung
cancer deaths per 1000 (95% CI: 6.1 to 16) for the same duration of
occupational exposure. These estimates are somewhat lower than the
corresponding estimates based on the Gibb cohort data, probably because
of the rather high average soluble Cr(VI) level (218 [mu]g/m\3\)
assumed by Braver et al. for plant workers throughout the 1950s. If
these assumed air levels led to an overestimate of worker exposure, the
resulting risks would be underestimated.
3. Gerin Cohort
Environ (Ex. 33-15) did a quantitative assessment of the observed
and expected lung cancer deaths in stainless steel welders classified
into four cumulative Cr(VI) exposure groups reported in Tables 2 and 3
of Gerin et al. (Ex. 7-120). The lung cancer data came from a large
combined multi-center welding study in which a statistically
significant excess lung cancer risk was observed for the whole cohort
and non-statistically significant elevated lung cancer mortality was
found for the stainless steel welder subcohorts (Ex. 7-114). A positive
relationship with time since first exposure was also observed for the
stainless steel welders (the type of welding with the highest exposure
to Cr(VI)) but not with duration of employment.
The exposure-response data from the Gerin study was only presented
for those stainless steel welders with at least five years employment.
Workers were divided into "ever stainless steel welders" and
"predominantly stainless steel welders" groups. The latter group were
persons known to have had extended time welding stainless steel only or
to have been employed by a company that predominantly worked stainless
steel. As stated in section VI.B.5, the cumulative exposure estimates
were not based on Cr(VI) air levels specifically measured in the cohort
workers, and therefore are subject to greater uncertainty than exposure
estimates from the chromate production cohort studies. Environ
restricted their analysis to the "ever stainless steel welders" since
that subcohort had the greater number of eligible subjects and person-
years of follow-up, especially in the important lower cumulative
exposure ranges. The person-years, observed numbers of lung cancers,
and expected numbers of lung cancers were computed starting 20 years
after the start of employment. Gerin et al. provided exposure-response
data on welders with individual work histories (about two-thirds of the
workers) as well as the entire subcohort. Regardless of the subcohort
examined, there was no obvious indication of a Cr(VI) exposure-related
effect on lung cancer mortality. A plausible explanation for this
apparent lack of exposure-response is the potentially severe exposure
misclassification resulting from the use of exposure estimates based on
the welding literature (rather than exposure measurements at the plants
used in the study, which were not available to the authors).
Environ used externally standardized models to fit the data (Ex.
33-15). They assumed that the cumulative Cr(VI) exposure for the
workers was at the midpoint of the reported range. A value of 2.5 mg/
m\3\-yr was assumed for the highest exposure group (e.g., >0.5 mg/m\3\-
yr), since Gerin et al. cited it as the mean value for the group, which
they noted to also include the "predominantly stainless steel
welders". All models fit the data adequately (p>0.28) with exposure
coefficients considerably lower than for the Gibb or Luippold cohorts
(Ex. 33-15, Table 6). In fact, the 95% confidence intervals for the
exposure coefficients overlapped 0, which would be expected when there
is no exposure-related trend.
Based on the best fitting model, a linear relative risk model (Ex.
33-15, Table 9, p. 44), the projected excess risk of lung cancer from a
working lifetime exposure to Cr(VI) at the previous PEL was 46 (95% CI:
0 to 130) cases per 1000 workers. The 95 percent confidence interval
around the maximum likelihood estimate reflects the statistical
uncertainty associated with risk estimates from the Gerin cohort.
Following the publication of the proposed rule, OSHA received
comments from Exponent (on behalf of a group of steel industry
representatives) stating that it is not appropriate to model exposure-
response for this cohort because there was not a statistically
significant trend in lung cancer risk with estimated exposure, and risk
of lung cancer did not increase monotonically with estimated exposure
(Ex. 38-233-4, pp. 7-8). OSHA disagrees. Because the best-fitting model
tested by Environ fit the Gerin data adequately, OSHA believes that it
is reasonable to generate risk estimates based on this model for
comparison with the risk estimates based on the Gibb and Luippold
cohorts. This allows OSHA to quantitatively assess the consistency
between its preferred estimates and risk estimates derived from the
Gerin cohort.
In post-hearing comments, Dr. Herman Gibb expressed support for
OSHA's approach. Dr. Gibb stated:
The epidemiologic studies of welders * * * conducted to date
have been limited in their ability to evaluate a lung cancer risk.
It is conceivable that differences in exposure * * * between [this
industry] and the chromate production industry could lead to
differences in cancer risk. Because there aren't adequate data with
which to evaluate these differences, it is appropriate to compare
the upper bounds [on risk] derived from the Gerin et al. * * *
[study] with those predicted from the chromate production workers to
determine if they are consistent.
OSHA agrees with Exponent that the results of the Gerin et al.
study were different from those of the Luippold (2003) and Gibb
cohorts, in that a statistically significant exposure-response
relationship and a monotonically increasing lung cancer risk with
exposure were not found in Gerin. Also, the maximum likelihood risk
estimates based on the Gerin cohort were somewhat lower than those
based on the Gibb and Luippold cohorts. However, OSHA believes the
lower risk estimates from the Gerin cohort may be explained by the
strong potential for bias due to Cr(VI) exposure misclassification and
possibly by the presence of co-exposures, as discussed in sections
VI.B.5 and VI.G.4. Part of the difference may also relate to
statistical uncertainty; note that the 95% confidence intervals (shown
in Table VI-7) overlap the lower end of OSHA's range based on the
preferred Gibb and Luippold (2003) studies.
4. Alexander Cohort
Environ (Ex. 33-15) did a quantitative assessment of the observed
and expected lung cancer incidence among aerospace workers exposed to
Cr(VI) classified into four cumulative chromate exposure groups,
reported in Table 4 of Alexander et al. (Ex. 31-16-3). The authors
stated that they derived "estimates of exposure to chromium [VI]"
based on the TWA measurements, but later on referred to "the index of
cumulative total chromate exposure (italics added) reported as [mu]g/
m\3\ chromate TWA-years" (Ex. 31-16-3, p. 1254). Alexander et al.
grouped the lung cancer data by cumulative exposure with and without a
ten year lag period. They found no statistically significant elevation
in lung cancer incidence among the chromate-exposed workers or clear
trend with cumulative chromate exposure.
For their analysis, Environ assumed that the cumulative exposures
were expressed in [mu]g/m\3\-yr of Cr(VI), rather than chromate
(CrO4-2) or chromic acid (CrO3).
Environ used an externally standardized linear relative risk model to
fit the unlagged data (Ex. 33-15). An additive risk model could not be
applied because person-years of observation were not reported by
Alexander et al. Environ assumed that workers were exposed to a
cumulative Cr(VI) exposure at the midpoint of the reported ranges. For
the open-ended high exposure category, Environ assumed a cumulative
exposure 1.5 times greater than the lower limit of 0.18 mg/m\3\-yr. The
model fit the data poorly (p=0.04) and the exposure coefficient was
considered to be 0 since positive values did not significantly improve
the fit. Given the lack of a positive trend between lung cancer
incidence and cumulative Cr(VI) exposure for this cohort, these results
are not surprising.
Following the publication of the proposed rule, OSHA received
comments from Exponent (on behalf of the Aerospace Industries
Association) stating that the Agency should not apply a linear model to
the Alexander et al. study to derive risk estimates for comparison with
the estimates based on the Gibb and Luippold (2003) cohorts (Ex. 38-
215-2, p. 10). Due to the poor fit of Environ's exposure-response model
to the Alexander cohort data, OSHA agrees with Exponent in this matter.
Risk estimates based on Alexander et al. are therefore not presented in
this risk assessment.
OSHA believes that there are several possible reasons for the lack
of a positive association between Cr(VI) exposure and lung cancer
incidence in this cohort. First, follow-up time was extremely short,
averaging 8.9 years per cohort member. Long-term follow-up of cohort
members is particularly important for determining the risk of lung
cancer, which typically has an extended latency period of roughly 20
years or more. One would not necessarily expect to see excess lung
cancer or an exposure-response relationship among workers who had been
followed less than 20 years since their first exposure to Cr(VI), as
most exposure-related cancers would not yet have appeared. Other
possible reasons that an exposure-response relationship was not
observed in the Alexander cohort include the young age of the cohort
members (median 42 years at end of follow-up), which also suggests that
occupational lung cancers may not yet have appeared among many cohort
members. The estimation of cumulative Cr(VI) exposure was also
problematic, drawing on air measurement data that did not span the
entire employment period of the cohort (there were no data for 1940 to
1974) and were heavily grouped into a relatively small number of
"summary" TWA concentrations that did not capture individual
differences in workplace exposures to Cr(VI).
F. Summary of Risk Estimates Based on Gibb, Luippold, and Additional
Cohorts
OSHA believes that the best estimates of excess lifetime lung
cancer risks are derived from the Gibb and Luippold cohorts. Due to
their large size and long follow-up, these two cohorts accumulated a
substantial number of lung cancer deaths that were extensively examined
by several different analyses using a variety of statistical
approaches. Cohort exposures were reconstructed from air measurements
and job histories over three or four decades. The linear relative risk
model fit the Gibb and Luippold data sets well. It adequately fit
several epidemiological data sets used for comparative analysis.
Environ and NIOSH explored a variety of nonlinear dose-response forms,
but none provided a statistically significant improvement over the
linear relative risk model.
The maximum likelihood estimates from a linear relative risk model
fit to the Gibb data are three- to five-fold higher than estimates
based on the Luippold data at equivalent cumulative
Cr(VI) exposures and the confidence limits around the projected risks
from the two data sets do not overlap. This indicates that the maximum
likelihood estimates derived from one data set are unlikely to describe
the lung cancer mortality observed in the other data set. Despite this
statistical inconsistency between the risk estimates, the differences
between them are not unreasonably great given the potential
uncertainties involved in estimating cancer risk from the data (see
section VI.G). Since the analyses based on these two cohorts are each
of high quality and their projected risks are reasonably close (well
within an order of magnitude), OSHA believes the excess lifetime risk
of lung cancer from occupational exposure to Cr(VI) is best represented
by the range of risks that lie between maximum likelihood estimates of
the Gibb and Luippold data sets.


BILLING CODE 4510-26-C
OSHA's best estimates of excess lung cancer cases from a 45-year
working lifetime exposure to Cr(VI) are presented in Table VI-7. As
previously discussed, several acceptable assessments of the Gibb data
set were performed, with similar results. The 2003 Environ model E1,
applying the Baltimore City reference population and ten exposure
categories based on a roughly equal number of person-years per group,
was selected to represent the range of best risk estimates derived from
the Gibb cohort, in part because this assessment employed an approach
most consistent with the exposure grouping applied in the Luippold
analysis (see Table VI-6). To characterize the statistical uncertainty
of OSHA's risk estimates, Table VI-7 also presents the 95% confidence
limits associated with the maximum likelihood risk estimates from the
Gibb cohort and the Luippold cohort.
OSHA finds that the most likely lifetime excess risk at the
previous PEL of 52 [mu]g/m\3\ Cr(VI) lies between 101 per 1000 and 351
per 1000, as shown in Table VI-7. That is, OSHA predicts that between
101 and 351 of 1000 workers occupationally exposed for 45 years at the
previous PEL would develop lung cancer as a result of their exposure.
The wider range of 62 per 1000 (lower 95% confidence bound, Luippold
cohort) to 493 per 1000 (upper 95% confidence bound, Gibb cohort)
illustrates the range of risks considered statistically plausible based
on these cohorts, and thus represents the statistical uncertainty in
the estimates of lung cancer risk. This range of risks decreases
roughly proportionally with exposure, as illustrated by the risk
estimates shown in Table VI-7 for working lifetime exposures at various
levels at and below the previous PEL.
The risk estimates for the Mancuso, Hayes, and Gerin data sets are
also presented in Table VI-7. (As discussed previously, risk estimates
were not derived from the Alexander data set.) The exposure-response
data from these cohorts are not as strong as those from the two
featured cohorts. OSHA believes that the supplemental assessments for
the Mancuso and Hayes cohorts support the range of projected excess
lung cancer risks from the Gibb and Luippold cohorts. This is
illustrated by the maximum likelihood estimates and 95% confidence
intervals shown in Table VI-7. The risk estimates and 95% confidence
interval based on the Hayes cohort are similar to those based on the
Luippold cohort, while the estimates based on the Mancuso cohort are
more similar to those based on the Gibb cohort. Also, OSHA's range of
best risk estimates based on the two primary cohorts for a given
occupational Cr(VI) exposure overlap the 95 percent confidence limits
for the Mancuso, Hayes, and Gerin cohorts. This indicates that the
Agency's range of best estimates is statistically consistent with the
risks calculated by Environ from any of these data sets, including the
Gerin cohort where the lung cancers did not show a clear positive trend
with cumulative Cr(VI) exposure.
Several commenters remarked on OSHA's use of both the Gibb cohort
and the Luippold cohort to define a preliminary range of risk estimates
associated with a working lifetime of exposure at the previous and
alternative PELs. Some suggested that OSHA should instead rely
exclusively on the Gibb study, due to its superior size, smoking data,
completeness of follow-up, and exposure information (Tr. 709-710, 769;
Exs. 40-18-1, pp. 2-3; 47-23, p. 3; 47-28, pp. 4-5). Others suggested
that OSHA should devise a weighting scheme to derive risk estimates
based on both studies but with greater weight assigned to the Gibb
cohort (Tr. 709-710, 769, Exs. 40-18-1, pp. 2-3; 47-23, p. 3), arguing
that "the use of the maximum likelihood estimate from the Luippold
study as the lower bound of OSHA's risk estimates * * * has the effect
of making a higher Permissible Exposure Limit (PEL) appear acceptable"
(Ex. 40-18-1, p. 3). OSHA disagrees with this line of reasoning. OSHA
believes that including all studies that provide a strong basis to
model the relationship between Cr(VI) and lung cancer, as the Luippold
study does, provides useful information and adds depth to the Agency's
risk assessment. OSHA agrees that in some cases derivation of risk
estimates based on a weighting scheme is an appropriate approach when
differences between the results of the two or more studies are believed
to primarily reflect sources of uncertainty or error in the underlying
studies. A weighting scheme might then be used to reflect the degree of
confidence in their respective results. However, the Gibb and Luippold
cohorts were known to be quite different populations, and the
difference between the risk estimates based on the two cohorts could
partly reflect variability in exposure-response. In this case, OSHA's
use of a range of risk defined by the two studies is appropriate for
the purpose of determining significance of risk at the previous PEL and
the alternative PELs that the Agency considered.
Another commenter suggested that OSHA should derive a "single
'best' risk estimate [taking] into account all of the six quantitative
risk estimates" identified by OSHA as featured or supporting risk
assessments in the preamble to the proposed rule, consisting of the
Gibb and Luippold cohorts as well as studies by Mancuso (Ex. 7-11),
Hayes (Ex. 7-14), Gerin (Ex. 7-120), and Alexander (Ex. 31-16-3) (Ex.
38-265, p. 76). The commenter, Mr. Stuart Sessions of Environomics,
Inc., proposed that OSHA should use a weighted average of risk
estimates derived from all six studies, weighting the Gibb and Luippold studies
more heavily than the remaining four "admittedly weaker studies" (Ex.
38-265, p. 78). During the public hearing, however, he stated that OSHA
may reasonably choose not to include some studies in the development of
its quantitative risk model based on certain criteria or qualifications
related to the principles of sound epidemiology and risk assessment
(Tr. 2484-2485). Mr. Sessions agreed with OSHA that sufficient length
of follow-up (>=20 years) is a critical qualification for a cohort to
provide an adequate basis for lung cancer risk assessment, admitting
that "if we are dealing with [a] long latency sort of effect and if
you only follow them for a few years it wouldn't be showing up with
anywhere near the frequency that you would need to get a statistically
significant excess risk" (Tr. 2485). This criterion supports OSHA's
decision to exclude the Alexander study as a primary data set for risk
assessment, due in part to the inadequate length of follow-up on the
cohort (average 8.9 years).
Mr. Sessions also agreed that the quality and comprehensiveness of
the exposure information for a study could be a deciding factor in
whether it should be used for OSHA's risk estimates (Tr. 2485-2487). As
discussed in the preamble to the proposed rule, significant uncertainty
in the exposure estimates for the Mancuso and Gerin studies was a
primary reason they were not used in the derivation of OSHA's
preliminary risk estimates (69 FR at 59362-3). Mancuso relied
exclusively on the air monitoring reported by Bourne and Yee (Ex. 7-98)
conducted over a single short period of time during 1949 to calculate
cumulative exposures for each cohort member, although the cohort
definition and follow-up period allowed inclusion of workers employed
as early as 1931 and as late as 1972. In the public hearing, Mr.
Sessions indicated that reliance on exposure data from a single year
would not necessarily "disqualify" a study from inclusion in the
weighted risk estimate he proposed, if "for some reason the exposure
hasn't changed much over the period of exposure" (Tr. 2486). However,
the Mancuso study provides no evidence that exposures in the
Painesville plant were stable over the period of exposure. To the
contrary, Mancuso stated that:
The tremendous progressive increase in production in the
succeeding years from zero could have brought about a concomitant
increase in the dust concentrations to 1949 that could have exceeded
the level of the first years of operation. The company instituted
control measures after the 1949 study which markedly reduced the
exposure (Ex. 7-11, p. 4).
In the Gerin et al. study, cohort members' Cr(VI) exposures were
estimated based on total fume levels and fume composition figures from
"occupational hygiene literature and and welding products
manufacturers' literature readily available at the time of the study",
supplemented by "[a] limited amount of industrial hygiene measurements
taken in the mid 1970s in eight of the [135] companies" from which the
cohort was drawn (Ex. 7-120, p. S24). Thus, cumulative exposure
estimates for workers in this cohort were generally not based on data
collected in their particular job or company. Gerin et al. explained
that the resulting "global average" exposure estimates "obscure a
number of between-plant and within-plant variations in specific factors
which affect exposure levels and would dilute a dose-response
relationship", including type of activity, * * * special processes,
arcing time, voltage and current characteristics, welder position, use
of special electrodes or rods, presence of primer paints and background
fumes coming from other activities (Ex. 7-120, p. S25).
Commenting on the available welding epidemiology, NIOSH emphasized
that wide variation in exposure conditions across employers may exist,
and should be a consideration in multi-employer studies (Ex. 47-19, p.
6). Gerin et al. recommended refinement and validation of their
exposure estimates using "more complete and more recent quantitative
data" and accounting for variability within and between plants, but
did not report any such validation for their exposure-response
analysis. OSHA believes that the exposure misclassification in the
Gerin study could be substantial. It is therefore difficult to place a
high degree of confidence in its results, and it should not be used to
derive the Agency's quantitative risk estimates. Comments received from
Dr. Herman Gibb support OSHA's conclusion. He stated that epidemiologic
studies of welders conducted to date do not include adequate data with
which to evaluate lung cancer risk (Ex. 47-8, p. 2).
Finally, Mr. Sessions agreed with OSHA that it is best to rely on
"independent studies on different cohorts of workers", rather than
including the results of two or more overlapping cohorts in the
weighted average he proposed (Tr. 2487). As discussed in the preamble
to the proposed rule, the Hayes et al. and Gibb et al. cohorts were
drawn from the same Baltimore chromate production plant (FR 69 at
59362). The workers in the subcohort of Hayes et al. analyzed by Braver
were first hired between 1945 and 1959; the Gibb cohort included
workers first hired between 1950 and 1974. Due to the substantial
overlap between the two cohorts, it is not appropriate to use the
results of the Hayes as well as the Gibb cohort in a weighted average
calculation (as proposed by Mr. Sessions).
Having carefully reviewed the various comments discussed above,
OSHA finds that its selection of the Gibb and Luippold cohorts to
derive a range of quantitative risk estimates is the most appropriate
approach for the Cr(VI) risk assessment. Support for this approach was
expressed by NIOSH, which stated that "the strength is in looking at
[the Gibb and Luippold studies] together * * * appreciating the
strengths of each" (Tr. 313). Several commenters voiced general
agreement with OSHA's study selection, even while disagreeing with
OSHA's application of these studies' results to specific industries.
Said one commenter, "[w]e concur with the selection of the two focus
cohorts (Luippold et al. 2003 and Gibb et al. 2000) as the best data
available upon which to base an estimate of the exposure-response
relationship between occupational exposure to Cr(VI) and an increased
lung cancer risk" (38-8, p. 6); and another, "[i]t is clear that the
data from the two featured cohorts, Gibb et al. (2000) and Luippold et
al. (2003), offer the best information upon which to quantify the risk
due to Cr(VI) exposure and an increased risk of lung cancer" (Ex. 38-
215-2, p. 16). Comments regarding the suitability of the Gibb and
Luippold cohorts as a basis for risk estimates in specific industries
will be addressed in later sections.
G. Issues and Uncertainties
The risk estimates presented in the previous sections include
confidence limits that reflect statistical uncertainty. This
statistical uncertainty concerns the limits of precision for
statistical inference, given assumptions about the input parameters and
risk models (e.g., exposure estimates, observed lung cancer cases,
expected lung cancer cases, linear dose-response). However, there are
uncertainties with regard to the above input and assumptions, not so
easily quantified, that may lead to underestimation or overestimation
of risk. Some of these uncertainties are discussed below.
1. Uncertainty With Regard to Worker Exposure to Cr(VI)
The uncertainty that may have the greatest impact on risk estimates
relates to the assessment of worker exposure. Even for the Gibb cohort,
whose exposures were estimated from roughly 70,000 air measurements
over a 35-year period, the calculation of cumulative exposure is
inherently uncertain. The methods used to measure airborne Cr(VI) did
not characterize particle size that determines deposition in the
respiratory tract (see section V.A). Workers typically differ from one
another with respect to working habits and they may have worked in
different areas in relation to where samples are taken. Inter-
individual (and intra-facility) variability in cumulative exposure can
only be characterized to a limited degree, even with extensive
measurement. The impact of such variability is likely less for
estimates of long-term average exposures when there were more extensive
measurements in the Gibb and Luippold cohorts in the 1960s through
1980s, but could affect the reliability of estimates in the 1940s and
1950s when air monitoring was done less frequently. Exposure estimates
that rely on annual average air concentrations are also less likely to
reliably characterize the Cr(VI) exposure to workers who are employed
for short periods of time. This may be particularly true for the Gibb
cohort in which a sizable fraction of cohort members were employed for
only a few months.
Like many retrospective cohort studies, the frequency and methods
used to monitor Cr(VI) concentrations may also be a source of
uncertainty in reconstructing past exposures to the Gibb and Luippold
cohorts. Exposures to the Gibb cohort in the Baltimore plant from 1950
until 1961 were determined based on periodic collection of samples of
airborne dust using high volume sampling pumps and impingers that were
held in the breathing zone of the worker for relatively short periods
of time (e.g., tens of minutes) (Ex. 31-22-11). The use of high volume
sampling with impingers to collect Cr(VI) samples may have
underestimated exposure since the accuracy of these devices depended on
an air flow low enough to ensure efficient Cr(VI) capture, the absence
of agents capable of reducing Cr(VI) to Cr(III), the proper storage of
the collected samples, and the ability of short-term collections to
accurately represent full-shift worker exposures. Further, impingers
would not adequately capture any insoluble forms of Cr(VI) present,
although other survey methods indicated minimal levels of insoluble
Cr(VI) were produced at the Baltimore facility (Ex. 13-18-14).
In the 1960s, the Baltimore plant expanded its Cr(VI) air
monitoring program beyond periodic high volume sampling to include
extensive area monitoring in 27 exposure zones around the facility.
Multiple short-term samples were collected (e.g., twelve one-hour or
eight three-hour samples) on cellulose tape for an entire 24 hour
period and analyzed for Cr(VI). Studies have shown that Cr(VI) can be
reduced to Cr(III) on cellulose filters under certain circumstances so
there is potential for underestimation of Cr(VI) using this collection
method (Ex. 7-1, p. 370). Monitoring was conducted prior to 1971, but
the results were misplaced and were not accessible to Gibb et al. The
area monitoring was supplemented by routine full-shift personal
monitoring of workers starting in 1977. The 24-hour area sampling
supplemented with personal monitoring was continued until plant closure
in 1985.
Some of the same uncertainties exist in reconstructing exposures
from the Luippold cohort. Exposure monitoring from operations at the
Painesville plant in the 1940s and early 1950s was sparse and consisted
of industrial hygiene surveys conducted by various groups (Ex. 35-61).
The United States Public Health Service (USPHS) conducted two
industrial hygiene surveys (1943 and 1951), as did the Metropolitan
Life Insurance Company (1945 and 1948). The Ohio Department of Health
(ODH) conducted surveys in 1949 and 1950. The most detailed exposure
information was available in annual surveys conducted by the Diamond
Alkali Company (DAC) from 1955 to 1971. Exponent chose not to consider
the ODH data in their analysis since the airborne Cr(VI) concentrations
reported in these surveys were considerably lower than values measured
at later dates by DAC. Excluding the ODH survey data in the exposure
reconstruction process may have led to higher worker exposure estimates
and lower predicted lung cancer risks.
There were uncertainties associated with the early Cr(VI) exposure
estimates for the Painesville cohort. Like the monitoring in the
Baltimore plant, Cr(VI) exposure levels were determined from periodic
short-term, high volume sampling with impingers that may have
underestimated exposures (Ex. 35-61). Since the Painesville plant
employed a "high-lime" roasting process to produce soluble Cr(VI)
from chromite ore, a significant amount of slightly soluble and
insoluble Cr(VI) was formed. It was estimated that up to approximately
20 percent of the airborne Cr(VI) was in the less soluble form in some
areas of the plant prior to 1950 (Ex. 35-61). The impingers were
unlikely to have captured this less soluble Cr(VI) so some reported
Cr(VI) air concentrations may have been underestimated for this reason.
The annual air monitoring program at the Painesville plant was
upgraded in 1966 in order to evaluate a full 24 hour period (Ex. 35-
61). Unlike the continuous monitoring at the Baltimore plant, twelve
area air samples from sites throughout the plant were collected for
only 35 minutes every two hours using two in-series midget impingers
containing water. The more frequent monitoring using the in-series
impinger procedure may be an improvement over previous high-volume
sampling and is believed to be less susceptible to Cr(VI) reduction
than cellulose filters. While the impinger collection method at the
Painesville plant may have reduced one source of potential exposure
uncertainty, another source of potential uncertainty was introduced by
failure to collect air samples for more than 40 percent of the work
period. Also, personal monitoring of workers was not conducted at any
time.
Concerns about the accuracy of the Gibb and Luippold exposure data
were expressed in comments following the publication of the proposed
rule. Several commenters suggested that exposures of workers in both
the Gibb and Luippold (2003) cohorts may have been underestimated,
resulting in systematic overestimation of risk in the analyses based on
these cohorts (Exs. 38-231, pp. 19-20; 38-233, p. 82; 39-74, p. 2; 47-
27, p. 15; 47-27-3, p. 1). In particular, the possibility was raised
that exposure measurements taken with the RAC sampler commonly used in
the 1960s may have resulted in lower reported Cr(VI) levels as a result
of reduction of Cr(VI) on the sample strip. Concerns were also raised
that situations of exceptionally high exposure may not have been
captured by the sampling plans at the Baltimore and Painesville plants
and that Cr(VI) concentrations in workers' breathing zones would have
been generally higher than concentrations measured in general area
samples taken in the two plants (Exs. 38-231, p. 19; 40-12-1, p. 2).
One commenter noted that "the exposure values identified in both the
Painesville and Baltimore studies are consistently lower than those
reported for a similar time period by alternative sources (Braver et
al. 1985; PHS 1953)" (Exs. 38-231, p. 19; 40-12-1, p. 2). It was also
suggested that impinger samples used to estimate exposures in the
Painesville plant and the impinger and RAC samples used between 1950 and 1985 in
the Baltimore plant did not efficiently capture particles smaller than
1 [mu]m in diameter, which were believed to have constituted a
substantial fraction of particles generated during the chromite ore
roasting process, and thus led to an underestimate of exposures (Ex.
47-27-3, pp. 1-4).
In his written testimony for the public hearing, Dr. Herman Gibb
addressed concerns about the type of samples on which the Gibb cohort
exposure estimates were based. Dr. Gibb stated, "[a] comparison of the
area and personal samples [collected during 1978-1985] found
essentially no difference for approximately two-thirds of the job
titles with a sufficient number of samples to make this comparison."
An adjustment was made for the remaining job titles, in which the area
samples were found to underestimate the breathing zone exposure, so
that the potential for underestimation of exposures based on general
area samples " * * * was accounted for and corrected * * * " in the
Gibb cohort exposure estimates (Ex. 44-4, pp. 5-6). Dr. Gibb also noted
that the publications claimed by commenters to have reported
consistently higher levels of exposure than those specified by the
authors of the Gibb et al. and Luippold et al. studies, in fact did not
report exposures in sufficient detail to provide a meaningful
comparison. In particular, Dr. Gibb said that the Public Health Service
(PHS) publication did not report plant-specific exposure levels, and
that Braver et al. did not report the locations or sampling strategies
used (Ex. 44-4, pp. 5-6).
OSHA agrees with Dr. Gibb that the use of RAC general area samples
in the Baltimore plant are unlikely to have caused substantial error in
risk estimates based on the Gibb cohort. A similar comparison and
adjustment between area and personal samples could not be performed for
the Luippold et al. cohort, for which only area samples were available.
The fact that most general area samples were similar to personal
breathing zone samples in the Gibb cohort does not support the
contention that reduction on the RAC sample strip or small particle
capture issues would have caused substantial error in OSHA's risk
estimates. Speculation regarding unusually high exposures that may not
have been accounted for in sampling at the Baltimore and Painesville
plants raises an uncertainty common to many epidemiological studies and
quantitative risk analysis, but does not provide evidence that
occasional high exposures would have substantially affected the results
of this risk assessment.
OSHA received comments from the Small Business Administration's
Office of Advocacy and others suggesting that, in addition to water-
soluble sodium dichromate, sodium chromate, potassium dichromate, and
chromic acid, some members of the Gibb and Luippold cohorts may have
been exposed to less soluble compounds such as calcium chromate (Tr.
1825, Exs. 38-7, p. 4; 38-8, p. 12; 40-12-5, p. 5). These less soluble
compounds are believed to be more carcinogenic than Cr(VI) compounds
that are water-soluble or water-insoluble (e.g. lead chromate). The
Painesville plant used a high-lime process to roast chromite ore, which
is known to form calcium chromate and lesser amounts of other less
water-soluble Cr(VI) compounds (Ex. 35-61). The 1953 USPHS survey
estimated that approximately 20 percent of the total Cr(VI) in the
roasting residue at the Painesville plant consisted of the less water-
soluble chromates (Ex. 2-14). The high lime roasting process is no
longer used in the production of chromate compounds.
Proctor et al. estimated that a portion of the Luippold cohort
prior to 1950 were probably exposed to the less water-soluble Cr(VI)
compounds due to the use of a high-lime roasting process, but that it
would amount to less than 20 percent of their total Cr(VI) exposure
(Ex. 35-61). The Painesville plant subsequently reduced and eliminated
exposure to Cr(VI) roasting residue through improvements in the
production process. A small proportion of workers in the Special
Products Division of the Baltimore plant may have been exposed to less
water-soluble Cr(VI) compounds during the occasional production of
these compounds over the years. However, the high-lime process believed
to generate less soluble compounds at the Painesville plant was not
used at the Baltimore plant, and the 1953 USPHS survey detected minimal
levels of less soluble Cr(VI) at this facility (Braver et al. 1985, Ex.
7-17).
OSHA agrees that some workers in the Luippold 2003 cohort
(Painesville plant) and perhaps in the Gibb cohort (Baltimore plant)
may have been exposed to minor amounts of calcium chromate and other
less-soluble Cr(VI) compounds. However, these exposures would have been
limited for most workers due to the nature of the production process
and controls that were instituted after the early production period at
the Painesville plant. The primary operation at the plants in
Painesville and Baltimore was the production of the water-soluble
sodium dichromate from which other primarily water-soluble chromates
such as sodium chromate, potassium dichromate, and chromic acid could
be made (Exs. 7-14; 35-61). Therefore, the Gibb and Luippold cohorts
were principally exposed to water-soluble Cr(VI). Risk of lung cancer
in these cohorts is therefore likely to reflect exposure to sodium
chromate and sodium dichromate, rather than calcium chromate.
The results of the recent German post-change cohort showed that
excess lung cancer mortality occurred among chromate-exposed workers in
plants exclusively using a no-lime production process (Ex. 48-4). Like
the Gibb cohort, the German cohort was exposed to average full-shift
Cr(VI) exposures well below the previous PEL of 52 [mu]g/m\3\ but
without the possible contribution from the more carcinogenic calcium
chromate (Exs. 48-1-2; Ex. 7-91). OSHA believes the elevated lung
cancer mortality in these post-change workers are further evidence that
occupational exposure to the less carcinogenic water-soluble Cr(VI)
present a lung cancer risk.
In their post-hearing brief, the Aerospace Industries Association
of America (AIA) stated:
OSHA's quantitative risk estimates are based on exposure
estimates derived from impinger and RAC samplers in the Painesville
and Baltimore chromate production plants. It is likely that these
devices substantially underestimated airborne levels of Cr(VI),
especially considering that particles were typically < 1 [mu]m. If
exposure in these studies were underestimated, the risk per unit
exposure was overestimated, and the risk estimates provided in the
proposed rule overstate lung cancer risks (Ex. 47-29-2, p. 4).
AIA supports its statements by citing a study by Spanne et al. (Ex. 48-
2) that found very low collection efficiencies (e.g. < 20 percent) of
submicron particles (i.e. < 1 [mu]m) using midget impingers. OSHA does
not dispute that liquid impinger devices, primarily used to measure
Cr(VI) air levels at the Painesville plant, are less effective at
collecting small submicron particles. However, OSHA does not believe
AIA has adequately demonstrated that the majority of Cr(VI) particles
generated during soluble chromate production are submicron in size.
This issue is further discussed in preamble section VI.G.4.a. Briefly,
the AIA evidence is principally based on a particle size distribution
from two airborne dust samples collected at the Painesville plant by an
outdated sampling device under conditions that essentially excludes
particles >5 [mu]m (Ex. 47-29-2, Figure 4).
OSHA believes it is more likely that Cr(VI) production workers in the
Gibb and Luippold cohorts were exposed to Cr(VI) mass as respirable
dust (i.e. < 10 [mu]m) mostly over 1 [mu]m in size. The Spanne et al.
study found that the impinger efficiency for particles greater than 2
[mu]m is above 80 percent. Cr(VI) exposure not only occurs during
roasting of chromite ore, where the smallest particles are probably
generated, but also during the leaching of water-soluble Cr(VI) and
packaging sodium dichromate crystals where particle sizes are likely
larger. Based on this information, OSHA does not have reason to believe
that the impinger device would substantially underestimate Cr(VI)
exposures during the chromate production process or lead to a serious
overprediction of risk.
The RAC samplers employed at the Baltimore plant collected airborne
particles on filter media, not liquid media. AIA provided no data on
the submicron particle size efficiency of these devices. For reasons
explained earlier in this section, OSHA finds it unlikely that use of
the RAC samplers led to substantial error in worker exposure estimates
for the Gibb cohort.
In summary, uncertainties associated with the exposure estimates
are a primary source of uncertainty in any assessment of risk. However,
the cumulative Cr(VI) exposure estimates derived from the Luippold
(2003) and Gibb cohorts are much more extensive than usually available
for a cancer cohort and are more than adequate as a basis for
quantitative risk assessment. OSHA does not believe the potential
inaccuracies in the exposure assessment for the Gibb and Luippold
(2003) cohorts are large enough to result in serious overprediction or
underprediction of risk.
2. Model Uncertainty, Exposure Threshold, and Dose Rate Effects
The models used to fit the observed data may also introduce
uncertainty into the quantitative predictions of risk. In the Preamble
to the Proposed Rule, OSHA solicited comments on whether the linear
relative risk model is the most appropriate approach on which to
estimate risk associated with occupational exposure to Cr(VI) (FR 69 at
59307). OSHA expressed particular interest in whether there is
convincing scientific evidence of a non-linear exposure-response
relationship and, if so, whether there are sufficient data to develop a
non-linear model that would provide more reliable risk estimates than
the linear approach that was used in the preliminary risk assessment.
OSHA received a variety of comments regarding the uncertainties
associated with using the risk model based on the Gibb and Luippold
cohorts to predict risk to individuals exposed over a working lifetime
to low levels of Cr(VI). OSHA's model assumes that the risk associated
with a cumulative exposure resulting from long-term, low-level exposure
is similar to the risk associated with the same cumulative exposure
from briefer exposures to higher concentrations, and that a linear
relative risk model adequately describes the cumulative exposure-
response relationship. These assumptions are common in cancer risk
assessment, and are based on scientifically accepted models of
genotoxic carcinogenesis. However, OSHA received comments from the
Small Business Administation's Office of Advocacy and others that
questioned the Agency's reliance on these assumptions in the case of
Cr(VI) (see e.g. Exs. 38-7, p. 2; 38-231, p. 18; 39-74, p. 2; 40-12-1,
p. 2; 38-106, p. 10, p. 23; 38-185, p. 4; 38-233, p. 87; 38-265-1, pp.
27-29; 43-2, pp. 2-3). Some comments suggested that a nonlinear or
threshold exposure-response model is an appropriate approach to
estimate lung cancer risk from Cr(VI) exposures. Evidence cited in
support of this approach rely on: (1) The lack of a statistically
significant increased lung cancer risk for workers exposed below a
cumulative Cr(VI) exposure of 1.0 mg/m\3\=yr (e.g., roughly equivalent
to 20 [mu]g/m\3\ TWA for a 45 year working lifetime) and below "a
highest reported eight hour average" Cr(VI) concentration of 52 [mu]g/
m\3\; (2) the lack of observed lung tumors at lower dose levels in rats
chronically exposed to Cr(VI) by inhalation and repeated intratracheal
installations; and (3) the existence of physiological defense
mechanisms within the lung, such as extracellular reduction of Cr(VI)
to Cr(III) and repair of DNA damage. These commenters argue that the
evidence suggests a sublinear nonlinearity or threshold in exposure-
response at exposures in the range of interest to OSHA.
The Small Business Administration's Office of Advocacy and several
other commenters stated that OSHA's risk model may overestimate the
risk to individuals exposed for a working lifetime at "low"
concentrations (Exs. 38-7, p. 2; 38-231, p. 18; 39-74, p. 2; 40-12-1,
p. 2) or at concentrations as high as 20-23 [mu]g/m\3\ (Exs. 38-7, p.
6; 38-106, p. 10, p. 23; 38-185, p. 4; 38-233, p. 87; 38-265-1, pp. 27-
29; 43-2, pp. 2-3), due to possible nonlinear features in the exposure-
response relationship for Cr(VI). These comments cited various
published analyses of the Luippold and Gibb cohorts, including the
Luippold et al. 2003 publication (Exs. 38-106, p. 10, p. 22; 38-233-4,
p. 17), the Proctor et al. 2004 publication (Ex. 38-233-4, p. 17), the
Crump et al. 2003 publication (Exs. 38-106, p. 22; 38-265-1, p. 27),
and an analysis conducted by Exponent on behalf of chromium industry
representatives (Ex. 31-18-15-1). The following discussion considers
each of these analyses, as well as the overall weight of evidence with
respect to cancer risk from low exposure to Cr(VI).
a. Linearity of the Relationship Between Lung Cancer Risk and
Cumulative Exposure
In the Luippold et al. 2003 publication (Ex. 33-10) and the Proctor
et al. 2004 publication (Ex. 38-216-10), the authors reported observed
and expected lung cancer deaths for five categories of cumulative
exposure. Lung cancer mortality was significantly elevated in
categories above 1.05 mg/m3-yr Cr(VI) (p < 0.05), and was
non-significantly elevated in the category spanning 0.20-0.48 mg/
m3-yr (8 observed lung cancer deaths vs. 4.4 expected), with
a slight deficit in lung cancer mortality for the first and third
categories (3 observed vs. 4.5 expected below 0.2 mg/m3-yr, 4 observed
vs. 4.4 expected at 0.48-1.04 mg/m3-yr) (Ex. 33-10, p. 455).
This analysis is cited by commenters who suggest that the lack of a
significantly elevated lung cancer risk in the range below 1.05 mg/
m3-yr may reflect the existence of a threshold or other
nonlinearity in the exposure-response for Cr(VI), and that OSHA's use
of a linear relative risk model in the preliminary risk assessment may
not be appropriate (Exs. 38-106, pp. 10-11; 38-233-4, p. 18). OSHA
received similar comments citing the Crump et al. (2003) publication,
in which the authors found a "consistently significant" trend of
increasing risk with increasing cumulative exposure for categories of
exposure above 1 mg/m3-yr (Ex. 35-58, p. 1157). The Exponent
analysis of the Gibb et al. cohort was also cited, which found that
lung cancer SMRs were not significantly elevated for workers with
cumulative exposures below 0.42 mg/m3-yrs Cr(VI) when
Baltimore reference rates and a six-category exposure grouping were
used (Ex. 31-18-15-1, Table 6).
Some commenters have interpreted these analyses to indicate
uncertainty about the exposure-response relationship at low exposure
levels. Others have asserted that "[c]redible health experts assessing
the same data as OSHA have concluded that 23 [mu]g/m3 is a protective
workplace standard (Ex. 38-185, p. 4) or that "[t]he Crump study
concluded that 23 [mu]g/m3 would be a standard that is
protective of workers health" (Ex. 47-35-1, p. 5). Contrary to these
assertions, it should be noted that the Gibb et al., Luippold et al.,
and Crump et al. publications do not include any statements concluding
that 23 [mu]g/m3 or any other exposure level is protective
against occupational lung cancer. OSHA has reviewed these analyses to
determine whether they provide sufficient evidence to support the use
of a nonlinear or threshold-based exposure-response model for the
Cr(VI) risk assessment, and whether they support the assertion that a
PEL higher than that proposed would protect workers against a
significant risk of lung cancer.
In discussing their results, Luippold et al. reported that
evaluation of a linear dose-response model using a chi-squared test
showed no significant departure from linearity and concluded that the
data are consistent with a linear dose-response model. They noted that
the results were also consistent with threshold or nonlinear effects at
low cumulative exposures, as they observed substantial increases in
cumulative exposure levels above approximately 1 mg/m3-yrs
(Ex. 33-10, p. 456). Ms. Deborah Proctor, lead author of the Proctor et
al. (2004) publication, confirmed these conclusions at the public
hearing, stating her belief that nonlinearities may exist but that the
data were also consistent with a linear dose response (Tr. 1845). The
authors of the Crump et al. 2003 publication (Ex. 35-58), in which
trend analyses were used to examine the exposure-response relationship
for cumulative exposure, stated that the data were " * * * neutral
with respect to these competing hypotheses" (Ex. 35-58, pp. 1159-
1160). Crump et al. concluded that their study of the Luippold cohort
" * * * had limited power to detect increases [in lung cancer risk] at
these low exposure levels" (Ex. 35-58, p. 1147). OSHA agrees with
Crump et al.'s conclusion that their study could not detect the
relatively small increases in risk that would be expected at low
exposures. With approximately 3000 person-years of observation time and
4.5 expected lung cancers in each of the three cumulative exposure
categories lower than 0.19 mg/m3-yrs Cr(VI) (Ex. 33-10, p.
455), analyses of the Luippold cohort cannot effectively discriminate
between alternative risk models for cumulative exposures that a worker
would accrue from a 45-year working lifetime of occupational exposure
at relatively low exposures (e.g., 0.045-0.225 mg/m3-yrs
Cr(VI), corresponding to a working lifetime of exposure at 1-5 [mu]g
Cr(VI)/m3).
The Exponent reanalysis of the Gibb cohort found that lung cancer
rates associated with exposures around 0.045 mg/m3-yrs
Cr(VI) and below were not significantly elevated in some analyses (Ex.
31-18-15-1, Table 6 p. 26). However, OSHA believes that this result is
likely due to the limited power of the study to detect small increases
in risk, rather than a threshold or nonlinearity in exposure-response.
In written testimony, Dr. Gibb explained that "[l]ack of a
statistically elevated lung cancer risk at lower exposures does not
imply that a threshold of response exists. As exposure decreases, so
does the statistical power of a given sample size to detect a
significantly elevated risk" (Ex. 44-4, p. 6). Exponent's analyses
found (non-significant) elevated risks for all exposure groups above
approximately 0.1 mg/m3-yrs, equivalent to 45 years of
occupational exposure at about 2.25 [mu]g/m3 Cr(VI) (Ex. 31-
18-15-1, p. 20, Table 3). Furthermore, Gibb et al.'s SMR analysis based
on exposure quartiles found statistically significantly elevated lung
cancer risks among workers with cumulative exposures well below the
equivalent of 45 years at the proposed PEL of 1 [mu]g/m3. As
Dr. Gibb commented at the hearing, the proposed PEL " * * * is within
the range of observation [of the studies] * * * In a sense, you don't
even need risk models" to show that workers exposed to cumulative
exposures equivalent to a working lifetime of exposure at or above the
proposed PEL have excess risk of lung cancer as a result of their
occupational exposure to Cr(VI)" (Tr. 121-122).
Furthermore, Robert Park of NIOSH reminded OSHA that "[a]nalysts
of both the Painesville and the Baltimore cohorts * * * did test for
deviation or departure from linearity in the exposure response and
found no significant effect. If there was a large threshold, you would
expect to see some deviance there" (Tr. 350-351). Post-hearing
comments from NIOSH indicated that further analysis of the Gibb data
provided no significant improvement in fit for nonlinear and threshold
models compared to the linear relative risk model (Ex. 47-19, p. 7).
Based on this evidence and on the previously discussed findings that
(1) linear relative risk models fit both the Gibb and Luippold data
sets adequately, and (2) the wide variety of nonlinear models tested by
various analysts failed to fit the available data better than the
linear model, OSHA believes that a linear risk model is appropriate and
that there is not convincing evidence to support the use of a threshold
or nonlinear exposure-response model, or to conclude that OSHA's risk
assessment has seriously overestimated risk at low exposures.
b. The Cumulative Exposure Metric and Dose-Rate Effects on Risk
The Small Business Administration's Office of Advocacy and several
other commenters questioned OSHA's reliance in the preliminary risk
assessment on models using cumulative exposure to estimate excess risk
of lung cancer, suggesting that cumulative exposures attained from
exposure to high concentrations of Cr(VI) for relatively short periods
of time, as for some individuals in the Gibb and Luippold cohorts, may
cause greater excess risk than equivalent cumulative exposures attained
from long-term exposure to low concentrations of Cr(VI) (Exs. 38-7, pp.
3-4, 38-215-2, pp. 17-18; 38-231, p. 18; 38-233, p. 82; 38-265-1, p.
27; 39-74, p. 2, 40-12-1, p. 2, 43-2, p. 2, 47-27, p. 14; 47-27-3, p.
1). This assertion implies that OSHA's risk assessment overestimates
risk from exposures at or near the proposed PEL due to a threshold or
dose-rate effect in exposure intensity. One commenter stated that
"[a]pplication of a linear model estimating lung cancer risk from
high-level expsoures . . . to very low-level exposure using the
exposure metric of cumulative dose will inevitably overestimate risk
estimates in the proposed PEL" (Ex. 47-27-3, p. 1). Comments on this
subject have cited analyses by Proctor et al. (2004) (Ex. 38-233-4, p.
17), Crump et al. (2003) (Exs. 38-106, p. 22; 38-265-1, p. 27),
Exponent (Ex. 31-18-15-1, pp. 31-34) and NIOSH (Ex. 47-19-1, p. 7); a
new study by Luippold et al. on workers exposed to relatively low
concentrations of Cr(VI) (Ex. 47-24-2); and mechanistic and animal
studies examining the potential for dose-rate effects in Cr(VI)-related
health effects (Exs. 31-18-7; 31-18-8; 11-7).
Of the two featured cohorts in OSHA's preliminary risk assessment,
the Gibb cohort is better suited to assess risk from exposure
concentrations below the previous PEL of 52 [mu]g Cr(VI)/m\3\. Contrary
to some characterizations of the cohort's exposures as too high to
provide useful information about risk under modern workplace conditions
(See e.g. Exs. 38-106, p. 21; 38-233, p. 82; 38-265-1, p. 28), most
members of the Gibb cohort had relatively low exposures, with
42[percnt] of the cohort members having a median annual average exposure
value below 10 [mu]g/m\3\ Cr(VI), 69[percnt] below 20 [mu]g/m\3\, and 91[percnt] below the
previous PEL (Ex. 35-295). In addition, Dr. Gibb indicated that
exposures in general were lower than suggested by some commenters (Tr.
1856, Ex. 38-215-2, p. 17). For example, about half of the total time
that workers were exposed was estimated to be below 14 [mu]g/m\3\
Cr(VI) from 1960-1985 (Ex. 47-8, p. 1).
Exponent calculated SMRs for six groups of workers in the Gibb
cohort, classified according to the level of their highest average
annual exposure estimates. They found that only the group of workers
whose highest exposure estimates were above approximately 95 [mu]g/m\3\
Cr(VI) had statistically significantly elevated lung cancer risk when
Baltimore reference rates were used (Ex. 31-18-15-1, p. 33). Exponent's
results are presented in Table VI-8 below, adapted from Table 10 in
their report (Ex. 31-18-15-1, p. 33).
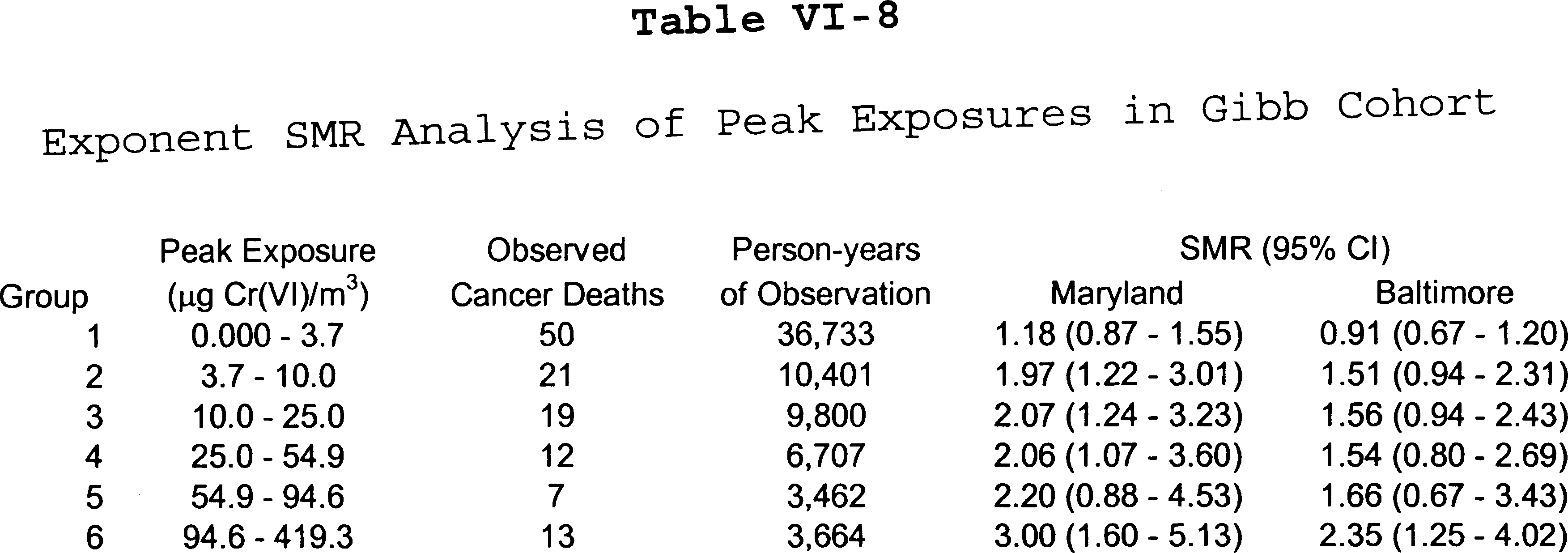
OSHA does not believe that Exponent's analysis of the Gibb data
provides convincing evidence of a threshold in exposure-response. While
the lower-exposure groups do not have statistically significantly
elevated lung cancer risk (p > 0.05) when compared with a Baltimore
reference population, the SMRs for all groups above 3.7 [mu]g/
m3 are consistently elevated. Moreover, the increased risk
approaches statistical significance, especially for those subgroups
with higher power (Groups 2 and 3). This can be seen by the lower 95%
confidence bound on the SMR for these groups, which is only slightly
below 1. The analysis suggests a lack of power to detect excess risk in
Groups 2-5, rather than a lack of excess risk at these exposure levels.
Analyses of the Luippold cohort by Crump et al. (Ex. 35-58) and
Proctor et al. (Ex. 38-216-10) used exposure estimates they called
"highest average monthly exposure" to explore the effects of exposure
intensity on lung cancer risk. They reported that lung cancer risk was
elevated only for individuals with exposure estimates higher than the
previous PEL of 52 [mu]g/m3 Cr(VI). Crump et al.
additionally found "statistically significant evidence of a dose-
related increase in the relative risk of lung cancer mortality" only
for groups above four times the previous PEL, using a series of Poisson
regressions modeling the increase in risk across the first two
subgroups and with the successive addition of higher-exposed subgroups
(Ex. 35-58, p. 1154).
As with the Gibb data, OSHA does not believe that the subgroup of
workers exposed at low levels is large enough to provide convincing
evidence of a threshold in exposure-response. In the Crump et al. and
Proctor et al. analyses, the groups for which no statistically
significant elevation or dose-related trends in lung cancer risk were
observed are quite small by the standards of cancer epidemiology (e.g.,
the Luippold cohort had only about 100 workers below the previous PEL
and about 40 workers within 1-3 times the previous PEL). Crump et al.
emphasized that " * * * this study had limited power to detect
increases [in lung cancer risk] at these low exposure levels" (Ex. 35-
58, p. 1147). The authors did not conclude that their results indicate
a threshold. They stated that their cancer potency estimates based on a
linear relative risk model using the cumulative exposure metric " * *
* are comparable to those developed by U.S. regulatory agencies and
should be useful for assessing the potential cancer hazard associated
with inhaled Cr(VI)" (Ex. 35-58, p. 1147).
OSHA discussed the Exponent, Crump et al. and Luippold et al. SMR
analyses of the Gibb and Luippold cohorts in the preamble to the
proposed rule, stating that the lack of a statistically significant
result for a subset of the entire cohort should not be construed to
imply a threshold (69 FR at 59382). During the hearing, Robert Park of
NIOSH expressed agreement with OSHA's preliminary interpretation,
adding that:
[W]e think that any interpretation of threshold in these studies
is basically a statistical artifact * * * It is important I think to
understand that any true linear or even just monotonic exposure
response that doesn't have a threshold will exhibit a threshold by
the methods that they used. If you stratify the exposure metric fine
enough and look at the lower levels, they will be statistically
insignificant in any finite study * * * telling you nothing about
whether or not in fact there is a threshold (Tr. 351).
To further explore the effects of highly exposed individuals on
OSHA's risk model, The Chrome Coalition suggested that OSHA should base
its exposure-response model on a subcohort of workers excluding those
who were exposed to " * * * an extraordinary exposure level for some
extended period of time* * * ", e.g., estimated exposures greater than
the previous PEL for more than one year (Ex. 38-231, p. 21). The Chrome
Coalition stated,
We are not aware of any study that has performed this type of
analysis but we believe that it should be a way of better estimating
the risk for exposures in the range that OSHA is considering for the
PEL (Ex. 38-231, p. 21).
To gauge the potential utility of such an analysis, OSHA examined the
subset of the Gibb cohort that was exposed for more than 365 days and
had average annual exposure estimates above the previous PEL of 52
[mu]g/m3 Cr(VI). The Agency found that the subcohort
includes only 82 such individuals, of whom 37 were reported as deceased
at the end of follow-up and five had died of lung cancer. In a cohort
of 2357 workers with 122 lung cancers out of 855 deaths, it is unlikely that
exclusion of a group this size would impact the results of a regression
analysis significantly, especially as the proportion of mortality
attributable to lung cancer is similar in the highly-exposed subgroup
and the overall cohort (5/37 0.135, 122/855 [cong] 0.143). The great
majority of the Gibb cohort members did not have the 'extraordinary'
exposure levels implied by the Chrome Coalition. As discussed
previously, most had relatively low exposures averaging less than 20
[mu]g/m3.
As discussed in their post-hearing comments, NIOSH performed
regression analyses designed to detect threshold or dose-rate effects
in the exposure-response relationship for the Gibb dataset (Ex. 47-19-
1, p. 7). NIOSH reported that "[t]he best fitting models had no
threshold for exposure intensity and the study had sufficient power to
rule out thresholds as large as 30 [mu]g/m\3\ CrO3 (15.6
[mu]g/m\3\ Cr(VI) * * * " and that there was no statistically
significant departure from dose-rate linearity when powers of annual
average exposure values were used to predict lung cancer risk (Ex. 47-
19-1, p. 7). This indicates that a threshold of approximately 20 [mu]g/
m\3\ Cr(VI) suggested in some industry comments is not consistent with
the Gibb cohort data. Based on these and other analyses described in
their post-hearing comments, NIOSH concluded that:
[E]xamination of non-linear features of the hexavalent chromium-
lung cancer response supports the use of the traditional (lagged)
"cumulative exposure paradigm * * * ": that is, linear exposure-
response with no threshold (Ex. 47-19-1, p. 7).
OSHA recognizes that, like most epidemiologic studies, neither the
Luippold nor the Gibb cohort provides ideal information with which to
identify a threshold or detect nonlinearities in the relationship
between Cr(VI) exposure and lung cancer risk, and that it is important
to consider other sources of information about the exposure-response
relationship at very low levels of Cr(VI) exposure. The Agency agrees
with Dr. Gibb's belief that " * * * arguments for a 'threshold' should
not be based on statistical arguments but rather on a biological
understanding of the disease process" (Ex. 44-4, p. 7) and Crump et
al.'s statement that " * * * one needs to consider supporting data
from mechanistic and animal studies" in order to determine the
appropriateness of assuming that a threshold (or, presumably, other
nonlinearity) in exposure-response exists (Ex. 35-58, p. 1159).
Experimental and mechanistic evidence and related comments relevant to
the issue of threshold and dose-rate effects are reviewed in the
following discussion.
c. Animal and Mechanistic Evidence Regarding Nonlinearities in
Cr(VI) Exposure-Response
In the NPRM, OSHA analyzed several animal and mechanistic studies
and did not find convincing evidence of a threshold concentration in
the range of interest (i.e. 0.25 to 52 [mu]g/m\3\). However, the Agency
recognized that evidence of dose rate effects in an animal instillation
study and the existence of extracellular reduction, DNA repair, and
other molecular pathways within the lung that protect against Cr(VI)-
induced respiratory tract carcinogenesis could potentially introduce
nonlinearities in Cr(VI) exposure-cancer response. OSHA solicited
comment on the scientific evidence for a non-linear exposure-response
relationship in the occupational exposure range of interest and whether
there was sufficient data to develop a non-linear model that would
provide more reliable risk estimates than the linear approach used in
the preliminary risk assessment (69 FR at 59307).
Some commenters believed the scientific evidence from animal
intratracheal instillation and inhalation of Cr(VI) compounds showed
that a linear risk model based on lung cancers observed in the Gibb and
Luippold cohorts seriously overpredicts lung cancer risk to workers
exposed at the proposed PEL (Exs. 38-216-1; 38-233-4; 38-231). The
research cited in support of this presumed non-linear response was the
intratracheal instillation study of Steinhoff et al. and the inhalation
study of Glaser et al. (Exs. 11-7; 10-11). For example, Elementis
Chromium states that:
Considering either the Steinhoff or Glaser studies, a calculated
risk based on the effect frequency at the highest daily exposure
would be considerably greater than that calculated from the next
lower daily exposure. We believe that the same effect occurs when
humans are exposed to Cr(VI) and consideration of this should be
taken when estimating risk at very low exposure levels based on
effects at much higher exposure levels (Ex. 38-216-1, p. 4).
Despite the different mode of Cr(VI) administration and dosing
schemes, the Steinhoff and Glaser studies both feature dose levels at
which there was no observed incidence of lung tumors. The Steinhoff
study found no significant lung tumor incidence in rats intratracheally
administered highly soluble sodium dichromate at 87 [mu]g Cr(VI)/kg or
less regardless of whether the dose was received five times a week or
once a week for 30 months. However, rats administered a higher dose of
437 [mu]g Cr(VI)/kg of sodium dichromate or a similar amount of the
slightly soluble calcium chromate once a week developed significant
increases (about 17 percent incidence) in lung tumors. The study
documented a 'dose rate effect' since the same total dose administered
more frequently (i.e. five times weekly) at a five-fold lower dose
level (i.e. 87 [mu]g Cr(VI)/kg) did not increase lung tumor incidence
in the highly soluble sodium dichromate-treated rats. The Glaser
inhalation study reported no lung tumors in rats inhaling 50 [mu]g
Cr(VI)/m\3\ of sodium dichromate or lower Cr(VI) concentrations for 22
hours/day, 7 days a week. However, the next highest dose level of 100
[mu]g Cr(VI)/m\3\ produced a 15 percent lung tumor incidence (i.e. 3 of
19 rats). Both studies are more fully described in Section V.B.7.a.
The apparent lack of lung tumors at lower Cr(VI) dose levels is
interpreted by the commenters to be evidence of a non-linear exposure-
response relationship and, possibly, an exposure threshold below which
there is no risk of lung cancer.
In written testimony, Dr. Harvey Clewell of ENVIRON Health Science
Institute addressed whether the Steinhoff, Glaser and other animal
studies provided evidence of a threshold for Cr(VI) induced lung
carcinogenicity (Ex. 44-5). He stated that the argument for the
existence of a threshold rests on two faulty premises:
(1) Failure to detect an increased incidence of tumors from a
given exposure indicates there is no carcinogenic activity at that
exposure, and
(2) Nonlinearities in dose response imply a threshold below
which there is no carcinogenic activity (Ex. 44-5, p. 13).
In terms of the first premise, Dr. Clewell states:
The ability to detect an effect depends on the power of the
study design. A statistically-based No Observed Adverse Effect Level
(NOAEL) in a toxicity study does not necessarily mean there is no
risk of adverse effect. For example, it has been estimated that a
typical animal study can actually be associated with the presence of
an effect in as many as 10% to 30% of the animals. Thus the failure
to observe a statistically significant increase in tumor incidence
at a particular exposure does not rule out the presence of a
substantial carcinogenic effect at that exposure (Ex. 44-5, p. 13-
14).
Dr. Clewell also addressed the second premise as it applies to the
Steinhoff instillation study as follows:
It has been suggested, for example, that the results of the
Steinhoff study suggest that dose rate is an important factor in the
carcinogenic potency of chrome (VI), and therefore, there must be a
threshold. But these data, while they do provide an indication of a
dose rate effect * * * they don't provide information about where and
whether a threshold or even a non-linearity occurs, and to what extent
it does occur at lower concentrations (Tr. 158-159).
OSHA agrees with Dr. Clewell that the absence of observed lung
tumor incidence at a given exposure (i.e. a NOAEL) in an animal study
should not be interpreted as evidence of a threshold effect. This is
especially true for clearly genotoxic carcinogens, such as Cr(VI),
where it is considered scientifically reasonable to expect some small,
but finite, probability that a very few molecules may damage DNA in a
single cell and eventally develop into a tumor. For this reason, it is
not appropriate to regard the lack of tumors in the Steinhoff or Glaser
studies as evidence for an exposure-response threshold.
Exponent, in a technical memorandum prepared for an ad hoc group of
steel manufacturers, raises the possibility that the lung tumor
responses in the Steinhoff and Glaser studies were the result of damage
to lung tissue from excessive levels of Cr(VI). Exponent suggests that
lower Cr(VI) exposures that do not cause 'respiratory irritation' are
unlikely to lead an excess lung cancer risk (Ex. 38-233-4). Exponent
went on to summarize:
In examining the weight of scientific evidence, for exposure
concentrations below the level which causes irritation, lung cancer
has not been reported. Not surprisingly, Cr(VI)-induced respiratory
irritation is an important characteristic of Cr(VI)-induced
carcinogenicity in both humans and animals * * * Based on the
information reviewed herein, it appears that the no effect level for
non-neoplastic respiratory irritation and lung cancer from
occupational exposure to Cr(VI) is approximately 20 [mu]g/m\3\. Thus
establishing a PEL of 1 [mu]g/m\3\ to protect against an excess lung
cancer risk is unnecessarily conservative (Ex. 38-233-4, p. 24).
In support of the above hypothesis, Exponent points out that only
the highest Cr(VI) dose level (i.e. 437 [mu]g Cr(VI)/kg) of sodium
dichromate employed in the Steinhoff study resulted in significant lung
tumor incidence. Tracheal instillation of this dose once a week
severely damaged the lungs leading to emphysematous lesions and
pulmonary fibrosis in the Cr(VI)-exposed rats. Lower Cr(VI) dose levels
(i.e. 87 [mu]g Cr(VI)/kg or less) of the highly water-soluble sodium
dichromate that caused minimal lung damage did not result in
significant tumor incidence. However, the study also showed that a
relatively low dose (i.e. 81 [mu]g Cr(VI)/kg) of slightly soluble
calcium chromate repeatedly instilled (i.e. five times a week) in the
trachea of rats caused significant lung tumor incidence (about 7.5
percent) in the absence of lung tissue damage. This finding is
noteworthy because it indicates that tissue damage is not an essential
requirement for Cr(VI)-induced respiratory tract carcinogenesis. The
same instilled dose of the slightly soluble calcium chromate would be
expected to provide a more persistent and greater source of Cr(VI) in
proximity to target cells within the lung than would the highly water-
soluble sodium dichromate. This suggests that the internal dose of
Cr(VI) at the tissue site, rather than degree of damage, may be the
critical factor determining lung cancer risk from low-level Cr(VI)
exposures.
Exponent applies similar logic to the results of the Glaser
inhalation study of sodium dichromate in rats. Exponent states:
In all experimental groups (i.e. 25, 50, and 100 [mu]g Cr(VI)/
m\3\), inflammation effects were observed, but at 100 [mu]g Cr(VI)/
m\3\ [the high dose group with significant lung tumor incidence],
effects were more severe, as expected (Ex. 38-233-4, p. 22).
This assessment contrasts with that of the study authors who remarked:
In this inhalation study, in which male Wistar rats were
continuously exposed for 18 months to both water soluble sodium
dichromate and slightly soluble chromium oxide mixture aerosols, no
clinical signs of irritation were obvious * * * For the whole time
of the study no significant effects were found from routine
hematology and clinico-chemical examinations in all rats exposed to
sodium dichromate aerosol (Ex. 10-11, p. 229).
The rats in the Glaser carcinogenicity study developed a focalized form
of lung inflammation only evident from microscopic examination. This
mild response should not be considered equivalent to the widespread
bronchiolar fibrosis, collapsed/distorted alveolar spaces and severe
damage found upon macroscopic examination of rat lungs instilled with
the high dose (437 [mu]g Cr(VI)/kg) of sodium dichromate in the
Steinhoff study. The non-neoplastic lung pathology (e.g. accumulation
of pigmentized macrophages) described following inhalation of sodium
dichromate at all air concentrations of Cr(VI) in the Glaser study are
more in line with the non-neoplastic responses seen in the lungs of
rats intratracheally instilled with lower dose levels of sodium
dichromate (i.e. 87 [mu]g Cr(VI)/kg or less) that did not cause tumor
incidence in the Steinhoff study. OSHA finds no evidence that severe
pulmonary inflammation occurred following inhalation of 100 [mu]g
Cr(VI)/m\3\ in the Glaser carcinogenicity study or that the lung tumors
observed in these rats were the result of 'respiratory irritation'. Dr.
Clewell also testified that lung damage or chronic inflammation is not
a necessary and essential condition for C(VI) carcinogenesis in the
Glaser study:
I didn't find any evidence that it [lung damage and chronic
inflammation] was necessary and essential. In particular, I think
the Glaser study was pretty good in demonstrating that there were
effects where they saw no evidence of irritation, or any clinical
signs of those kinds of processes (Tr. 192).
Subsequent shorter 30-day and 90-day inhalation exposures with
sodium dichromate in rats were undertaken by the Glaser group to better
understand the non-neoplastic changes of the lung (Ex. 31-18-11). The
investigation found a transitory dose-related inflammatory response in
the lungs at exposures of 50 [mu]g Cr(VI)/m\3\ and above following the
30 day inhalation. This initial inflammatory response did not persist
during the 90 day exposure study except at the very highest dose levels
(i.e. 200 and 400 [mu]g Cr(VI)/m\3\). Significant increases in
biomarkers for lung tissue damage (such as albumin and lactate
dehydrogenase (LDH) in bronchioalveolar lavage fluid (BALF) as well as
bronchioalveolar hyperplasia) also persisted through 90 days at these
higher Cr(VI) air levels, especially 400 [mu]g Cr(VI)/m\3\. The study
authors considered the transient 30-day responses to represent
adaptive, rather than persistent pathological, responses to Cr(VI)
challenge. A dose-related elevation in lung weights due to
histiocytosis (i.e. accumulation of lung macrophages) was seen in all
Cr(VI)-administered rats at both time periods. The macrophage
accumulation is also likely to be an adaptive response that reflects
lung clearance of inhaled Cr(VI). These study results are more fully
described in section V.C.3.
OSHA believes that Cr(VI)-induced carcinogenesis may be influenced
not only by the total Cr(VI) dose retained in the respiratory tract but
also by the rate at which the dose is administered. Exponent is correct
that one possible explanation for the dose rate effect observed in the
Steinhoff study may be the widespread, severe damage to the lung caused
by the immediate instillation of a high Cr(VI) dose to the respiratory
tract repeated weekly for 30 months. It is biologically plausible that
the prolonged cell proliferation in response to the tissue injury would
enhance tumor development and progression compared to the same total Cr(VI)
instilled more frequently at smaller dose levels that do not cause widespread
damage to the respiratory tract. This is consistent with the opinion of Dr. Clewell
who testified that:
I would not say that it [respiratory tract irritation, lung
damage, or chronic inflammation] is necessary and sufficient, but
rather it exacerbates an underlying process. If there is a
carcinogenic process, then increased cell proliferation secondary to
irritation is going to put mitogenic pressure on the cells, and this
will cause more likelihood of a transformation (Tr. 192).
OSHA notes that increased lung tumor incidence was observed in
animals instilled with lower dose levels of calcium chromate in the
Steinhoff study and after inhalation of sodium dichromate in the Glaser
study. These Cr(VI) exposures did not trigger extensive lung damage and
OSHA believes it unlikely that the lung tumor response from these
treatments was secondary to 'respiratory irritation' as suggested by
Exponent. The more thorough investigation by the Glaser group did not
find substantive evidence of persistent tissue damage until rats
inhaled Cr(VI) at doses two- to four-fold higher than the Cr(VI) dose
found to elevate lung tumor incidence in the their animal cancer
bioassay.
Exponent goes on to estimate a NOAEL (no observable adverse effect
level) for lung histopathology in the Steinhoff study. They chose the
lowest dose level (i.e. 3.8 [mu]g Cr(VI)/kg) in the study as their
NOAEL based on the minimal accumulation of macrophages found in the
lungs instilled with this dose of sodium dichromate five times weekly
(Ex. 38-233-4, p. 21). Exponent calculates that this lung dose is
roughly equivalent to the daily dose inhaled by a worker exposed to 27
[mu]g Cr(VI)/m\3\ using standard reference values (e.g. 70 kg human
inhaling 10 m\3\/day over a daily 8 hour work shift). Exponent
considers this calculated Cr(VI) air level as a threshold below which
no lung cancer risk is expected in exposed workers.
However, Steinhoff et al. instilled Cr(VI) compounds directly on
the trachea rather than introducing the test compound by inhalation,
and was only able to characterize a significant dose rate effect at one
cumulative dose level. For these reasons, OSHA considers the data
inadequate to reliably determine the human exposures where this
potential dose transition might occur and to confidently predict the
magnitude of the resulting non-linearity. NIOSH presents a similar view
in their post-hearing comments:
NIOSH disagrees with Dr. Barnhardt's analysis [Ex. 38-216-1] and
supports OSHA's view that the Steinhoff et al. [1986] rat study
found a dose-rate effect in rats under the specified experimental
conditions, that this effect may have implications for human
exposure and that the data are insufficient to use in a human risk
assessment for Cr(VI) * * * The study clearly demonstrates that,
within the constraints of the experimental design, a dose rate
effect was observed. This may be an important consideration for
humans exposed to high levels of Cr(VI). However, quantitative
extrapolation of that information to the human exposure scenario is
difficult (Ex. 47-19-1, p. 8).
Exponent also relies on a case investigation of the benchmark dose
methodology applied to the pulmonary biomarker data measured in the 90-
day Glaser study (Ex. 40-10-2-8). In this instance, the benchmark doses
represent the 95 percent lower confidence bound on the Cr(VI) air level
corresponding a 10 percent increase relative to unexposed controls for
a chosen biomarker (e.g. BALF total protein, albumin, or LDH). The
inhaled animal doses were adjusted to reflect human inhalation and
deposition in the respiratory tract as well as continuous environmental
exposure (e.g. 24 hours/day, 7 days/week) rather than an occupational
exposure pattern (e.g. 8 hours/day, 5 days/week). The benchmark doses
were reported to range from 34 to 140 [mu]g Cr(VI)/m\3\.
Exponent concludes that "these [benchmark] values are akin to a
no-observed-adverse-effect level NOAEL in humans to which uncertainty
factors are added to calculate an RfC [i.e. Reference Concentration
below which adverse effects will not occur in most individuals]" and
"taken as a whole, the studies of Glaser et al. suggest that both non-
neoplastic tissue damage and carcinogenicity are not observed among
rats exposed to Cr(VI) at exposure concentrations below 25 [mu]g/m\3\"
(Ex. 38-233-4, p. 22). Since the Exponent premise is that Cr(VI)-
induced lung cancer only occurs as a secondary response to
histopathological changes in the respiratory tract, the suggested 25
[mu]g Cr(VI)/m\3\ is essentially being viewed as a threshold
concentration below which lung cancer is presumed not to occur.
In his written testimony, Dr. Clewell indicated that the tumor data
from the Glaser cancer bioassay was more appropriately analyzed using
linear, no threshold exposure-response model rather than the benchmark
uncertainty factor approach that presumes the existence of threshold
exposure-response.
The bioassay of Glaser et al. provides an example of a related
difficulty of interpreting data from carcinogenicity studies. The
tumor outcome appears to be nonlinear (0/18, 0/18, and 3/19 at
0.025, 0.05, and 0.1 mg Cr/m\3\). However, although the outcomes are
restricted to be whole numbers (of animals), they should not be
evaluated as such. Because the nature of cancer as a stochastic
process, each observed outcome represents a random draw from a
Poisson distribution. Statistical dose-response modeling, such as
the multistage model used by OSHA, is necessary to properly
interpret the cancer dose-response. In the case of Glaser et al.
(1986) study, such modeling would produce a maximum likelihood
estimate of the risk at the middle dose that was greater than zero.
In fact, the estimated risk at the middle dose would be on the order
of several percent, not zero. Therefore, suggesting a lack of lung
cancer risk at a similar human exposure would not be a health
protective position (Ex. 44-5, p. 14).
The U.S. Environmental Protection Agency applied a linearized (no
threshold) multistage model to the Glaser data (Ex. 17-101). They
reported a maximum likelihood estimate for lifetime lung cancer risk of
6.3 per 1000 from continuous exposure to 1 [mu]g Cr(VI)/m\3\. This risk
would be somewhat less for an occupational exposure (e.g. 8 hours/day,
5 days/week) to the same air level and would be close to the excess
lifetime risk predicted by OSHA (i.e. 2-9 per 1000).
In summary, OSHA does not believe the animal evidence demonstrates
that respiratory irritation is required for Cr(VI)-induced
carcinogenesis. Significant elevation in lung tumor incidence was
reported in rats that received Cr(VI) by instillation or inhalation at
dose levels that caused minimal lung damage. Consequently, OSHA
believes it inappropriate to consider a NOAEL (such as 25 [mu]g/m\3\)
where lung tumors were not observed in a limited number of animals to
be a threshold concentration below which there is no risk. Statistical
analysis of the animal inhalation data using a standard dose-response
model commonly employed for genotoxic carcinogens, such as Cr(VI), is
reported to predict risks similar to those estimated by OSHA from the
occupational cohorts of chromate production workers. While the rat
intratracheal instillation study indicates that a dose rate effect may
exist for Cr(VI)-induced carcinogenesis, it can not be reliably
determined from the data whether the effect would occur at the
occupational exposures of interest (e.g. working lifetime exposures at
0.25 to 52 [mu]g Cr(VI)/m\3\) without a better quantitative
understanding of Cr(VI) dosimetry within the lung. Therefore, OSHA does
not believe that the animal data show that cumulative Cr(VI)
exposure is an inappropriate metric to estimate lung cancer risk.
Exponent used the clinical findings from chromate production
workers in the Gibb and Luippold cohorts to support their contention
that 'respiratory irritation' was key to Cr(VI)-induced lung cancer
(Ex. 28-233-4, p. 18-19). They noted that over 90 percent of chromate
production workers employed at the Painesville plant during the 1930s
and 1940s, including some Luippold cohort members, were reported to
have damaged nasal septums. Based on this, Exponent concludes:
Thus, it is possible that the increased incidence of lung cancer
in these workers (i.e. SMR of 365 from Luippold et al. cohort
exposed during the 1940s) is at least partially due to respiratory
system tissue damage resulting from high Cr(VI) concentrations to
which these workers were exposed. These exposures clearly exceed a
threshold for both carcinogenic and non-carcinogenic (i.e.
respiratory irritation) health effects (Ex. 38-233-4, p. 18).
Exponent noted that about 60 percent of the Gibb cohort also suffered
ulcerated nasal septum tissue. The mean estimated annual Cr(VI) air
level at time of diagnosis was about 25 [mu]g Cr(VI)/m\3\. Ulcerated
nasal septum was found to be highly correlated with the average annual
Cr(VI) exposure of the workers as determined by a proportional hazards
model. These findings, again, led Exponent to suggest that:
It may be reasonable to surmise that the high rates of lung
cancer risk observed among the featured cohorts (i.e. Gibb and
Luippold) was at least partially related to respiratory irritation
(Ex. 38-233-4, p. 19).
In its explanations, Exponent assumes that the irritation and
damage to nasal septum tissue found in the exposed workers also occurs
elsewhere in the respiratory tract. Exponent provided no evidence that
Cr(VI) concentrations that damage tissue at the very front of the nose
will also damage tissue in the bronchoalveolar regions where lung
cancers are found. A national medical survey of U.S. chromate
production workers conducted by the U.S. Public Health Service in the
early 1950s found greater than half suffered nasal septum perforations
(Ex. 7-3). However, there was little evidence of non-cancerous lung
disease in the workers. The survey found only two percent of the
chromate workers had chronic bronchitis which was only slightly higher
than the prevalence in nonchromate workers at the same plants and less
than had been reported for ferrous foundry workers. Just over one
percent of the chromate production workers in the survey were found to
have chest X-ray evidence consistent with pulmonary fibrosis. This led
the U.S. Public Health Service to conclude "on the basis of X-ray data
we cannot confirm the presence of pneumoconiosis from chromate
exposure" (Ex. 7-3, p. 80). An earlier report noted fibrotic areas in
the autopsied lungs of three Painesville chromate production workers
employed during the 1940s who died of lung cancer (Ex. 7-12). The
authors attributed the fibrotic lesions to the large amounts of
chromite (a Cr(III) compound) ore found in the lungs.
Exponent correctly noted that prevalence of nasal septum ulceration
in the Gibb cohort was "significantly associated with [average annual]
Cr(VI) exposure concentrations" using a proportional hazards model
(Ex. 38-233-4, p. 19). However, other related symptomatology, such as
nasal irritation and perforation, was not found to be correlated with
annual average Cr(VI) air levels. This led the authors to suggest that
nasal septum tissue damage was more likely related to short-term,
rather than annual, Cr(VI) air levels. Nasal septum ulceration was also
not a significant predictor of lung cancer when the confounding effects
of smoking and cumulative Cr(VI) exposure were accounted for in the
proportional hazards model (Ex. 31-22-11). The authors believed the
lack of correlation probably reflected cumulative Cr(VI) as the
dominant exposure metric related to the elevated lung cancer risk in
the workers, rather than the high, short-term Cr(VI) air levels thought
to be responsible for the high rate of nasal septum damage. The
modeling results are not consistent with nasal septum damage as a
predictor of Cr(VI)-induced lung cancer in chromate production workers.
Dr. Herman Gibb confirmed this in oral testimony:
* * * I was curious to see if [respiratory] irritation might be
predictive of lung cancer. We did univariate analyses and found that
a number of them were [predictive]. But whenever you looked at, when
you put it into the regression model, none of them were. In other
words, [respiratory] irritation was not predictive of the lung
cancer response (Tr. 144).
OSHA does not believe the evidence indicates that tissue damage in
the nasal septum of chromate production workers exposed to Cr(VI) air
levels around 20 [mu]g/m\3\ is responsible for the observed excess lung
cancers. The lung cancers are found in the bronchioalveolar region, far
removed from the nasal septum. Careful statistical analysis of the Gibb
cohort did not find a significant relationship between clinical
symptoms of nasal septum damage (e.g. ulceration, persistent bleeding,
perforation) and lung cancer mortality. A 1951 U.S. Public Health
Service medical survey found a high prevalence of nasal septum damage
with few cases of chronic non-neoplastic lung disease (e.g. chronic
bronchitis, pulmonary fibrosis). This suggests that the nasal septum
damage caused by high Cr(VI) air concentrations was not mirrored by
damage in lower regions of the respiratory tract where lung cancer
takes place. Given these findings, it seems unlikely that the lower
Cr(VI) air levels experienced by the Gibb cohort caused pervasive
bronchioalveolar tissue damage that would be responsible for the
clearly elevated lung cancer incidence in these workers. Therefore, the
Agency does not concur with Exponent that there is credible evidence
from occupational cohort studies that the high rates of lung cancer are
related to tissue damage in the respiratory tract or that occupational
exposure to 20 [mu]g Cr(VI)/m\3\ represents a 'no effect' level for
lung cancer.
Some commenters felt that certain physiological defense mechanisms
that protect against the Cr(VI)-induced carcinogenic process introduce
a threshold or sublinear dose-response (Exs. 38-233-4; 38-215-2; 38-
265). Some physiological defenses are thought to reduce the amount of
biologically active chromium (e.g. intracellular Cr(V), Cr(III), and
reactive oxygen species) able to interact with critical molecular
targets within the lung cell. A prime example is the extracellular
reduction of permeable Cr(VI) to the relatively impermeable Cr(III)
which reduces Cr(VI) uptake into cells. Other defense mechanisms, such
as DNA repair and apoptosis, can interfere with carcinogenic
transformation and progression. These defense mechanisms are presented
by commenters as highly effective at low levels of Cr(VI) but are
overwhelmed at high dose exposures and, thus, could "provide a
biological basis for a sublinear dose-response or a threshold below
which there is expected to be no increased lung cancer risk (Ex. 38-
215-2, p. 29).
One study, cited in support of an exposure-response threshold,
determined the amount of highly soluble Cr(VI) reduced to Cr(III) in
vitro by human bronchioalveolar fluid and pulmonary macrophage
fractions over a short period (Ex. 31-18-7). These specific activities
were used to estimate an "overall reducing capacity" of the lung. As
previously discussed, cell membranes are permeable to Cr(VI) but not
Cr(III), so only Cr(VI) enters cells to any appreciable extent. The
authors interpreted these data to mean that high
levels of Cr(VI) would be required to "overwhelm" the reduction
capacity before significant amounts of Cr(VI) could enter lung cells
and damage DNA, thus creating a biological threshold to the exposure--
response (Ex. 31-18-8).
There are several problems with this threshold interpretation. The
in vitro reducing capacities were determined in the absence of cell
uptake. Cr(VI) uptake into lung cells happens concurrently and in
parallel with its extracellular reduction, so it cannot be concluded
from the study data that a threshold reduction capacity must be
exceeded before uptake occurs. The rate of Cr(VI) reduction to Cr(III)
is critically dependant on the presence of adequate amounts of
reductant, such as ascorbate or GSH (Ex. 35-65). It has not been
established that sufficient amounts of these reductants are present
throughout the thoracic and alveolar regions of the respiratory tract
to create a biological threshold. Moreover, the in vitro activity of
Cr(VI) reduction in epithelial lining fluid and alveolar macrophages
was shown to be highly variable among individuals (Ex. 31-18-7, p.
533). It is possible that Cr(VI) is not rapidly reduced to Cr(III) in
some workers or some areas of the lung. Finally, even if there was an
exposure threshold created by extracellular reduction, the study data
do not establish the dose range in which the putative threshold would
occur.
Other commenters thought extracellular reduction and other
physiological defenses were unlikely to produce a biological threshold
(Exs. 44-5; 40-18-1). For example, Dr. Clewell remarked:
Although studies attempted to estimate capacities of Cr(VI) (De
Flora et al., 1997) the extracellular reduction and cellular uptake
of Cr(VI) are parallel and competing kinetic processes. That is,
even at low concentrations where reductive capacity is undiminished,
a fraction of Cr(VI) will still be taken up into cells, as
determined by the relative rates of reduction and transport. For
this reason, reductive capacities should not be construed to imply
"thresholds" below which Cr(VI) will be completely reduced prior
to uptake. Rather, they indicate that there is possibly a "dose-
dependent transition", i.e. a nonlinearity in concentration
dependence of the cellular exposure to Cr(VI). Evaluation of the
concentration-dependence of the cellular uptake of Cr(VI) would
require more data than is currently available on the relative
kinetics of dissolution, extracellular reduction, and cellular
uptake as well as on the homeostatic response to depletion of
reductive resources (e.g. reduction of glutathione reductase) (Ex.
44-5, p. 16)
The same logic applies to other 'defense mechanisms' such as DNA
repair and apoptosis. Despite the ability of cells to repair DNA damage
or to undergo apoptosis (i.e. a form of programmed cell death) upon
exposure to low levels of Cr(VI), these protections are not absolute.
Since a single error in a critical gene may trigger neoplastic
transformation and DNA damage increases with intracellular
concentration of Cr(VI), it stands to reason that there may be some
risk of cancer even at low Cr(VI) levels. If the protective pathways
are saturable (e.g. protective capacity overwhelmed) then it might be
manifested as a dose transition or nonlinearity. However, as explained
above, an extensive amount of kinetic modeling data would be needed to
credibly predict the dose level at which a potential dose transition
occurs. OSHA agrees with Dr. Clewell that "in the absence of such a
biologically based [kinetic] dose-response model it is impossible to
determine either the air concentration of Cr(VI) at which the
nonlinearity might occur or the extent of the departure from a linear
dose-response that would result. Therefore, the assumption of a linear
dose-response is justified" (Ex. 44-5, p.17-18).
In conclusion, OSHA believes that examination of the Gibb and
Luippold cohorts, the new U.S. cohorts analyzed in Luippold et al.
(2005), and the best available animal and mechanistic evidence does not
support a departure from the traditional linear, cumulative exposure-
based approach to cancer risk assessment for hexavalent chromium.
OSHA's conclusion is supported by several commenters (see e.g. Tr. 121,
186, Exs. 40-10-2, p. 6; 44-7). For example, NIOSH stated:
It is not appropriate to employ a threshold dose-response
approach to estimate cancer risk from a genotoxic carcinogen such as
Cr(VI) [Park et al. 2004]. The scientific evidence for a
carcinogenicity threshold for Cr(VI) described in the Preamble [to
the proposed rule] consists of the absence of an observed effect in
epidemiology studies and animal studies at low exposures, and in
vitro evidence of intracellular reduction. The epidemiologic and
animal studies lack the statistical power to detect a low-dose
threshold. In both the NIOSH and OSHA risk assessments, linear no-
threshold risk models provided good fit to the observed cancer data.
The in vitro extracellular reduction studies which suggested a
theoretical basis for a non-linear reseponse to Cr(VI) exposure were
conducted under non-physiologic conditions. These results do not
demonstrate a threshold of response to Cr(VI) exposure (Ex. 40-10-2,
p. 6).
OSHA's position is also supported by Dr. Herman Gibb's testimony at the
hearing that a linear, no-threshold model best characterizes the
relationship between Cr(VI) exposure and lung cancer risk in the Gibb
cohort (Tr. 121). Statements from Ms. Deborah Proctor and Crump et al.
(who conducted analyses utilizing the Luippold cohort) also indicated
that these data are consistent with the traditional linear model (Tr.
1845, Exs. 33-10, p. 456; 35-58, pp. 1159-1160). The significant excess
risk observed in the Gibb cohort, which was best suited to address risk
from low cumulative or average exposures, contradicts comments to the
effect that "[i]ncreased lung cancers have been demonstrated only at
workplace exposures significantly higher than the existing standard * *
* " (Ex. 38-185, p. 4) or that characterized OSHA's risk assessment
for the proposed PEL as "speculative" (Ex. 47-35-1, p. 4) or
"seriously flawed" (Ex. 38-106, p. 23). OSHA believes that the clear
excess risk among workers with cumulative exposures equivalent to those
accrued over a 45-year working lifetime of low-level exposure to
Cr(VI), combined with the good fit of linear exposure-response models
to the Gibb and Luippold (2003) datasets and the lack of demonstrable
nonlinearities or dose-rate effects, constitute strong evidence of risk
at low exposures in the range of interest to OSHA.
3. Influence of Smoking, Race, and the Healthy Worker Survivor Effect
A common confounder in estimating lung cancer risk to workers from
exposure to a specific agent such as Cr(VI) is the impact of cigarette
smoking. First, cigarette smoking is known to cause lung cancer.
Ideally, lung cancer risk attributable to smoking among the Cr(VI)-
exposed cohorts should be controlled or adjusted for in characterizing
exposure-response. Secondly, cigarette smoking may interact with the
agent (i.e., Cr(VI)) or its biological target (i.e., susceptible lung
cells) in a manner that enhances or even reduces the risk of developing
Cr(VI)-induced lung cancer from occupational exposures, yet is not
accounted for in the risk model. The Small Business Administration's
Office of Advocacy commented that such an interactive effect may have
improperly increased OSHA's risk estimates (Ex. 38-7, p. 4).
OSHA believes its risk estimates have adequately accounted for the
potential confounding effects of cigarette smoking in the underlying
exposure-lung cancer response data, particularly for the Gibb cohort.
One of the key issues in this regard is whether or not the reference
population utilized to derive the expected number of lung cancers
appropriately reflects the smoking behavior of the cohort members. The
risk analyses of the Gibb cohort by NIOSH and Environ indicate that
cigarette smoking was properly controlled for in the exposure-response
modeling. NIOSH applied a smoking-specific correction factor that
included a cumulative smoking term for individual cohort members (Ex.
33-13). Environ applied a generic correction factor and used lung
cancer mortality rates from Baltimore City as a reference population
that was most similar to the cohort members with respect to smoking
behavior and other factors that might affect lung cancer rates (Ex. 33-
12). Environ also used internally standardized models that did not
require use of a reference population and included a smoking-specific
(yes/no) variable. All these models predicted very similar estimates of
risk over a wide range of Cr(VI) exposures. There was less information
about smoking status for the Luippold cohort. However, regression
modeling that controlled for smoking indicated that it was not a
significant confounding factor when relating Cr(VI) exposure to the
lung cancer mortality (Ex. 35-58).
Smoking has been shown to interact in a synergistic manner (i.e.,
combined effect of two agents are greater than the sum of either agent
alone) with some lung carcinogens, most notably asbestos (Ex. 35-114).
NIOSH reported a slightly negative but nonsignficant interaction
between cumulative Cr(VI) exposure and smoking in a model that had
separate linear terms for both variables (Ex. 33-13). This means that,
at any age, the smoking and Cr(VI) contributions to the lung cancer
risk appeared to be additive, rather than synergistic, given the
smoking information in the Gibb cohort along with the cumulative
smoking assumptions of the analysis. In their final linear relative
risk model, NIOSH included smoking as a multiplicative term in the
background rate in order to estimate lifetime lung cancer risks
attributable to Cr(VI) independent of smoking. Although this linear
relative risk model makes no explicit assumptions with regard to an
interaction between smoking and Cr(VI) exposure, the model does assume
a multiplicative relationship between the background rate of lung
cancer in the reference population and Cr(VI) exposure. Therefore, to
the extent that smoking is a predominant influence on the background
lung cancer risk, the linear relative risk model implicitly assumes a
multiplicative (e.g., greater than additive and synergistic, in most
situations) relationship between cumulative Cr(VI) exposure and
smoking. Since current lung cancer rates reflect a mixture of smokers
and non-smokers, OSHA agrees with the Small Business Administration's
Office of Advocacy that the excess lung cancer risks from Cr(VI)
exposure predicted by the linear relative risk model may overestimate
the risks to non-smokers to some unknown extent. By the same token, the
model may underestimate the risk from Cr(VI) exposure to heavy smokers.
Because there were so few non-smokers in the study cohorts
(approximately 15 percent of the exposed workers and four lung cancer
deaths in the Gibb cohort), it was not possible to reliably estimate
risk for the nonsmoking subpopulation.
Although OSHA is not aware of any convincing evidence of a specific
interaction between cigarette smoking and Cr(VI) exposure, prolonged
cigarette smoking does have profound effects on lung structure and
function that may indirectly influence lung cancer risk from Cr(VI)
exposure (Ex. 33-14). Cigarette smoke is known to cause chronic
irritation and inflammation of the respiratory tract. This leads to
decreases in airway diameter that could result in an increase in Cr(VI)
particulate deposition. It also leads to increased mucous volume and
decreased mucous flow, that could result in reduced Cr(VI) particulate
clearance. Increased deposition and reduced clearance would mean
greater residence time of Cr(VI) particulates in the respiratory tract
and a potentially greater probability of developing bronchogenic
cancer. Chronic cigarette smoking also leads to lung remodeling and
changes in the proliferative state of lung cells that could influence
susceptibility to neoplastic transformation. While the above effects
are plausible consequences of cigarette smoking on Cr(VI)-induced
carcinogenesis, the likelihood and magnitude of their occurrence have
not been firmly established and, thus, the impact on risk of lung
cancer in exposed workers is uncertain.
Differences in lung cancer incidence with race may also introduce
uncertainty in risk estimates. Gibb et al. reported differing patterns
for the cumulative exposure-lung cancer mortality response between
whites and non-whites in their cohort of chromate production workers
(Ex. 31-22-11). In the assessment of risk from the Gibb cohort, NIOSH
reported a strong interaction between cumulative Cr(VI) exposure and
race, such that nonwhites had a higher cumulative exposure coefficient
(i.e., higher lung cancer risk) than whites based on a linear relative
risk model (Ex. 33-13). If valid, this might explain the slightly lower
risk estimates in the predominantly white Luippold cohort. However,
Environ found that including race as an explanatory variable in the Cox
proportional hazards model C1 did not significantly improve model fit
(p=0.15) once cumulative Cr(VI) exposure and smoking status had been
considered (Ex. 33-12).
NIOSH suggested that exposure or smoking misclassification might
plausibly account for the Cr(VI) exposure-related differences in lung
cancer by race seen in the Gibb cohort (Ex. 33-13, p. 15). It is
possible that such misclassification might have occurred as a result of
systematic differences between whites and non-whites with respect to
job-specific Cr(VI) exposures at the Baltimore plant, unrecorded
exposure to Cr(VI) or other lung carcinogens when not working at the
plant, or in smoking behavior. Unknown differences in biological
processes critical to Cr(VI)-induced carcinogenesis could also
plausibly account for an exposure-race interaction. However, OSHA is
not aware of evidence that convincingly supports any of these possible
explanations.
Another source of uncertainty that may impact the risk estimates is
the healthy worker survivor effect. Studies have consistently shown
that workers with long-term employment status have lower mortality
rates than short-term employed workers. This is possibly due to a
higher proportion of ill individuals and those with a less healthy
lifestyle in the short term group (Ex. 35-60). Similarly, worker
populations tend to be healthier than the general population, which
includes both employed and unemployed individuals. As a result,
exposure-response analyses based on mortality of long-term healthy
workers will tend to underestimate the risk to short-term workers and
vice versa, even when their cumulative exposure is similar. Also, an
increase in disease from occupational exposures in a working population
may not be detected when workers are compared to a reference population
that includes a greater proportion less healthy individuals.
The healthy worker survivor effect is generally thought to be less
of a factor in diseases with a multifactorial causation and long onset,
such as cancer, than in diseases with a single cause or short onset.
However, there is evidence of a healthy worker effect in several
studies of workers exposed to Cr(VI), as discussed further in the next
section ("Suitability of Risk Estimates for Cr(VI) Exposures in Other
Industries"). In these studies, the healthy worker survivor effect may
mask increased lung cancer mortality due to occupational Cr(VI) exposure.
4. Suitability of Risk Estimates for Cr(VI) Exposures in Other
Industries
At issue is whether the excess lung cancer risks derived from
cohort studies of chromate production workers are representative of the
risks for other Cr(VI)-exposed workers (e.g., electroplaters, painters,
welders). Typically, OSHA has used epidemiologic studies from one
industry to estimate risk for other industries. For example, OSHA
relied on a cohort of cadmium smelter workers to estimate the excess
lung cancer risk in a wide range of affected industries for its cadmium
standard (57 FR at 42102, 9/14/1992). This approach is usually
acceptable because exposure to a common agent of concern is the primary
determinant of risk and not some other factor unique to the workplace.
However, in the case of Cr(VI), workers in different industries are
exposed to various Cr(VI) compounds that may differ in carcinogenic
potency depending to a large extent on water solubility. The chromate
production workers in the Gibb and Luippold cohorts were primarily
exposed to certain highly water-soluble chromates. As more fully
described in section V.B. of the Cancer Effects section, the scientific
evidence indicates that all Cr(VI) compounds are carcinogenic but that
the slightly soluble chromates (e.g. calcium chromate, strontium
chromate, and some zinc chromates) exhibit greater carcinogenicity than
the highly water soluble chromates (e.g. sodium chromate, sodium
dichromate, and chromic acid) or the water insoluble chromates (e.g.
lead chromates) provided the same dose is delivered and deposited in
the respiratory tract of the worker. It is not clear from the available
scientific evidence whether the carcinogenic potency of water-insoluble
Cr(VI) compounds would be expected to be more or less than highly
water-soluble Cr(VI) compounds. Therefore, OSHA finds it prudent to
regard both types of Cr(VI) compounds to be of similar carcinogenic
potency.
The primary operation at the chromate production plants in
Painesville (Luippold cohort) and Baltimore (Gibb cohort) was the
production of the highly water-soluble sodium dichromate. Sodium
dichromate served as a starting material for the production of other
highly water-soluble chromates such as sodium chromate, potassium
dichromate, and chromic acid (Exs. 7-14; 35-61). As a result, the Gibb
and Luippold cohorts were principally exposed to water-soluble Cr(VI).
In the NPRM, OSHA requested comment on whether its risk estimates based
on the exposure-response data from these two cohorts of chromate
production workers were reasonably representative of the risks expected
from equivalent exposures to different Cr(VI) compounds encountered in
other industry sectors. Of particular interest was whether the
preliminary risk estimates from worker cohorts primarily engaged in the
production of the highly water soluble sodium chromate and sodium
dichromate would substantially overpredict lung cancer risk for workers
with the same level and duration of exposure to Cr(VI) but involving
different Cr(VI) compounds or different operations. These operations
include chromic acid aerosol in electroplating operations, the less
water soluble Cr(VI) particulates encountered during pigment production
and painting operations, and Cr(VI) released during welding, as well as
exposure in other applications.
OSHA received comments on this issue from representatives of a wide
range of industries, including chromate producers, specialty steel
manufacturers, construction and electric power companies that engage in
stainless steel welding, the military and aerospace industry that use
anti-corrosive primers containing Cr(VI), the surface finishing
industry, color pigment manufacturers, and the Small Business
Administration's Office of Advocacy (Exs. 38-231, 38-233; 38-8; 47-5;
40-12-4; 38-215; 40-12-5; 38-106; 39-43; 38-7). Many industry
commenters expressed concerns about the appropriateness of the
underlying Gibb and Luippold data sets and the methodology (e.g. linear
instead of threshold model) used to generate the lung cancer risk
estimates. These issues have been addressed in other parts of section
VI. The color pigment manufacturers asserted that lead chromate
pigments, unlike other Cr(VI) compounds, lacked carcinogenic potential.
This issue was addressed in section V.B.9 of the Health Effects
section. In summary, OSHA finds lead chromate and other water-insoluble
Cr(VI) compounds to be carcinogenic. The Agency further concludes that
it is reasonable to regard water insoluble Cr(VI) compounds to be of
similar carcinogenic potency to highly soluble Cr(VI) compounds. Based
on this conclusion, OSHA no longer believes that its risk projections
will underestimate the lung cancer risk for workers exposed to
equivalent levels of water-insoluble Cr(VI), as suggested in the NPRM
(69 FR at 59384).
Several commenters encouraged OSHA to rely on cohort studies that
examined the lung cancer mortality of workers in their particular
industry in lieu of the chromate production cohorts. Members of the
aircraft industry and their representatives commented that OSHA failed
to consider the results from several large cohort studies that showed
aerospace workers were not at increased risk of lung cancer (Exs. 38-
106; 38-215-2; 44-33; 47-29-2). In addition, Boeing Corporation and the
Aeropspace Industries Association (AIA) provided data on the size
distribution of Cr(VI) aerosols generated during primer spraying
operations which showed most particles to be too large for deposition
in the region of the respiratory tract where lung cancer typically
occurs (Exs. 38-106-2; 38-215-2; 47-29-2). The Specialty Steel Industry
maintained that epidemiological data specific to alloy manufacturing
and experience within the their industry show that the lung cancer risk
estimated by OSHA is unreasonably high for steel workers exposed to the
proposed PEL of 1 [mu]g Cr(VI)/m\3\ (Ex. 38-233, p. 82). Several
comments argued that there was a lack of scientific evidence for a
quantifiable exposure-response relationship between Cr(VI) exposure
from stainless steel welding (Exs. 38-8; 38-233-4). The commenters went
on to suggest that the OSHA quantitative Cr(VI) exposure-lung cancer
response model derived from the chromate production cohorts should not
be used to characterize the risk to welders. The suitability of the
OSHA risk estimates for these particular industries is further
discussed below.
a. Aerospace Manufacture and Maintenance. Most of the comments on
suitability of OSHA risk estimates were provided by AIA (Exs. 38-215;
47-29-2), Exponent on behalf of AIA (Exs. 38-215-2; 44-33), and the
Boeing Corporation (Exs. 38-106; 38-106-1). Cr(VI) is used as an anti-
corrosive in primers and other coatings applied to the aluminum alloy
structural surfaces of aircraft. The principal exposures to Cr(VI)
occur during application of Cr(VI) primers and coatings and mechanical
sanding of the painted surfaces during aircraft maintenance. Cr(VI)
exposures are usually in the form of the slightly soluble strontium and
zinc chromates used in primers and chromic acid found in other
treatments and coatings designed to protect metal surfaces.
Cohort Studies of Aerospace Workers. AIA commented that:
OSHA has all but ignored a substantial body of evidence of
studies showing no increased risk of lung cancer in aerospace
workers * * *. While epidemiologic studies show a link between lung
cancer and chromium VI exposure in other industries [e.g. chromate
production], that relationship is not established in the aerospace
industry (Ex.38-106, p. 16).
Aerospace commenters pointed to several cohort studies from aircraft
manufacturing and maintenance sites that did not find significantly
elevated lung cancer mortality in workers (Exs. 31-16-3; 31-16-4; 35-
213; 35-210). However, OSHA believes that the vast majority of workers
in these cohorts were not routinely engaged in jobs involving potential
Cr(VI) exposures.
Only two of the above studies (i.e., the Alexander and Boice
cohorts) specifically investigated the relationship between Cr(VI)
exposures and lung cancer mortality (Exs. 31-16-3; 31-16-4). The
Alexander cohort was evaluated as a supplemental data set for
quantitative risk assessment in sections VI.B.6 and VI.E.4. Briefly,
there were 15 observed lung cancer cases in the Alexander et al. study
with 19.5 expected (Ex. 31-16-3). There was no evidence of a positive
trend between cumulative Cr(VI) exposure and lung cancer incidence. The
lack of excess lung cancers was probably, in large part, due to the
short follow-up period (median nine years per member) and young age of
the cohort (median 42 years at the end of follow-up). Lung cancer
generally occurs 20 or more years after initial exposure to a
carcinogenic agent and mostly in persons aged 55 years and older. There
was no Cr(VI) air monitoring data for a significant portion of the
study period and reconstruction of worker exposure was reduced to a
limited number of 'summary time-weighted average exposure levels' based
on job category (Ex. 31-16-3). These limitations may have caused
inaccuracies in the worker exposure estimates that could lead to
potential misclassification of exposure, and, thus may also have
contributed to the lack of a positive Cr(VI) exposure--lung cancer
response.
In the their technical comments on behalf of the AIA, Exponent
considered the Boice cohort to be "the largest, best defined, most
completely ascertained, and followed for the longest duration" of the
epidemiological studies examining lung cancer mortality and other
health outcomes of aerospace workers (Ex. 38-215-2, p. 10). The Boice
cohort (previously described in section V.B.6) consisted of 77,965
aerospace workers employed over a thirty-year period at a large
aircraft manufacturing plant in California (Ex. 31-16-4). The average
duration of employment was over ten years and thirty percent of the
cohort was deceased. Therefore, the Boice cohort was larger, older, and
had greater follow-up than the Alexander cohort. Unfortunately, Cr(VI)
air measurements were sparse in recent years and entirely absent during
early years of plant operation so, unlike the Alexander cohort,
quantitative Cr(VI) exposure reconstruction was not attempted. Instead,
all jobs were qualitatively categorized by the chemicals involved
(e.g., chromates, trichloroethylene, perchloroethylene, etc.) and their
frequency of chemical usage (routine, intermittent, or no exposure).
Duration of potential chemical exposure, including Cr(VI), was
determined for the cohort members based on work history (Ex. 47-19-15).
There were 3634 workers in the cohort believed to have routine
exposures to Cr(VI), mostly in painting/primer operations or operating
process equipment used for plating and corrosion protection. Another
3809 workers were thought to have potential 'intermittent exposure' to
chromates. Most workers with potential exposure to Cr(VI) also had
potential exposures to the chlorinated solvents tricholoroethylene
(TCE) and perchloroethylene (PCE). Because of an inadequate amount of
Cr(VI) exposure data, OSHA was unable to use the Boice study for
quantitative risk assessment.
The Boice et al. study did not find excess lung cancer among the
45,323 aircraft factory workers when compared against the race-, age-,
calendar year-, and gender-adjusted rates for the general population of
the State of California (SMR=97). This is not a surprising result
considering more than 90 percent did not work in jobs that routinely
involve Cr(VI) exposure. Factory workers potentially exposed to Cr(VI)
also did not have significantly elevated lung cancer mortality
(SMR=102; 95% CI: 82-126) relative to the California general population
based on 87 observed lung cancer deaths. However, workers engaged in
spray painting/priming operations that likely had the highest potential
for Cr(VI) exposure did experience some excess lung cancer mortality
(SMR=111; 95% CI: 80-151) based on 41 deaths, but the increase was not
statistically significant.
As commonly encountered in factory work, there was evidence of a
'healthy worker effect' in this aerospace cohort that became
increasingly pronounced in workers with long-term employment. The
healthy worker effect (HWE) refers to the lower rate of disease
relative to the general population sometimes observed in long-term
occupational cohorts. For example, the Boice cohort factory workers
employed for 20 years had statistically significant lower rates of
death than a standardized California reference population for all
causes (SMR=78; 95% CI: 75-81), lung cancer (SMR=70; 95% CI: 61-80),
heart disease (SMR=79; 95% CI: 74-83), cerebrovascular disease (SMR=67;
95% CI: 56-78), non-malignant respiratory disease (SMR=65; 95% CI: 57-
74), and cirrhosis of the liver (SMR=67; 95% CI: 51-88) among other
specific causes (Ex. 31-16-4, Table 5). The study authors note that
"these reductions [in disease mortality] seem in part due to the
initial selection into the workforce and the continued employment of
healthy people [i.e. healthy worker effect] that is often found in
occupational studies" (Ex. 31-16-4, p. 592). If not properly accounted
for in mortality analysis, HWE can mask evidence of disease risk. Mr.
Robert Park, senior epidemiologist from NIOSH, confirmed this at the
public hearing when addressing implications of HWE for Cr(VI) lung
cancer risk in the Boice cohort.
This [Boice cohort] is a population where you would expect to
see a very dramatic healthy worker effect * * * so just off the top,
I would say any [relative risk] estimates for lung cancer in the
Boice population based on SMRs, I would want to adjust upwards by
0.9, for example, if the real SMR ought to be around 0.9 due to the
healthy worker effect. So if you do that in their population, they
have classified some workers as [routinely] exposed to chromates,
about 8 percent of the population. They observe a SMR of 1.02 in
that group. If you look at some of the other groupings in that
study, for example, assembly has an SMR of 0.92, fabrication, which
is basically make all the parts, 0.92, maintenance, 0.79. So a lot
of evidence for healthy worker effect in general in that population.
So the chromate group actually is at least 10 or 12 percent higher
in their lung cancer SMR. Now again, the numbers are small, you'd
have to have a very huge study for an SMR of 1.1 or 1.15 to be
statistically significant. So it is not. But it is a hint (Tr. 345-
347).
OSHA agrees with Mr. Park that the relative risks for lung cancer
in the Boice cohort are likely understated due to HWE. This is also
illustrated in the study analysis of the lung cancer morality patterns
by exposure duration to specific chemicals using internal cohort
comparisons. The internal analysis presumably minimize any biases (e.g.
smoking, HWE) that might exist from comparisons to the general
population. The results for workers potentially exposed to Cr(VI),
trichloroethylene (TCE), and perchloroethylene (PCE) are presented in
Table VI-9.

As shown in the table, there was a statistically significant
decline in relative risk of lung cancer among factory workers with
duration of TCE exposure (p< 0.01) and PCE exposure (p=0.02). This
mirrors the decline with increasing employment duration seen in
comparison with the general California population and strongly suggests
the internal cohort analysis failed to adequately adjust for HWE.
The table shows that, despite the downward influence of HWE on lung
cancer risk, there was a slight nonsignificant upward trend in excess
lung cancer mortality with duration of exposure to Cr(VI). The result
is that aircraft workers potentially exposed to chromate for five or
more years had 50 to 70 percent greater lung cancer mortality than
coworkers with a similar duration of potential exposure to the
chlorinated solvents. The relative excess is even more noteworthy given
that the subgroups had considerable overlap (e.g., many of the same
workers in the PCE and TCE groups were also in the chromate group).
This implies that a subset of Cr(VI) workers not exposed to chlorinated
solvents, possibly spray painters routinely applying Cr(VI) primers
over many years, may be at greater lung cancer risk than other Cr(VI)-
exposed members of the cohort.
The AIA and its technical representative, Exponent, objected to
OSHA reliance on the non-statistically significant upward trend in
excess lung cancers with increasing Cr(VI) exposure duration described
above (Exs. 38-215-2; 47-29-2). Exponent stated:
Statistical tests for trend indicated there is no evidence for a
trend of increasing risk of lung cancer with increasing years
exposed to chromate (P< 0.20). OSHA seems to have 'eye-balled' the
estimates and felt confident accepting the slight and non-
significant increases among risk estimates with overlapping
confidence intervals as evidence of a "slightly positive" trend.
However, OSHA's interpretation is an overstatement of the finding
and should be corrected in the final rule (Ex. 38-215-2, p. 13).
OSHA does not agree with these comments and believes it has
objectively interpreted the trend data in a scientifically legitimate
fashion. The fact that an upward trend in lung cancer risk with Cr(VI)
exposure duration fails to meet a statistical confidence of 95 percent
does not mean the relationship does not exist. For example, a trend
with a p-value of 0.2 means random chance will not explain the
relationship 80 percent of the time. The positive trend is all the more
notable given that it occurs in spite of a significant downward trend
in lung cancer mortality with years of employment. In other words,
aerospace workers exposed to Cr(VI) experienced a slightly greater lung
cancer mortality with increasing number of years exposed even while
their co-workers exposed to other chemicals were experiencing a
substantially lower lung cancer mortality with increasing years
exposed.
In its post-hearing comments, NIOSH calculated the observed excess
lung cancer risk to the Boice spray painters expected to have the
highest Cr(VI) exposures (SMR=1.11) to be 21 percent higher than the
minimally Cr(VI)-exposed assembly workers (SMR=0.92). NIOSH assumed the
painters were exposed to 15 [mu]g CrO3/m3 (i.e.,
the arithmetic mean of Cr(VI) air sampling data in the plant between
1978 to 1991) for 10 years (i.e., the approximate average duration of
employment) to derive an excess risk per mg CrO3/
m3 of 1.4 (Ex. 47-19-1). NIOSH noted that this was very
close to the excess risk per mg CrO3/m3 of 1.44
determined from their risk modeling of the Gibb cohort (Ex. 33-13). In
a related calculation, OSHA derived the expected excess risk ratio from
its linear relative risk model using a dose coefficient consistent with
the Gibb and Luippold data sets. Assuming the Boice spray painters were
exposed to 10 [mu]g Cr(VI)/m3 (90th percentile of plant air
sampling data converted from [mu]g CrO3 to [mu]g Cr(VI)) for
12 years (average employment duration of Boice factory workers), the
model predicts a risk ratio 1.20 which is also very close to the
observed excess risk ratio of 1.21 calculated from the observed SMR
data for spray painters above. These calculations suggest that the
excess lung cancer mortality observed in the Boice subcohort of Cr(VI)-
exposed aerospace workers is consistent with excess risks predicted
from models based on the Gibb and Luippold cohort of chromate
production workers.
The other cohort studies of aerospace workers cited by AIA were not
informative with regard to the association between Cr(VI) and lung
cancer. A cohort study by Garabrandt et al. of 14,067 persons employed
by an aircraft manufacturing company found significantly reduced excess
lung cancer mortality (SMR=80; 95% CI: 68-95) compared to adjusted
rates in the U.S. and San Diego County populations (Ex. 35-210). The
mean duration of follow-up was only 16 years and the study authors are
careful to state that the study can not rule out excess risk for
diseases, such as lung cancer, that have long latencies of 20 years or
more. The consistently low all-cause and cancer mortalities reported in
the study strongly suggest the presence of a healthy worker effect.
Another cohort study by Blair et al. of 14,457 aircraft maintenance
workers at Hill Air Force base in Utah did not find elevated lung
cancer mortality (SMR=90; 95% CI: 60-130) when compared to the general
population of Utah (Ex. 35-213). However, the study was exclusively
designed to investigate cancer incidence of chlorinated solvents (e.g.
TCE, PCE, methylene chloride) and makes no mention of Cr(VI). This was
also the case for a cohort study by Morgan et al. of 20,508 aerospace
workers employed at a Hughes Aircraft manufacturing
plant, which found no excess lung cancer mortality (SMR=0.96; 95% CI:
87-106) compared to the general U.S. population. However, a detailed
investigation of jobs at a large aircraft manufacturing facility (i.e.
facility studied by Boice et al.) found that only about 8 percent of
employees had potential for routine Cr(VI) exposure (Ex. 47-19-15). If
this is representative of the workforce in the other studies cited
above, it is doubtful whether a Cr(VI)-related increase in lung cancer
from a small proportion of workers would be reflected in the mortality
experience of the entire cohort, most of whom would not have been
exposed to Cr(VI).
In summary, OSHA does not find convincing evidence from the
aerospace cohort studies that the Agency's quantitative risk assessment
overstates the lung cancer risk to Cr(VI)-exposed workers. An
association between Cr(VI) exposure and lung cancer was never addressed
in most cohorts relied upon by the aerospace industry. Job analysis
shows that only a minor proportion of all aerospace workers are engaged
in workplace activities that routinely lead to Cr(VI) exposure. This
could explain the lack of excess lung cancer mortality found in studies
characterizing the mortality experience of all aerospace workers.
Alexander et al. identified a cohort of Cr(VI) exposed workers, made
individual worker estimates of cumulative Cr(VI) exposures, and found
no exposure-related trend with lung cancer incidence. However, the
absence of exposure-response could be the result of a number of study
limitations including the young age of the cohort (e.g. majority of
workers were under 50 years of age, when lung cancer incidence is
relatively uncommon), the inadequate follow-up period (e.g. majority of
workers followed < 10 years), and the potential for exposure
misclassification (e.g. Cr(VI) exposure levels prior to 1975 were not
monitored). Boice et al. also identified a subcohort of aerospace
workers with potential Cr(VI) exposure but lacked adequate air sampling
to investigate a quantitative relationship between Cr(VI) exposure and
lung cancer response. There was a significant decline in relative lung
cancer risk with length of employment among factory workers as well as
those exposed to chlorinated solvents, indicating a strong healthy
worker survivor effect among this pool of workers. The healthy worker
effect may have masked a significant trend in lung cancer with Cr(VI)
exposure duration. Risk projections based on the OSHA linear model were
found to be statistically consistent with the relative risk ratios
observed in the Boice cohort.
Cr(VI) Particle Size Distribution During Aerospace Operations.
Differences in the size of Cr(VI) aerosols generated during chromate
production and aerospace operations is another reason representatives
of the aircraft industry believe the OSHA risk estimates overstate risk
to aerospace workers (Exs. 38-106; 38-106-1; 38-215-2; 39-43; 44-33;
47-29-2). The submitted particle size data indicated that spraying
Cr(VI) primers mostly generates large aerosol droplets (e.g.
> 10 [mu]m) not expected to penetrate beyond the very upper portions of
the respiratory tract (e.g. nasal passages, larynx). Some aerospace
commenters also cited research showing that the few respirable primer
particulates that reach the lower regions of the lung contain less
Cr(VI) per particle mass than the larger non-respirable particles (Exs.
44-33; 38-106; 39-43). As a result, aerospace commenters contend that a
very small proportion of Cr(VI) aerosols generated by aircraft primer
operations deposit in the bronchioalveolar regions of the lung where
lung cancer occurs. OSHA agrees that the particle size studies
submitted to the record sufficiently demonstrate that a relatively
small proportion of Cr(VI) reaches the critical regions of the lung as
a result of these aircraft spraying operations. However, the Agency
believes the reduction in lung cancer risk from this lower Cr(VI)
particle burden is likely offset by the greater carcinogenic activity
of the slightly soluble strontium and zinc chromates inhaled during
spray primer application. Evaluation of the study data provided to the
record and the rationale behind the OSHA position are described below.
The Agency reviewed the information provided by Boeing on the
particle size of paint aerosols from typical spraying equipment used in
aerospace applications. Boeing provided size characterization of paint
aerosol from their in-house testing of spray paint equipment (Ex. 38-
106-1, p. 8-11). They measured droplet size distributions of non-
chromated polyurethane enamels generated by high volume low pressure
(HVLP) and electrostatic air spray guns under typical settings. The
particle size was measured 10 to 12 inches from the nozzle of the gun
using laser diffraction techniques. Boeing found the median volumetric
droplet diameter (Dv50) of the paint particles to be in the range of 17
to 32 [mu]m under the test conditions. Less than 0.5 percent of
droplets in the spray were 5 [mu]m and smaller (e.g. typical of
particles that deposit in the bronchioalveolar region). Boeing
concluded:
In typical operations and products, the best aerosol size is a
distribution with mass median diameter of about 30-40 microns, and a
relatively monodisperse distribution. As a result, the fraction of
the spray that is < 5 micron is about 1% or less; in overspray
perhaps [ap]2%. Therefore the deposited dose would be far less than
from exposure to an equal concentration of a smaller aerosol size,
and estimates of risk based on studies of other industry sectors are
not relevant to evaluation of risk in aerospace paint spraying (Ex.
38-106-1, p. 16).
Although Boeing used a non-chromated enamel paint in their studies,
they contend that the results would be representative of the particle
size distribution for a Cr(VI) primer using the same equipment under
similar conditions.
Boeing also submitted recent publications by the UCLA Center for
Occupational and Environmental Health measuring the Cr(VI) particle
size distribution during spray painting operations at an aerospace
manufacturing facility (Ex. 38-106-1). The UCLA group investigated
particle size distributions of Cr(VI) primers sprayed from HVLP
equipment in a lab bench-scale spray booth and in a field study of
spray booths at an aerospace facility (Ex. 38-106-1, attachment 6). The
tested primers contained the slightly soluble strontium chromate. The
study data are presented in two papers by Sabty-Daily et al. The
aerosol particles were collected at different locations several meters
from the spray gun in the bench-scale paint booth using a cascade
impactor. Full shift personal breathing zone samples from workers
spraying primer were also collected with a cascade impactor in the
field studies. The mass median aerodynamic diameter (MMAD) for Cr(VI)
particles in the field study was reported to be 8.5 [mu]m with a
geometric standard deviation of 2.2 [mu]m. On average, 62 percent of
the Cr(VI) mass was associated with non-respirable particles >10 [mu]m.
Taking into account deposition efficiency, it was estimated that less
than five percent of the Cr(VI) would potentially deposit in the lower
regions of the respiratory tract where lung cancer occurs. The bench
scale study gave particle distributions similar to the field studies.
It was shown that particle size decreases slightly as gun atomization
pressure increases. Particles in the direct spray were generally larger
than the overspray. Particle size was shown to decrease with distance
to the target surface due to evaporation of solvent.
Both Sabty-Daily articles and the Boeing submission made reference
to another study that measured particle size distribution of a HVLP-
generated paint aerosol in the breathing zone of the worker (Ex. 48-3).
Paint droplets were collected on polycarbonate filters with 0.2 [mu]m
pore size. Aerosol size was measured using a microscopic method that
minimizes bias from solvent evaporation. The breathing zone MMAD in the
overspray was reported to be 15 to 19 [mu]m with a GSD of 1.7 [mu]m. In
another study, LaPuma et al. investigated the Cr(VI) content of primer
particles from an HVLP spray gun using a cascade impactor (Ex. 31-2-2).
They reported that smaller particles (i.e. < 7 [mu]m) contained
disproportionately less Cr(VI) per mass of dry paint than larger
particles.
Boeing concluded that "the particle size distribution reported by
Sabty-Daily et al. (2004a) significantly underestimate the size
distribution of paint aerosol" (Ex. 38-106-1, p. 14). They state that
"in typical [spraying] operations and products the best aerosol size
is a distribution with mass median diameter of about 30-45 microns"
(Ex. 38-106-1, p. 16). This particle size is larger than 15 to 20 [mu]m
reported in independent breathing zone measurements of spray paint
aerosol collected on conventional sampling media (i.e. polycarbonate
filters) (Carlton and Flynn, 1997).
The Boeing rationale for dismissing the UCLA data was that the
cascade impactor had low collection efficiency for larger particles
relative to the Boeing laser diffraction method, which Boeing believes
is more accurate over the entire size distribution. OSHA notes,
however, that Boeing did not characterize aerosol particles in the
breathing zone of workers spraying Cr(VI) primer. Their study
characterized droplet size from an non-chromated enamel spray directly
out of the spray gun prior to contact with the target surface. While
collection efficiency accounts for some of the particle size
difference, other factors may also have contributed. These factors
include the composition of the spray paint, the sampling location, and
the degree of solvent evaporation. OSHA considers Cr(VI) primer
droplets with an average MMAD of 7 to 20 [mu]m, as measured in
breathing zone studies, to best represent the particle size inhaled by
a worker during spraying operations, since this range was measured in
breathing zone studies. The majority of these droplet particles would
not be expected to penetrate regions of the respiratory tract where
lung cancers occur.
While aerosol particle size during spray application of Cr(VI)
primers has been measured, AIA acknowledged that the particle size
distribution during sanding procedures has not been well studied (Exs.
38-106; 47-29-2). However, they believe that most of the particles
released as a result of sanding and grinding operations to remove old
paint coatings from aircraft are non-respirable (e.g. >10 [mu]m). OSHA
is not aware of reliable data in the record to support or refute this
claim.
The Cr(VI) particle size data from spray primer and sanding
applications in aerospace need to be evaluated against Cr(VI) particle
size during chromate production to determine its impact on OSHA risk
estimates. Boeing observed that the high temperature calcination
process that oxidizes chromite ore to sodium chromate would likely lead
to a high proportion of respirable fume (Ex. 38-106). During post-
hearing comments, AIA provided a figure from the 1953 U.S. Public
Health Service survey report that indicated the geometric mean airborne
dust particle size in a chromate production plant was 0.3 to 0.4 m in
size (Ex. 47-29-2, p. 3). The data came from a thermal precipitator
analysis of one-hour dust samples collected from the roasting and
leaching areas of the plant (Ex. 7-3). An independent 1950 industrial
hygiene survey report of the Painesville plant from the Ohio Department
of Health indicates the median size of the in-plant dust was 1.7
microns and the median size of the mist generated during the leaching
operations was 3.8 microns (Ex. 7-98). The measurement method used to
determine this particle size was not clear from the survey report.
The thermal precipitator used by the U.S. Public Health Service
survey is an older sampling device specifically used to characterize
particles smaller than 5 [mu]m. The thermal precipitator collection
efficiency for particles >5 [mu]m was considered suspect due to
gravitational and inertial effects caused by the very low air flow
rates (e.g. 6 ml/min) necessary to operate the device. The survey
figure shows that 95 percent of collected particles were smaller than 1
[mu]m. However, this is probably an inflated percentage given that the
thermal precipitator is unable to effectively collect particles outside
the fine and ultrafine range (e.g. greater than about 5 [mu]m).
In their post-hearing brief, AIA introduced an Exponent microscopic
analysis of particles claimed to be landfilled 'roast residue'
generated as airborne dust from the Painesville plant 'decades' earlier
(Ex. 47-29-2). AIA stated that "the particle diameters ranged from
0.11 to 9.64 [mu]m and that 82 percent of the particles were less than
2.5 [mu]m (Ex. 47-29-2, p. 3). OSHA was unable to verify the nature of
the landfill dust or determine its relevance from the information
provided by AIA.
In the same submission, AIA referenced several experimental and
animal studies as evidence that small particles less than 2.5 [mu]m in
diameter cause greater lung toxicity than larger particles (Ex. 47-29-
2). AIA concluded that:
It is important for OSHA to recognize in the quantitative risk
assessment that the particles to which the featured chromate
production workers were exposed were fine [particle diameters 0.1-
2.5 [mu]m] and ultrafine particles [particle diameters < 0.1 [mu]m]
and that particles of this size range are known to be associated
with greater toxicity than larger particles. Thus, the quantitative
cancer risk estimates based on these studies are very conservative
and likely overestimate risks for Cr(VI) exposures in other
industries, most notably aerospace (Ex. 47-29-2, p. 7).
The above studies showed that fine/ultrafine particles penetrate
into the alveolar region of the lung, are slowly cleared from
respiratory tract, and can lead to pulmonary inflammation and non-
neoplastic respiratory disease. OSHA agrees that fine/ultrafine
particles can disrupt pulmonary clearance and cause chronic
inflammation if sufficient amounts are inhaled. However, AIA did not
provide data that demonstrated the Gibb and Luippold workers were
routinely exposed to levels of small particles that would trigger
serious lung toxicity.
AIA also referred to a human epidemiological study that reported
the excess risk of lung cancer mortality from airborne fine/ultrafine
particles (i.e. 8 percent increase per 10 [mu]g/m\3\ in particles) to
be similar to the excess risk of cardiopulmonary disease (i.e. 6
percent increase with each 10 [mu]g/m\3\ in particles). AIA suggested
these results were evidence that the excess lung cancer mortality
attributed to Cr(VI) in chromate production cohorts were, in large
part, due to fine/ultrafine particles. However, the Luippold cohort had
an excess mortality from lung cancer (SMR=239) that was 10.6-fold
higher than the excess mortality of heart disease (SMR=113) (Ex. 33-
10). The Gibb cohort had an excess mortality from lung cancer that was
5.7-fold higher than the excess mortality of arteriosclerotic heart
disease (SMR=114) (Ex. 33-11). These mortality patterns are not
consistent with the small particle study results above and strongly
indicate fine/ultrafine particles are not the primary cause of excess
lung cancer among the chromate production workers in the Luippold and
Gibb cohorts. Given the information provided, OSHA does not have reason
to expect that exposure to fine/ultrafine particles in the Luippold and
Gibb cohorts had a substantial quantitative impact on its estimates of
lung cancer risk from exposure to Cr(VI).
Based on the evidence presented, OSHA believes the production of
sodium chromate and dichromate likely generated a greater proportion of
respirable Cr(VI) particles than the aerospace spray priming
operations. The roasting operation that oxidizes trivalent chromite ore
and soda ash to hexavalent sodium chromate salts would be expected to
generate a small particle fume based on information from other high
temperature calcination processes (e.g. beryllium oxide production).
This is supported by a small amount of particle size information from
the 1940s and 1950s (Ex. 7-98). However, there are insufficient data to
reliably determine the median diameter of Cr(VI) particles or otherwise
characterize the particle size distribution generated during sodium
chromate production in the breathing zone of the worker. It should also
be recognized that significant Cr(VI) exposures occurred during other
chromate production operations, such as leaching sodium chromate from
the roast, separating sodium dichromate crystals, and drying/bagging
the final purified sodium dichromate product. There is no information
on particle size for these operations, but it is reasonable to expect
greater proportions of larger particles than generated during the
roasting process. For these reasons, there is some degree of
uncertainty with regard to size distribution of Cr(VI) aerosols inhaled
by chromate production workers.
OSHA agrees with the aerospace industry that the reduced proportion
of respirable particles from spray primer operations relative to
chromate production will tend to lower the lung cancer risk from
equivalent Cr(VI) exposures. This is because less Cr(VI) will reach the
bronchioalveolar regions of the respiratory tract where lung cancer
occurs. However, the chemical form of Cr(VI) must also be considered.
Spray primer and painting operations expose workers to the slightly
soluble strontium and zinc chromates while chromate production workers
are exposed primarily to highly soluble sodium chromate/dichromate.
As explained earlier in section V.B.9 on carcinogenic effects,
animal and mechanistic evidence suggest that the slightly soluble
strontium and zinc chromates are more carcinogenic than the highly
soluble Cr(VI) compounds when equivalent doses are delivered to
critical regions of the respiratory tract. Slightly soluble Cr(VI)
compounds produced a higher incidence of bronchogenic tumors than
highly soluble Cr(VI) compounds (e.g. sodium dichromate, chromic acid)
when instilled in the respiratory tract of rats at similar dosing and
other experimental conditions (Ex. 11-2; 11-7). For example,
intrabronchial instillation of strontium chromate produced a 40 to 60-
fold greater tumor incidence than instillation of sodium dichromate in
one study (Ex. 11-2). Unlike the highly soluble Cr(VI) compounds, the
less water soluble Cr(VI) compounds are better able to provide a
persistent source of high Cr(VI) concentration within the immediate
microenvironment of the lung epithelia facilitating cellular uptake of
chromate ion into target cells. The greater carcinogenicity of the
slightly soluble Cr(VI) compounds have led to ACGIH TLVs that are from
5-fold (i.e. zinc chromates) to 100-fold (i.e. strontium chromates)
lower than the TLV for highly water soluble Cr(VI) compounds.
For these reasons, the risk reductions achieved from the lower
Cr(VI) particle burden that reaches the bronchioalveolar region of the
lung may, to a large extent, be offset by the greater carcinogenic
activity of the Cr(VI) compounds that are inhaled during aircraft spray
painting operations. Since significant lung cancer risk exists at
Cr(VI) air levels well below the new PEL (e.g. 0.5-2.5 [mu]g/m\3\)
based on chromate production cohorts, the risk would also likely be
significant even if the lung cancer risk from similar Cr(VI) exposures
in aerospace operations is slightly lower. Therefore, OSHA believes
that the risk models based on the Gibb and Luippold data sets will
provide reasonable estimates of lung cancer risk for aerospace workers
exposed to equivalent levels of Cr(VI). However, based on the lower
lung burden expected after considering the particle size distribution
evidence submitted to the record, OSHA no longer believes that its risk
projections will underestimate lung cancer risk for aerospace workers
exposed to strontium or zinc chromates, as suggested in the NPRM (69 FR
at 59384).
b. Specialty Steel Industry and Stainless Steel Welding.
Collier Shannon Scott submitted comments to OSHA on behalf of a
group of steel and superalloy industry trade associations and companies
including the Specialty Steel Industry of North America (SSINA), the
Steel Manufacturers Association (SMA), and the American Iron and Steel
Institute (AISI) as well as various individual companies. They
requested that OSHA "seriously consider" the results of the Arena et
al. (1998) study of workers employed in the high nickel alloys industry
(Tr. 661), as well as studies by Huvinen et al. (1996, 2002) and Moulin
et al. (1990) on stainless steel production workers (Exs. 38-233, p.
85; 47-5, p. 10) and by Danielsen et al. (1996) on Norweigen stainless
steel welders (Ex. 47-5, p. 10). On behalf of the SSINA, Ms. Joan
Fessler testified that the Arena et al. study (Ex. 38-233-2), also
referred to as the "Redmond Study", found no relationship between
Cr(VI) exposure and lung cancer, and in general " * * * no strong
epidemiological evidence causally associating occupational exposures
with excess risk" (Tr. 662). Ms. Fessler concluded that the study
results " * * * stand in stark contrast to the extrapolated estimates
of cancer risk OSHA has developed from the chromate worker cohorts to
develop the proposed rule" (Tr. 662) and "[show] that there is no
significant excess risk of lung cancer for workers in the steel
industry" (Ex. 40-12-4, p. 2). She cited studies conducted by Huvinen
et al. as additional evidence that workers in the stainless steel
production industry do not have excess risk of lung cancer from Cr(VI)
exposure (Tr. 663).
OSHA reviewed the Arena et al. (1998) study, which examined
mortality in a cohort of 31,165 workers employed at 13 U.S. high nickel
alloy plants for at least one year between 1956 and 1967 (Ex. 38-233-2,
p. 908). The focus of the study is nickel exposure; it does not report
how many of the cohort members were exposed to Cr(VI) or the levels of
Cr(VI) exposure to which they may have been exposed. Therefore there
does not appear to be any basis for SSINA's conclusion that "[t]here
was no strong epidemiological evidence causally associating
occupational exposures with excess risk" in the study and that "[n]o
dose response relationship was demonstrated * * * " (Tr. 662). Ms.
Fessler stated, in response to a question by Dr. Lurie of Public
Citizen, that there is no information in the study on Cr(VI) exposures
with which to assess a dose-response relationship between occupational
exposure to Cr(VI) and excess lung cancer risk in the cohort (Tr. 685).
Without any information on the proportion of workers that were exposed
to Cr(VI) or the levels to which they were exposed, one cannot
determine that there is no carcinogenic effect of Cr(VI) exposure, or
that the results of the Arena study contradict OSHA's risk estimates.
To more meaningfully compare the lung cancer risk predicted by
OSHA's risk model and that observed in the Arena et al. study, OSHA
estimated Cr(VI) exposures for the cohort members based in part on
exposures in the stainless steel industry. High-nickel alloys that
contain chromium are roughly comparable to stainless steel in terms of
chromium content and the temperatures at which they are melted. This
in turn determines the amount of trivalent chromium that converts to h
exavalent chromium in the heating process. For example, cast stainless steels
with high nickel composition (e.g. Cast 18-38, Cast 12-60, Cast 15-65,
and Cast 15-35) have chromium content ranging from 10-21% and have
melting points between 2350 and 2450 degrees Fahrenheit. Other high-nickel
alloys with chromium content, such as Hastelloy alloys C and G, Incoloy,
Nimonic, and Inconel, range from 13 to 22% chromium (except Incoloy 804=29.7% Cr)
with melting points of 2300-2600 degrees Fahrenheit. Stainless steels,
in general, have 12-30% chromium content and melting points between
2350 and 2725 degrees Fahrenheit.
For this analysis OSHA projected that the proportion of workers in
each production job category is approximately similar in stainless
steel and high-nickel alloy production. For example, OSHA assumed that
the percent of alloy production workers who are furnace operators is,
as in steel production, about 5%. Assuming that both the Cr(VI)
exposures typical of various production jobs and the proportion of
workers employed in each job are roughly similar, workers in the Arena
cohort producing high-nickel stainless steels and alloys containing
chromium are likely to have Cr(VI) exposures comparable to those
generally found in stainless steel production. Workers' exposures were
estimated using the exposure profile shown in Table III-62 of the Final
Economic Analysis section on steel mills (Ex. 49-1).
Not all workers in the Arena et al. cohort had Cr(VI) exposures
comparable to those in stainless steel facilities. As discussed by Ms.
Fessler at the hearing, exposure to " * * * [c]hrome was not uniform
in all [industries included in the study] because some of those
industries * * * did only high nickel work or nickel mining or whatever
specific nickel work there was" (Tr. 683). OSHA assumed that Cr(VI)
exposures of workers producing high-nickel alloys without chromium
content, such as Duranickel, Permanickel, Hastelloy alloys B, D, and G,
and Monel alloys, are similar to those found in carbon steel mills and
other non-stainless facilities, which according to comments submitted
by Collier Shannon Scott:
* * * may generate Cr(VI) due to trace levels of chromium in
feedstock materials or the inadvertent melting of stainless steel
scrap, as well as during various maintenance and welding operations
(Ex. 38-233, p. 10).
Exposure levels for Arena cohort workers producing these alloys were
estimated using the carbon steel exposure profile shown in Table III-64
of the Final Economic Analysis section on steel mills (Ex. 49-1).
Table VI-10 below shows the risk ratios (ratio of excess plus
background cancers to background only cancers) predicted by OSHA's
model for workers producing high-nickel alloys with and without
chromium content. The percentage of workers with 8-hour TWA exposures
in each range shown below are calculated for Ni-Cr alloys and non-Cr
alloys using profiles developed for the Final Economic Analysis
sections on stainless steel and carbon steel industries, respectively
(Ex. 49-1). An average exposure duration of 20 years was assumed. While
it was not clear how long workers were exposed on average, the reported
length of follow-up in the study indicates that the duration of
exposure was probably less than 20 years for most workers. Risk ratios
were calculated assuming that workers were followed through age 70. The
average age at end of follow-up was not clear from the Arena et al.
publication. Over half of the original cohort was under 30 as of 1978,
and follow-up ended in 1988 (Ex. 38-233-2, p. 908). Follow-up through
age 70 may therefore lead OSHA's model to overestimate risk in this
population, but would probably not lead to underestimation of risk.

The Arena et al. study reported lung cancer rates among white males
(who comprised the majority of the cohort) about 2%-13% higher than
background depending on the reference population used. The table above
illustrates that with reasonable assumptions about exposures in the
Arena cohort, OSHA's risk model predicts excess risks as low as those
reported by Arena et al. OSHA's model predicts the highest risks (1-6%
higher than background) among workers producing alloy mixtures similar
to stainless steel in chromium content. Unfortunately, it is not clear
from the Arena et al. publication how many of the workers were involved
in production of chromium-containing alloys. If an even split is
assumed between workers producing alloys with and without chromium
content in the Arena et al. cohort, OSHA's model predicts a lung cancer
rate between 0.8% and 3.8% higher than background.
More precise information about the level or duration of cohort
members' exposures might increase or decrease OSHA's model predictions
somewhat. For example, some workers in the historical alloy industry
would have had higher exposures than their modern-day counterparts, so
that better exposure information may lead to somewhat higher model
predictions. On the other hand, better information on the duration of
exposure and workers' age at the end of follow-up would lower the model
predictions, because this analysis made assumptions likely to
overestimate both. The analysis presented here should be interpreted
cautiously in light of the considerable uncertainty about the actual
exposures to the Arena cohort members, and the fact that OSHA's model
predictions are based on a lifetable using year 2000 U.S. all-cause
mortality data (rather than data from the time period during which the
cohort was followed). This analysis is not intended to provide a
precise estimate of risk from exposure to Cr(VI) in the Arena cohort,
but rather to demonstrate that the relatively low excess risk seen in
the cohort is reasonably consistent with the excess risk that OSHA's
model would predict at low exposures. It illustrates that OSHA's risk
model does not predict far higher risk than was observed in this
cohort. Rather, the majority of workers in alloy production would be
predicted to have relatively low risk of occupational lung cancer based
on their relatively low exposure to Cr(VI).
Regarding the Huvinen et al. (1996, 2002) studies, the comments
submitted by Collier Shannon Scott state that "there was not a
significant increase in the incidence of any disease, including lung
cancer, as compared to the control population" (Ex. 38-233, p. 85).
However, the authors also noted that risk of cancer could not be
excluded because the follow-up time was short and the exposed group was
young and small (Ex. 38-233-3, p. 747).
In addition to the small size (109 workers) and young age (mean
43.3 years) of the Cr(VI)-exposed group in the Huvinen et al. study
population, the design of this study limits its relevance to the issue
of lung cancer risk among stainless steel workers. The subjects were
all employed by the company at the time of the study. Individuals with
lung cancer would be expected to leave active employment, and would not
have been surveyed in the study. The authors made only a limited
attempt to track former workers: Those who met the study criteria of 8
years' employment in a single production department were surveyed by
mailed questionnaire (Ex. 38-233-3, p. 743), and no follow-up on
nonrespondents was reported. A second study conducted on the original
study group five years later was again limited to employed workers, as
those who had left the company " * * * could not be contacted" (Ex.
38-233-3, p. 204). Due to the short follow-up period and the
restriction to living workers (still employed or survey respondents),
these studies are not well suited to identify lung cancer cases.
Post-hearing comments stated that " * * * OSHA has failed to even
consider specific epidemiological studies performed on stainless steel
production workers and welders that would be far more relevant than the
chromate production studies OSHA relied upon for its analysis" (Ex.
47-5, p. 10). In particular, they suggest that OSHA should consider a
study by Danielsen et al. (1996) on Norweigian boiler welders and a
study by Moulin et al. (1990) on French stainless steel production
workers (Ex. 47-5, p. 10). However, the Moulin et al. study (Ex. 35-
282), was discussed in the Preamble to the Proposed Rule (69 FR at
59339). OSHA concluded that the association between Cr(VI) and
respiratory tract cancer in this and similar studies is difficult to
assess because of co-exposures to other potential carcinogens such as
asbestos, polycyclic aromatic hydrocarbons, nickel, and the lack of
information on smoking (69 FR at 59339).
The Danielsen et al. study was not evaluated in the NPRM, but is
similar to other studies of welders evaluated by OSHA in which excess
risk of lung cancer did not appear to be associated with stainless
steel welding. In Danielsen et al., as in most other welding studies,
no quantitative information on Cr(VI) exposure was available, there was
potential confounding by smoking and asbestos exposure, and there
appeared to be an overall healthy worker effect in the study (625
deaths vs. 659 expected). Therefore, OSHA does not believe that
Danielsen et al. contributes significant information beyond that in the
studies that are reviewed in Section V.B.4 of this preamble. OSHA's
interpretation and conclusions regarding the general findings of
welding cohort studies, discussed below in the context of comments
submitted by the Electric Power Research Institute, apply to the
results of Danielsen et al. as well.
The Electric Power Research Institute (EPRI), Exponent, and others
submitted comments to OSHA that questioned whether the Agency's
exposure-response model, based on the Gibb and Luippold chromate
production industry cohorts, should be used to estimate lung cancer
risks to welders exposed to Cr(VI) (Exs. 38-8; 38-233-4; 39-25, pp. 2-
3). EPRI stated that:
OSHA's review of the toxicology, epidemiology, and mechanistic
data associated with health effects among welders was thorough and
accurate. We concur with the selection of the two focus cohorts
(Luippold et al. 2003 and Gibb et al. 2000) as the best data
available upon which to base an estimate of the exposure-response
relationship between occupational exposure to Cr(VI) and an
increased lung cancer risk"; however * * * it may be questionable
whether that relationship should be used for stainless steel welders
given that a positive relationship between exposure to Cr(VI) and
lung cancer risk was not observed in most studies of welder cohorts
(Ex. 38-8, pp. 6-7).
EPRI's concerns, like other comments submitted to OSHA on risk to
welders, are based primarily on the results of the Gerin et al. (1993)
study and on several studies comparing stainless steel and mild steel
welders.
As discussed above in Section V., Gerin et al. (1993) is the only
available study that attempts to relate estimated cumulative Cr(VI)
exposure and lung cancer risk among welders. While excess lung cancer
risks were found among stainless steel welders, there was no clear
relationship observed between the estimated amount of Cr(VI) exposure
and lung cancer (Ex. 38-8, p. 8). This led the authors to suggest that
the elevated risks might be " * * * related to other exposures such as
cigarette smoking, background asbestos exposure at work or other
occupational or environmental risks * * * " rather than to Cr(VI)
exposure. On the other hand, Gerin et al. stated that " * * * the
welding fume exposures in these populations may be too low to demonstrate
a gradient of risk", or misclassification of exposure might obscure the
dose-response relationship (Ex. 7-120, pp. S25-S26), a point with which EPRI
expressed agreement (Ex. 38-8, p. 8).
OSHA agrees with Gerin et al. that co-exposures to carcinogens such
as nickel, asbestos, and cigarette smoke may have contributed to the
elevated lung cancer risks among welders. OSHA also agrees with the
authors that exposure misclassification may explain the absence of a
clear relationship between Cr(VI) and lung cancer in this study. Gerin
et al. derived their exposure data primarily from literature on welding
fume, as well as from a limited number of industrial hygiene
measurements taken in the mid 1970s in eight of the 135 companies
participating in the study (Ex. 7-120, p. S24, p. S27). Their exposure
estimates took account of the welding process used and the base metal
welded by individuals in the cohort, but they apparently had no
information on other important items, such as the size of the work
piece and weld time, which were identified by EPRI as factors affecting
the level of Cr(VI) exposure from welding (Ex. 38-8, p. 5).
EPRI also identified ventilation as a particularly important
determinant of exposure (Ex. 38-8, p. 5). Gerin et al. did not appear
to have individual information on ventilation use for their exposure
estimates, relying instead on "information on the history of welding
practice * * * obtained from each company on the basis of an ad hoc
questionnaire" that described for each company the average percent of
time that welders used local ventilation, operated in confined or open
areas, and worked indoors or outdoors (Ex. 7-120, p. S23). The use of
local ventilation, time spent welding in confined areas, and time spent
welding outdoors may have varied considerably from worker to worker
within any single company. In this case exposure estimates based on
company average information would tend to overestimate exposure for
some workers and underestimate it for others, thus weakening the
appearance of an exposure-response relationship in the cohort.
Gerin et al. also stated that the average exposure values they
estimated do not account for a number of factors which affect welders'
exposure levels, including " * * * type of activity (e.g. maintenance,
various types of production), special processes, arcing time, voltage
and current characteristics, welder position, use of special electrodes
or rods, presence of primer paints and background fumes coming from
other activities" (Ex. 7-120, p. S25). They noted that the resulting
difficulty in the construction of individual exposure estimates is
exacerbated by aggregation of data across small cohorts from many
different companies that may have different exposure conditions (Ex. 7-
120, p. S25). According to Gerin et al., exposure misclassification of
this sort may have obscured a dose-response relationship in this cohort
(Ex. 7-120, p. S25). The authors suggest that their estimates should be
checked or corrected " * * * with data coming from well-documented
industrial hygiene studies or industrial hygiene data banks including
information on the major relevant factors" (Ex. 7-120, p. S26). OSHA
believes that there is insufficient information to determine why a
clear relationship between Cr(VI) exposure and lung cancer is not
observed in the Gerin et al. study, but agrees with the authors that
exposure misclassification and the influence of background exposures
may explain this result.
EPRI noted the apparent lack of a relationship between exposure
duration and lung cancer risk in the Gerin et al. cohort (Ex. 38-8, p.
10). Duration of exposure is expected to show a relationship with
cancer risk if duration serves as a reasonable proxy for a measure of
exposure (e.g. cumulative exposure) that is related to risk. Since
cumulative exposure is equal to exposure duration multiplied by average
exposure level, duration of exposure may correlate reasonably well with
cumulative exposure if average exposure levels are similar across
workers, or if workers with longer employment tend to have higher
average exposure levels. In a cohort where exposure duration is
believed to correlate well with cumulative exposure, the absence of a
relationship between exposure duration and disease risk could be
interpreted as evidence against a relationship between cumulative
exposure and risk.
High variation in average exposures among workers, unrelated to the
duration of their employment, would tend to reduce the correlation
between exposure duration and cumulative exposure. If, as EPRI states,
Cr(VI) exposure depends strongly on process, base metal, and other work
conditions that vary from workplace to workplace, then duration of
exposure may not correlate well with cumulative exposure across the 135
companies included in the Gerin et al. study. The lack of a positive
relationship between exposure duration and lung cancer in the Gerin et
al. cohort may therefore signify that duration of exposure is not a
good proxy for the amount of exposure accumulated by workers, and
should not be interpreted as evidence against an exposure-response
relationship.
In post-hearing comments Mr. Robert Park of NIOSH discussed other
issues related to exposure duration in the Gerin et al. and other
welding cohorts:
Several factors may impact the interpretation of [the Gerin et
al. (1993) and Simonato et al. (1991) welder cohort studies] and are
consistent with an underlying risk associated with duration * * *.
The healthy worker survivor effect is a form of confounding in which
workers with long employment durations systematically diverge from
the overall worker population on risk factors for mortality. For
example, because smoking is a risk factor for disease, disability
and death, long duration workers would tend to have a lower smoking
prevalence, and hence lower expected rates of diseases that are
smoking related, like lung cancer. Not taking this into account
among welders might result in long duration welders appearing to
have diminished excess risk when, in fact, excess risk continues to
increase with time (Ex. 47-19-1, p. 6).
Mr. Park also emphasized the special importance of detailed information
for individual workers in multi-employer studies with exposure
conditions that vary widely across employers. He notes that high worker
turnover in highly exposed jobs " * * * could result in long duration
welding employment appearing to have lower risk than some shorter
duration [welding] employment when it does not" (Ex. 47-19-1, p. 6).
EPRI compared the risk of lung cancer among a subset of workers in
the Gerin cohort exposed to high cumulative levels of Cr(VI) to the
risk found among chromate production workers in the Gibb et al. and
Luippold et al. studies. "Focusing on the highest exposure group, SMRs
for the cohorts of stainless steel workers studied by Gerin et al
(1993) * * * range from 133 to 148 for exposures >1.5 mg-yrs/m\3\ * *
*. By comparison, the SMR from the Luippold et al. (2003) cohort is 365
for cumulative exposures of 1.0 to 2.69 mg-yrs/m\3\", a difference
that EPRI argues " * * * draws into question whether the exposure-
specific risk estimates from the chromate production industry can be
extrapolated to welders" (Ex. 38-8, p. 25). It is not clear why EPRI
chose to focus on the high exposure group, which had a minimum of 1.5
mg/m\3\-years cumulative Cr(VI) exposure, a mean of 2.5 mg/m\3\-years,
and no defined upper limit. Compared to the other exposure groups
described by Gerin et al., this group is likely to have had more
heterogenous exposure levels; may be expected to have a stronger
healthy worker effect due to the association between high cumulative
exposure and long employment history; and is the least comparable to
either workers exposed for a working lifetime at the proposed PEL (1
[mu]g/m\3\ * 45 years = 0.045 mg/m\3\-years cumulative exposure) or
welders in modern-day working conditions, who according to an IARC
review cited in EPRI's comments typically have exposure levels less
than 10 [mu]g/m\3\ (< 0.45 mg/m\3\-years cumulative exposure over 45
years) (Ex. 38-8, p. 4). In addition, the majority of the observation
time in the Luippold et al. cohort and the vast majority in the Gibb et
al. cohort is associated with exposure estimates lower than 1.5 mg/
m\3\-years Cr(VI) (Ex. 33-10, p. 455, Table 3; 25, p. 122, Table VI).
It should be noted that the levels of excess lung cancer risk
observed among welders in the Gerin et al. cohort and chromate
production workers in the Gibb and Luippold cohorts are quite similar
at lower cumulative exposure ranges that are more typical of Cr(VI)
exposures experienced in the cohorts. For example, the group of welders
with estimated cumulative exposures ranging from 50 to 500 [mu]g-yrs/
m\3\ has an SMR of 230. Chromate production workers from the Gibb and
Luippold cohorts with cumulative exposures within this range have
comparable SMRs, ranging from 184 to 234, as shown in Table VI-11
below. For reference, 45 years of occupational exposure at
approximately 1.1 [mu]g/m\3\ Cr(VI) would result in a cumulative
exposure of 50 [mu]g-yrs/m\3\; 45 years of occupational exposure at
approximately 11.1 [mu]g/m\3\ Cr(VI) would result in a cumulative
exposure of 500 [mu]g-yrs/m\3\.

OSHA performed an analysis comparing the risks predicted by OSHA's
models, based on the Gibb and Luippold data collected on chromate
production workers, with the lung cancer deaths reported for the
welders in the Gerin et al. study. Gerin et al. presented observed and
expected lung cancer deaths for four categories of cumulative exposure:
< 50 [mu]g-yrs/m\3\, 50-500 [mu]g-yrs/m\3\, 500-1500 [mu]g-yrs/m\3\, and
1500+ [mu]g-yrs/m\3\. The great majority of the Gerin et al. data on
stainless steel welders (98% of person-years) are in the highest three
categories, while the lowest category is extremely small (< 300 person-
years of observation). OSHA's preferred risk models (based on the Gibb
and Luippold cohorts) were used to predict lung cancer risk for each of
the three larger exposure categories. The OSHA predictions were derived
using the mean values from each exposure range, except for the open-
ended highest category, for which Gerin et al. reported a mean exposure
level of 2500 [mu]g-yrs/m\3\ (Ex. 7-120, p. S26). The ratio of
predicted to background lung cancer deaths, which approximately
characterizes the expected SMRs for these exposure groups, was
calculated for each group.
The OSHA model predictions were calculated assuming that workers
were first exposed to Cr(VI) at age 29, the average age at the start of
employment reported by Gerin et al. (Ex. 7-120, p. S26). The SMRs
reported by Gerin et al. were calculated for welders with at least five
years of employment and at least 20 years of follow-up. However, the
average duration of employment and follow-up was not evident from the
publication. The OSHA model predictions were therefore calculated using
a range of reasonable assumptions about the duration of employment over
which workers were exposed (5, 10, 15, and 20 years) and the length of
follow-up (30, 40, and 50 years).
Table VI-12 below presents the SMRs reported by Gerin et al. for
stainless steel welders in the three highest exposure categories,
together with the ratio of predicted to background lung cancer deaths
from OSHA's risk models. It should be noted that the ratio was
calculated using year 2000 U.S. lung cancer mortality rates, while the
SMRs reported by Gerin et al. were calculated using national lung
cancer mortality rates for the nine European countries represented in
the study (Ex. 7-114).

Table VI-12 shows that the range of risk ratios predicted by OSHA's
model is higher than the ratios reported for the highest exposure group
in the Gerin et al. cohort, consistent with EPRI's observations (Ex.
38-8, p. 25). However, the risk ratios predicted by OSHA's model are
consistent with the Gerin SMRs for the 500-1500 [mu]g-yrs/m\3\
cumulative exposure range. For the 50-500 [mu]g-yrs/m\3\ cumulative
exposure range, the OSHA prediction falls slightly below the lung
cancer mortality ratio observed for the Gerin et al. cohort. The OSHA
predictions for each group overlap with the 95% confidence intervals of
the Gerin et al. SMRs, suggesting that sampling error may partly
account for the discrepancies between the observed and predicted risk
ratios in the lowest and highest exposure groups.
As previously discussed, OSHA believes that the lack of a clear
exposure-response trend in the Gerin et al. study may be partly
explained by exposure misclassification. As shown in Table VI-12, the
highest exposure group has lower risk than might be expected based on
OSHA's preferred risk models, while the lowest exposure group appears
to have higher risk than OSHA's models would predict. This overall
pattern of generally elevated but non-increasing SMRs across the three
larger exposure groups in the Gerin study is consistent with
potentially severe exposure misclassification. The higher-than-
predicted risks among welders in the lowest exposure group could
similarly reflect misclassification. However, it is not possible to
determine with certainty that exposure misclassification is the cause
of the differences between the risk predicted by OSHA's model and that
observed in the Gerin cohort.
Finally, EPRI cites the generally similar relative risks found
among stainless steel and mild steel welders as further evidence that
exposure to Cr(VI) may not carry the same risk of lung cancer in
welding operations as it does in the chromate production industry. EPRI
states:
[I]t is reasonable to expect that if Cr(VI) were a relevant risk
factor for welders in the development of lung cancer, and certain
types of welding involve Cr(VI) more than other types, then
subgroups of welders who are more exposed to Cr(VI) by virtue of the
type of welding they do should have higher rates of lung cancer than
welders not exposed to Cr(VI) in their welding occupation;
in particular, " * * *stainless steel welders should have a higher
risk of lung cancer than welders of mild steel" (Ex. 38-8, p. 13).
OSHA believes that EPRI's point would be correct if the subgroups in
question are similar in terms of other important risk factors for lung
cancer, such as smoking, co-exposures, and overall population health.
However, no analysis comparing stainless steel welders with mild steel
welders has properly controlled for these factors, and in fact there
have been indications that mild steel welders may be at greater risk of
lung cancer than stainless steel welders from non-occupational causes.
As discussed by EPRI, "[r]esults from cohort studies of stainless
steel welders with SMRs much less than 100 support an argument that the
healthy worker effect might be more marked among stainless steel
workers compared to mild steel welders'; also " * * *stainless steel
welders are generally more qualified and paid more than other welders"
(Ex. 38-8, p. 16), a socioeconomic factor that suggests possible
differences in lung cancer risk due to smoking, community exposures, or
occupational exposures from employment other than welding.
Comments submitted by Exponent (Ex. 38-233-4) and EPRI (Ex. 38-8)
compare the Cr(VI) compounds found in welding fumes and those found in
the chromate production environments of the Gibb and Luippold cohorts.
Exponent stated that "[t]he forms of Cr(VI) to which chromate
production workers were historically exposed are primarily the soluble
potassium and sodium chromates" found in stainless steel welding
fumes. Less soluble forms of Cr(VI) are also found in stainless steel
welding fumes in limited amounts, as discussed in the 1990 IARC
monograph on welding (Ex. 35-242, p. 460), and are believed to have
been present in limited amounts at the plants where the Gibb and
Luippold workers were employed (Ex. 38-233-4, p. 4). Exponent concludes
that, while it is difficult to compare the exposures of welders to
chromate production workers, " * * *there is no obvious difference * *
* in solubility * * * " that would lead to a significantly lesser risk
from Cr(VI) exposure in welding as compared to the Gibb and Luippold
cohort exposures (Ex. 38-233-4, p. 3, p. 11). OSHA believes that the
similarity in the solubility of Cr(VI) exposures to welders and
chromate production workers supports the Agency's use of its risk model
to describe Cr(VI)-related risks to welders.
Exponent and others (Exs. 38-8; 39-25) commented on the possibility
that the bioavailability of Cr(VI) may nevertheless differ between
welders and chromate production workers, stating that " * * *
bioavailability of Cr(VI)-containing particles from welding fumes may
not be specifically related to solubility of the Cr(VI) chemical
species in the fume" (Ex. 38-233-4, p. 11). In this case, Exponent
argues,
delivered doses of Cr(VI) to the lung could be quite dissimilar
among welders as compared to chromate production industry workers
exposed to the same Cr(VI) chemical species at the same Cr(VI)
airborne concentrations (Ex. 38-233-4, p. 11).
However, Exponent provided no data or plausible rationale that would
support a Cr(VI) bioavailability difference between chromate production
and welding. The low proportion of respirable Cr(VI) particles that
apparently limits bioavailability of inhaled Cr(VI) during aircraft
spray priming operations described previously is not an issue with
welding. High temperature welding generates fumes of small
respirable-size Cr(VI) particles able to penetrate the bronchoalveolar
region of the lung. OSHA finds no evidence indicating that Cr(VI) from
welding is less bioavailable than Cr(VI) from soluble chromate
production.
In summary, OSHA agrees with EPRI and other commenters that
evidence of an exposure-response relationship is not as strong in
studies of Cr(VI)-exposed welders compared to studies of chromate
production workers. OSHA believes that the available welding studies
are less able to detect an exposure-response relationship, due to the
potentially severe exposure misclassification, occupational exposure to
other cancer causing agents, and the general lack of information with
which to control for any differences in background lung cancer risk
between Cr(VI)-exposed and unexposed welders. In contrast, the two
featured cohorts had sufficient information on workers' Cr(VI)
exposures and potential confounding exposures to support a reliable
exposure-response assessment. These are the primary factors that led
OSHA to determine (like EPRI and Exponent) that the Luippold and Gibb
cohorts are the best data available on which to base a model of
exposure-response between Cr(VI) and lung cancer (Exs. 38-8, p. 6; 38-
233-4, p. 1). Moreover, EPRI admitted that examination of " * * * the
forms of Cr(VI) to which welders are exposed, exposure concentrations,
and other considerations such as particle size * * * " identified " *
* * no specific basis * * * " for a difference in Cr(VI)-related lung
cancer risk among welders and the Gibb and Luippold chromate production
cohorts (Ex. 38-8, p. 7). OSHA concludes that it is reasonable and
prudent to estimate welders' risk using the exposure-response model
developed on the basis of the Gibb et al. and Luippold et al. datasets.
H. Conclusions
OSHA believes that the best quantitative estimates of excess
lifetime lung cancer risks are those derived from the data sets
described by Gibb et al. and Luippold et al. Both data sets show a
significant positive trend in lung cancer mortality with increasing
cumulative Cr(VI) exposure. The exposure assessments for these two
cohorts were reconstructed from air measurements and job histories over
three or four decades and were superior to those of other worker
cohorts. The linear relative risk model generally provided the best fit
among a variety of different models applied to the Gibb et al. and
Luippold et al. data sets. It also provided an adequate fit to three
additional data sets (Mancuso, Hayes et al., and Gerin et al.). Thus,
OSHA believes the linear relative risk model is the most appropriate
model to estimate excess lifetime risk from occupational exposure to
Cr(VI). Using the Gibb et al. and Luippold et al. datasets and a linear
relative risk model, OSHA concludes that the lifetime lung cancer risk
is best expressed by the three-to five-fold range of risk projections
bounded by the maximum likelihood estimates from the two featured data
sets. This range of projected risks is within the 95 percent confidence
intervals from all five data sets.
OSHA does not believe that it is appropriate to employ a threshold
dose-response approach to estimate cancer risk from a genotoxic
carcinogen, such as Cr(VI). Federal agencies, including OSHA, assume an
exposure threshold for cancer risk assessments to genotoxic agents only
when there is convincing evidence that such a threshold exists (see
e.g. EPA, Guidelines for Carcinogen Risk Assessment, March 2005, pp. 3-
21). In addition, OSHA does not consider absence of a statistically
significant effect in an epidemiologic or animal study that lacks power
to detect such effects to be convincing evidence of a threshold or
other non-linearity. OSHA also does not consider theoretical reduction
capacities determined in vitro with preparations that do not fully
represent physiological conditions within the respiratory tract to be
convincing evidence of a threshold. While physiological defense
mechanisms (e.g. extracellular reduction, DNA repair, apoptosis) can
potentially introduce dose transitions, there is no evidence of a
significantly non-linear Cr(VI) dose-lung cancer response in the
exposures of interest to OSHA. Finally, as previously discussed, linear
no-threshold risk models adequately fit the existing exposure-response
data.
The slightly soluble Cr(VI) compounds produced a higher incidence
of respiratory tract tumors than highly water soluble or highly water
insoluble Cr(VI) compounds in animal studies that tested Cr(VI)
compounds under similar experimental conditions. This likely reflects
the greater tendency for chromates of intermediate water solubility to
provide a persistent high local concentration of solubilized Cr(VI) in
close proximity to the target cell. Highly soluble chromates rapidly
dissolve and diffuse in the aqueous fluid lining the epithelia of the
lung and are more quickly cleared from the respiratory tract. Thus,
these chromates are less able to achieve the higher and more persistent
local concentrations within close proximity of the lung cell surface
than the slightly water soluble chromates. Water insoluble Cr(VI)
particulates are also able to come in close contact with the lung cell
surface but do not release readily absorbed chromate ions into the
biological environment as rapidly. OSHA concludes that slightly soluble
Cr(VI) compounds are likely to exhibit a greater degree of
carcinogenicity than highly water soluble or water insoluble Cr(VI)
when the same dose is delivered to critical target cells in the
respiratory tract of the exposed worker. OSHA also believes it
reasonable to regard water insoluble Cr(VI) to be of similar
carcinogenic potency to highly water soluble Cr(VI) compounds in the
absence of convincing scientific evidence to indicate otherwise.
The Gibb and Luippold cohorts were predominantly exposed to highly
water-soluble chromates, particularly sodium chromate and dichromate.
After evaluating lung cancer rates in other occupational cohort studies
with respect to the forms of Cr(VI) in the workplace, reliability in
the Cr(VI) exposure data, and the presence of potentially confounding
influences (e.g. smoking) and bias (e.g. healthy worker survivor bias)
as well as information on solubility, particle size, cell uptake, and
other factors influencing delivery of Cr(VI) to lung cells, OSHA finds
the risks estimated from the Gibb and Luippold cohorts adequately
represent risks to workers exposed to equivalent levels of Cr(VI)
compounds in other industries.
As with any risk assessment, there is some degree of uncertainty in
the projection of risks that results from the data, assumptions, and
methodology used in the analysis. The exposure estimates in the Gibb et
al. and Luippold et al. data sets relied, to some extent, on a paucity
of air measurements using less desirable sampling techniques to
reconstruct Cr(VI) exposures, particularly in the 1940s and 1950s.
Additional uncertainty is introduced when extrapolating from the cohort
exposures, which usually involved exposures to higher Cr(VI) levels for
shorter periods of time to an equivalent cumulative exposure involving
a lower level of exposure for a working lifetime. The study cohorts
consisted mostly of smokers, but detailed information on their smoking
behavior was unavailable. While the risk assessments make some
adjustments for the confounding effects of smoking, it is unknown
whether the assessments fully account for any interactive effects that
smoking and Cr(VI) exposure may have on
carcinogenic action. In any case, OSHA does not have reason to believe
the above uncertainties would introduce errors that would result in
serious overprediction or underprediction of risk.
OSHA's estimate of lung cancer risk from a 45 year occupational
exposure to Cr(VI) at the previous PEL of 52 [mu]g/m\3\ is 101 to 351
excess deaths per 1000 workers. This range, which is defined by maximum
likelihood estimates based on the Gibb and Luippold epidemiological
cohorts, is OSHA's best estimate of excess risk. It does not account
for statistical uncertainty, or for other potential sources of
uncertainty or bias. The wider range of 62 to 493 excess deaths per
1000 represents the statistical uncertainty associated with OSHA's
excess risk estimate at the previous PEL, based on lowest and highest
95% confidence bounds on the maximum likelihood estimates for the two
featured data sets. The excess lung cancer risks at alternative 8 hour
TWA PELs that were under consideration by the Agency were previously
shown in Table VI-7, together with the uncertainty bounds for the
primary and supplemental studies at these exposure concentrations. The
45-year exposure estimates satisfy the Agency's statutory obligation to
consider the risk of material impairment for an employee with regular
exposure to the hazardous agent for the period of his working life (29
U.S.C. 651 et seq.). Occupational risks from Cr(VI) exposure to less
than a full working lifetime are considered in Section VII on the
Significance of Risk and in Section VIII on the Benefits Analysis.
VII. Significance of Risk
In promulgating health standards, OSHA uses the best available
information to evaluate the risk associated with occupational
exposures, to determine whether this risk is severe enough to warrant
regulatory action, and to determine whether a new or revised rule will
substantially reduce this risk. OSHA makes these findings, referred to
as the "significant risk determination", based on the requirements of
the OSH Act and the Supreme Court's interpretation of the Act in the
"benzene" decision of 1980 (Industrial Union Department, AFL-CIO v.
American Petroleum Institute, 448 U.S. 607). The OSH Act directs the
Secretary of Labor to:
set the standard which most adequately assures, to the extent
feasible, on the basis of the best available evidence, that no
employee will suffer material impairment of health or functional
capacity even if such employee has regular exposure to the hazard *
* * for the period of his working life [6(b)(5)].
OSHA's authority to promulgate regulations to protect workers is
limited by the requirement that standards be "reasonably necessary and
appropriate to provide safe or healthful employment" [3(8)].
In the benzene decision, the Supreme Court's interpretation of
Section 3(8) further defined OSHA's regulatory authority. The Court
stated:
By empowering the Secretary to promulgate standards that are
"reasonably necessary or appropriate to provide safe or healthful
employment and places of employment," the Act implies that, before
promulgating any standard, the Secretary must make a finding that
the workplaces in question are not safe (IUD v. API 448 U.S. at
642).
"But 'safe' is not the equivalent of 'risk-free' ", the Court
maintained. "[T]he Secretary is required to make a threshold finding
that a place of employment is unsafe-in the sense that significant
risks are present and can be eliminated or lessened by a change in
practices" (IUD v. API, 448 U.S. at 642). It has been Agency practice
in regulating health hazards to establish this finding by estimating
risk to workers using quantitative risk assessment, and determining the
significance of this risk based on judicial guidance, the language of
the OSH Act, and Agency policy considerations.
The Agency has considerable latitude in defining significant risk
and in determining the significance of any particular risk. The Court
did not stipulate a means to distinguish significant from insignificant
risks, but rather instructed OSHA to develop a reasonable approach to
the significant risk determination. The Court stated that "it is the
Agency's responsibility to determine in the first instance what it
considers to be a 'significant' risk", and it did not express "any
opinion on the* * *difficult question of what factual determinations
would warrant a conclusion that significant risks are present which
make promulgation of a new standard reasonably necessary or
appropriate" (448 U.S. at 659). The Court also stated that, while
OSHA's significant risk determination must be supported by substantial
evidence, the Agency "is not required to support the finding that a
significant risk exists with anything approaching scientific
certainty" (448 U.S. at 656). Furthermore,
A reviewing court [is] to give OSHA some leeway where its
findings must be made on the frontiers of scientific knowledge [and]
* * * the Agency is free to use conservative assumptions in
interpreting the data with respect to carcinogens, risking error on
the side of overprotection rather than underprotection [so long as
such assumptions are based on] a body of reputable scientific
thought (448 U.S. at 655, 656).
To make the significance of risk determination for a new or
proposed standard, OSHA uses the best available scientific evidence to
identify material health impairments associated with potentially
hazardous occupational exposures, and, when possible, to provide a
quantitative assessment of exposed workers' risk of these impairments.
OSHA has reviewed extensive epidemiological and experimental research
pertaining to adverse health effects of occupational Cr(VI) exposure,
including lung cancer, and has established quantitative estimates of
the excess lung cancer risk associated with previously allowable Cr(VI)
exposure concentrations and the expected impact of the new PEL. OSHA
has determined that long-term exposure at the previous PEL would pose a
significant risk to workers' health, and that adoption of the new PEL
and other provisions of the final rule will substantially reduce this
risk.
A. Material Impairment of Health
As discussed in Section V of this preamble, there is convincing
evidence that exposure to Cr(VI) may cause a variety of adverse health
effects, including lung cancer, nasal tissue damage, asthma, and
dermatitis. OSHA considers these conditions to be material impairments
of health, as they are marked by significant discomfort and long-
lasting adverse effects, can have adverse occupational and social
consequences, and may in some cases have permanent or potentially life-
threatening consequences. Based on this finding and on the scientific
evidence linking occupational Cr(VI) to each of these effects, OSHA
concludes that exposure to Cr(VI) causes "material impairment of
health or functional capacity" within the meaning of the OSH Act.
1. Lung Cancer
OSHA considers lung cancer, an irreversible and frequently fatal
disease, to be a clear material impairment of health. OSHA's finding
that inhaled Cr(VI) causes lung cancer is based on the best available
epidemiological data, reflects substantial evidence from animal and
mechanistic research, and is consistent with the conclusions of other
government and public health organizations, including NIOSH, EPA,
ACGIH, NTP, and IARC (Exs. 35-117; 35-52; 35-158; 17-9-D; 18-3, p.
213). The Agency's primary evidence comes from two epidemiological
studies that show significantly increased incidence of lung cancer
among workers in the chromate production industry (Exs. 25; 33-10). The
high quality of the data collected in these studies and the analyses
performed on them has been confirmed by OSHA and by independent peer
review. Supporting evidence of Cr(VI) carcinogenicity comes from
occupational cohort studies in chromate production, chromate pigment
production, and chromium plating, and by cell culture research into the
processes by which Cr(VI) disrupts normal gene expression and
replication. Studies demonstrating uptake, metabolism, and genotoxicity
of a variety of soluble and insoluble Cr(VI) compounds support the
Agency's position that all Cr(VI) compounds should be regulated as
occupational carcinogens (Exs. 35-148; 35-68; 35-67; 35-66; 12-5; 35-
149; 35-134).
2. Non-Cancer Impairments
While OSHA has relied primarily on the association between Cr(VI)
inhalation and lung cancer to demonstrate the necessity of the
standard, the Agency has also determined that several other material
health impairments can result from exposure to airborne Cr(VI). As
shown in several cross-sectional and cohort studies, inhalation of
Cr(VI) can cause ulceration of the nasal passages and perforation of
the nasal septum (Exs. 35-1; 7-3; 9-126; 35-10; 9-18; 3-84; 7-50; 31-
22-12). Nasal tissue ulcerations are often accompanied by swelling and
bleeding, heal slowly, and in some cases may progress to a permanent
perforation of the nasal septum that can only be repaired surgically.
Inhalation of Cr(VI) may also lead to asthma, a potentially life-
threatening condition in which workers become allergic to Cr(VI)
compounds and experience symptoms such as coughing, wheezing, and
difficulty in breathing upon exposure to small amounts of airborne
Cr(VI). Several case reports have documented asthma from Cr(VI)
exposure in the workplace, supporting Cr(VI) as the sensitizing agent
by bronchial challenge (Exs. 35-7; 35-12; 35-16; 35-21).
During the comment period, NIOSH requested that OSHA consider
allergic contact dermatitis (ACD) as a material impairment of health
due to occupational exposure to Cr(VI). NIOSH reasoned:
Dermal exposure to Cr(VI) through skin contact * * * may lead to
sensitization or allergic contact dermatitis. This condition, while
not life-threatening, is debilitating and marked by significant
discomfort and long-lasting adverse effects; it can have adverse
occupational and social consequences and should be a material
impairment to the health of affected workers * * * Including
allergic contact dermatitis in OSHA's determination of material
impairment of health draws attention to the fact that Cr(VI) is both
a dermal exposure hazard and an inhalation hazard, and alerts
employers that they should seek to minimize exposure to both routes
(Ex. 40-10-2, p. 3)
OSHA fully agrees with the NIOSH comment. There is strong evidence
that unprotected skin contact with Cr(VI)-containing materials and
solutions can cause ACD as well as irritant dermatitis and skin
ulceration (see section V.D). ACD is a delayed hypersensitivity
response. The worker initially becomes sensitized to Cr(VI) following
dermal exposure. Once a worker becomes sensitized, brief exposures to
small amounts of Cr(VI) can trigger symptoms such as redness, swelling,
itching, and scaling. ACD is characterized by the initial appearance of
small raised papules that can later develop into blisters and dry
thickened, cracked skin. The allergic condition is persistent, causing
some workers to leave their jobs (Ex. 35-320). Symptoms of ACD
frequently continue long after occupational exposure to Cr(VI) ends,
since sensitized individuals can react to contact with Cr(VI) in
consumer products and other non-occupational sources.
Skin exposure to Cr(VI) compounds can also cause a non-allergic
form of dermatitis. This skin impairment results from direct contact
with Cr(VI) doses that damage or irritate the skin, but do not involve
immune sensitization. This form of dermatitis can range from mild
redness to severe burns and ulcers, known as "chrome holes", that
penetrate deep into tissues. Once the worker is removed from exposure,
the skin ulcers heal slowly, often with scarring.
B. Risk Assessment
When possible, epidemiological or experimental data and statistical
methods are used to characterize the risk of disease that workers may
experience under the currently allowable exposure conditions, as well
as the expected reduction in risk that would occur with implementation
of the new PEL. The Agency finds that the available epidemiological
data are sufficient to support quantitative risk assessment for lung
cancer among Cr(VI)-exposed workers. Using the best available studies,
OSHA has identified a range of expected risk from regular occupational
exposure at the previous PEL (101-351 excess lung cancer deaths per
1000 workers) and at the new PEL of 5 [mu]g/m\3\ (10-45 per 1000
workers), assuming a working lifetime of 45 years' exposure in each
case. These values represent the best estimates of multiple analysts
working with data from two extensively studied worker populations, and
are highly consistent across analyses using a variety of modeling
techniques and assumptions. While some attempts have been made to
assess the relationship between Cr(VI) exposure level and noncancer
adverse health effects, the Agency does not believe that a reliable
quantitative risk assessment can be performed for noncancer effects at
this time, and has therefore characterized noncancer risk
qualitatively.
For estimates of lung cancer risk from Cr(VI) exposure, OSHA has
relied upon data from two cohorts of chromate production workers. The
Gibb cohort, which originates from a chromate production facility in
Baltimore, Maryland, includes 2357 workers who began work between 1950
and 1974 and were followed up through 1992 (Ex. 33-11). The extensive
exposure documentation available for this cohort, the high statistical
power afforded by the large cohort size, and the availability of
information on individual workers' race and smoking status provide a
strong basis for risk analysis. The Luippold cohort, from a facility in
Painesville, Ohio, includes 482 workers who began work between 1940 and
1972, worked for at least one year at the plant, and were followed up
through 1997 (Ex. 33-10). This cohort also provides a strong basis for
risk analysis, in that it has high-quality documentation of worker
Cr(VI) exposure and mortality, a long period of follow-up, and a large
proportion of relatively long-term employees (55% were employed for
longer than 5 years).
1. Lung Cancer Risk Based on the Gibb Cohort
Risk assessments were performed on the Gibb cohort data by Environ
International Corporation (Ex. 33-12), under contract with OSHA; Park
et al., as part of an ongoing effort by NIOSH (Ex. 33-13); and Exponent
on behalf of the Chrome Coalition (Ex. 31-18-15-1). A variety of
statistical models were considered, allowing OSHA to identify the most
appropriate models and assess the resulting risk estimates' sensitivity
to alternate modeling approaches. Models were tried with additive and
relative risk assumptions; various exposure groupings and lag times;
linear and nonlinear exposure-response functions; external and internal
standardization; reference lung cancer rates from city-, state-, and
national-level data; inclusion and exclusion of short-term workers; and
a variety of ways to control for the effects of smoking. OSHA's
preferred approach, a relative risk model using Baltimore lung cancer
reference rates, and NIOSH's preferred approach, a relative risk model
using detailed smoking information and U.S. lung cancer reference
rates, are among several models that use reasonable assumptions and
provide good fits to the data. As discussed in section VI, the Environ,
Park et al., and linear Exponent models yield similar predictions of
excess risk from exposure at the previous PEL and the new PEL (see
Tables VI-2 and VI-3). OSHA's preferred models (from the Gibb data set)
predict about 300-350 excess lung cancers per 1000 workers exposed for
a working lifetime of 45 years at the previous PEL and about 35-45
excess lung cancers per 1000 workers at the new PEL of 5 [mu]g/m\3\.
Environ and Crump et al. performed risk assessments on the Luippold
cohort, exploring additive and relative risk models, linear and
quadratic exposure-response functions, and several exposure groupings
(Exs. 35-59; 35-58). Additive and relative risk models by both analyst
groups fit the data adequately with linear exposure-response. All
linear models predicted similar excess risks, from which OSHA has
selected preferred estimates based on the Crump et al. analysis of
about 100 excess lung cancer deaths per 1000 workers exposed for 45
years at the previous PEL, and ten excess lung cancer deaths per 1000
workers at the new PEL.
2. Lung Cancer Risk Based on the Luippold Cohort
The risk assessments performed on the Luippold cohort yield
somewhat lower estimates of lung cancer risk than those performed on
the Gibb cohort. This discrepancy is probably not due to statistical
error in the risk estimates, as the confidence intervals for the
estimates do not overlap. The risk estimates based on the Gibb and
Luippold cohorts are nonetheless reasonably close. OSHA believes that
both cohorts support reasonable estimates of lung cancer risk, and
based on their results has selected a representative range of 101-351
per 1000 for 45 years' occupational exposure at the previous PEL and
10-45 per 1000 for 45 years' occupational exposure at the new PEL for
the significant risk determination. OSHA's confidence in these risk
estimates is further strengthened by the results of the independent
peer review to which the risk assessment was submitted, which supported
the Agency's approach and results. OSHA also received several comments
in support of its risk estimates (Exs. 44-7, 38-222; 39-73-1). A full
analysis of major comments on the results of OSHA's quantitative risk
assessment can be found in section VI.F.
3. Risk of Non-Cancer Impairments
Although nasal damage and asthma may be associated with
occupational exposure to airborne Cr(VI), OSHA has determined that
there are insufficient data to support a formal quantitative risk
assessment for these effects. Available occupational studies of Cr(VI)-
induced nasal damage are either of cross-sectional study design, do not
provide adequate data on short-term airborne Cr(VI) exposure over an
entire employment period, or do not account for possible contribution
from hand-to-nose transfer of Cr(VI) (Exs. 31-22-12; 9-126; 35-10; 9-
18). Occupational asthma caused by Cr(VI) has been documented in
clinical case reports but asthma occurrence has not been linked to
specific Cr(VI) exposures in a well-conducted epidemiological
investigation. The Agency has nonetheless made careful use of the best
available scientific information in its evaluation of noncancer health
risks from occupational Cr(VI) exposure. In lieu of a quantitative
analysis linking the risk of noncancer health effects, such as damage
to nasal tissue, with specific occupational exposure conditions, the
Agency has qualitatively considered information on the extent of these
effects and occupational factors affecting risk, as discussed below.
Damage to the nasal mucosa and septum can occur from inhalation of
airborne Cr(VI) or transfer of Cr(VI) on workers' hands to the interior
of the nose. Epidemiological studies have found varying, but
substantial, prevalence of nasal damage among workers exposed to high
concentrations of airborne Cr(VI). In the cohort of 2357 chromate
production workers studied by Gibb et al., over 60% experienced nasal
tissue ulceration at some point during their employment, with half of
these workers' first ulcerations occurring within 22 days from the date
they were hired (Ex. 31-22-12). The authors found a statistically
significant relationship between nasal ulceration and workers'
contemporaneous exposures, with about half of the workers who developed
ulcerations first diagnosed while employed in a job with average
exposure concentrations greater than 20 [mu]g/m3. Nasal
septum perforations were reported among 17% of the Gibb cohort workers,
and developed over relatively long periods of exposure (median time 172
days from hire date to diagnosis).
A high prevalence of nasal damage was also found in a study of
Swedish chrome platers (Ex. 9-126). Platers exposed to average 8-hour
Cr(VI) concentrations above 2 [mu]g/m3 with short-term
excursions above 20 [mu]g/m3 from work near the chrome bath
had a nearly 50 percent prevalence (i.e. 11 out of 24 workers) of nasal
ulcerations and septum perforations. These data, along with that from
the Gibb cohort, suggest a substantial and clearly significant risk of
nasal tissue damage from regular short-term exposures above 20 [mu]g/
m3. More than half of the platers (i.e. 8 of 12 subjects)
with short-term excursions to somewhat lower Cr(VI) concentrations
between 2.5 and 11 [mu]g/m3 had atrophied nasal mucosa (i.e.
cellular deterioration of the nasal passages) but not ulcerations or
perforations. This high occurrence of nasal atrophy was substantially
greater than found among the workers with mean Cr(VI) levels less than
2 [mu]g/m3 (4 out of 19 subjects) and short-term Cr(VI)
exposures less than 1 [mu]g/m3 (1 of 10 subjects) or among
the office workers not exposed to Cr(VI) (0 of 19 subjects). This
result is consistent with a concentration-dependant gradation in
response from relatively mild nasal tissue atrophy to the more serious
nasal tissue ulceration with short-term exposures to Cr(VI) levels
above about 10 [mu]g/m3. For this reason, OSHA believes
short-term Cr(VI) exposures regularly exceeding about 10 [mu]g/
m3 may still result in a considerable risk of nasal
impairment. However, the available data do not allow a precise
quantitative estimation of this risk.
While dermal exposure to Cr(VI) can cause material impairment to
the skin, a credible quantitative assessment of the risk is not
possible because few occupational studies have measured the amounts of
Cr(VI) that contact the skin during job activities; studies rarely
distinguish dermatitis due to Cr(VI) from other occupational and non-
occupational sources of dermatitis; and immune hypersensitivity
responses, such as ACD, have an exceedingly complex dose-response.
C. Significance of Risk and Risk Reduction
The Supreme Court's benzene decision of 1980 states that "before
he can promulgate any permanent health or safety standard, the
Secretary [of Labor] is required to make a threshold finding that a
place of employment is unsafe--in the sense that significant risks are
present and can be eliminated or lessened by a change in practices"
(IUD v. API, 448 U.S. at 642). The Court broadly describes the range of
risks OSHA might determine to be significant:
It is the Agency's responsibility to determine in the first
instance what it considers to be a "significant" risk. Some risks
are plainly acceptable and others are plainly unacceptable. If, for
example, the odds are one in a billion that a person will die from
cancer by taking a drink of chlorinated water, the risk clearly
could not be considered significant. On the other hand, if the odds
are one in a thousand that regular inhalation of gasoline vapors
that are 2 percent benzene will be fatal, a reasonable person might
well consider the risk significant and take the appropriate steps to
decrease or eliminate it. (IUD v. API, 448 U.S. at 655).
The Court further stated, "The requirement that a "significant" risk
be identified is not a mathematical straitjacket * * *. Although the
Agency has no duty to calculate the exact probability of harm, it does
have an obligation to find that a significant risk is present before it
can characterize a place of employment as "unsafe"' and proceed to
promulgate a regulation (IUD v. API, 448 U.S. at 655).
Table VII-1 presents the estimated excess risk of lung cancer
associated with various levels of Cr(VI) exposure allowed under the
current rule, based on OSHA's risk assessment and assuming either 20
years' or 45 years' occupational exposure to Cr(VI) as indicated. The
purpose of the OSH Act, as stated in Section 6(b), is to ensure "that
no employee will suffer material impairment of health or functional
capacity even if such employee has regular exposure to the hazard * * *
for the period of his working life." 29 U.S.C. 655(b)(5). Taking a 45-
year working life from age 20 to age 65, as OSHA has always done in
significant risk determinations for previous standards, the Agency
finds an excess lung cancer risk of approximately 100 to 350 per 1000
workers exposed at the previous PEL of 52 [mu]g/m3 Cr(VI).
This risk is clearly significant, falling well above the level of risk
the Supreme Court indicated a reasonable person might consider
acceptable. Even assuming only a 20-year working life, the excess risk
of about 50 to 200 per 1000 workers is still clearly significant. The
new PEL of 5 [mu]g/m3 Cr(VI) is expected to reduce these
risks substantially, to below 50 excess lung cancers per 1000 workers.
However, even at the new PEL, the risk posed to workers with a lifetime
of regular exposure is still clearly significant.

Workers exposed to concentrations of Cr(VI) lower than the new PEL
and for shorter periods of time may also have significant excess cancer
risk. The Agency's risk estimates are roughly proportional to duration
for any given exposure concentration. The estimated risk to workers
exposed at any fixed concentration for 10 years is about one-half the
risk to workers exposed for 20 years; the risk for five years' exposure
is about one-fourth the risk for 20 years. For example, about 11 to 55
out of 1000 workers exposed at the previous PEL for five years are
expected to develop lung cancer as a result of their exposure. Those
exposed to 10 [mu]g/m3 Cr(VI) for 5 years have an estimated
excess risk of about 2-12 lung cancer deaths per 1000 workers. It is
thus not only workers exposed for many years at high levels who have
significant cancer risk under the old standard; even workers exposed
for shorter periods at levels below the previous PEL are at substantial
risk, and will benefit from implementation of the new PEL.
To further demonstrate significant risk, OSHA compares the risk
from currently permissible Cr(VI) exposures to risks found across a
broad variety of occupations. The Agency has used similar occupational
risk comparisons in the significant risk determination for substance-
specific standards promulgated since the benzene decision. This
approach is supported by evidence in the legislative record that
Congress intended the Agency to regulate unacceptably severe
occupational hazards, and not "to establish a utopia free from any
hazards"(116 Cong. Rec. 37614 (1970), Leg. Hist 480), or to address
risks comparable to those that exist in virtually any occupation or
workplace. It is also consistent with Section 6(g) of the OSH Act,
which states:
In determining the priority for establishing standards under
this section, the Secretary shall give due regard to the urgency of
the need for mandatory safety and health standards for particular
industries, trades, crafts, occupations, businesses, workplaces or
work environments.
Fatal injury rates for most U.S. industries and occupations may be
obtained from data collected by the Bureau of Labor Statistics. Table
VII-2 shows average annual fatality rates per 1000 employees for
several industries between 1992 and 2001, as well as projected
fatalities per 1000 employees for periods of 20 and 45 years based on
these annual rates (Ex. 35-305). While it is difficult to compare
aggregate fatality rates meaningfully to the risks estimated in the
quantitative risk assessment for Cr(VI), which target one specific
hazard (inhalation exposure to Cr(VI)) and health outcome (lung
cancer), these rates provide a useful frame of reference for
considering risk from Cr(VI) inhalation. Regular exposures at high
levels, including the previous PEL of 52 [mu]g/m3 Cr(VI),
are expected to cause substantially more deaths per 1000 workers from
lung cancer than result from occupational injuries in most private
industry. At the new PEL of 5 [mu]g/m3 Cr(VI) the Agency's
estimated range of excess lung cancer mortality overlaps the fatality
risk for mining and approaches that for construction, but still clearly exceeds
the risk in lower-risk industries such as manufacturing.

Because there is little available information on the incidence of
occupational cancer, risk from Cr(VI) exposure cannot be compared with
overall risk from other workplace carcinogens. However, OSHA's previous
risk assessments provide estimates of risk from exposure to certain
carcinogens. These risk assessments, like the current assessment for
Cr(VI), were based on animal or human data of reasonable or high
quality and used the best information then available. Table VII-3 shows
the Agency's best estimates of cancer risk from 45 years' occupational
exposure to several carcinogens, as published in the preambles to final
rules promulgated since the benzene decision in 1980.
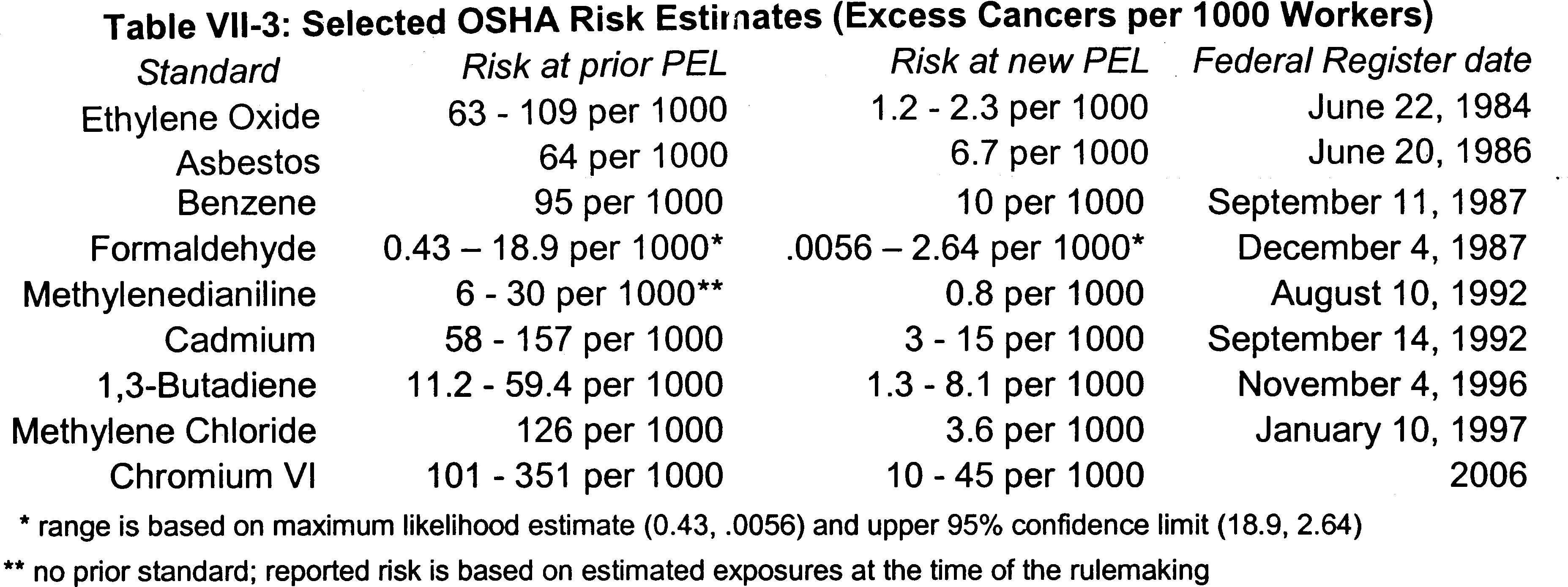
The Cr(VI) risk estimate at the previous PEL is higher than many
risks the Agency has found to be significant in previous rules (Table
VII-3, "Risk at Previous PEL"). The estimated risk from lifetime
occupational exposure to Cr(VI) at the new PEL is 10-45 excess lung
cancer deaths per 1000 workers, a range which overlaps the estimated
risks from exposure at the current PELs for benzene and cadmium (Table
VII-3, "Risk at new PEL").
Based on the results of the quantitative risk assessment, the
Supreme Court's guidance on acceptable risk, comparison with rates of
occupational fatality in various industries, and comparison with cancer
risk estimates developed in previous rules, OSHA finds that the risk of
lung cancer posed to workers under the previous permissible level of
occupational Cr(VI) exposure is significant. The new PEL of 5 is
expected to reduce risks to workers in Cr(VI)-exposed occupations
substantially (by about 8- to 10-fold). OSHA additionally finds that
nasal tissue ulceration and septum perforation can occur under exposure
conditions allowed by the previous PEL leading to an additional health
risk beyond the significant lung cancer risk present. The reduction of
the Cr(VI) PEL from 52 [mu]g/m3 to 5 [mu]g/m3 is
expected to substantially reduce workers' risk of nasal tissue damage.
With regard to dermal effects from Cr(VI) exposure, OSHA believes that
provision of appropriate protective clothing and adherence to
prescribed hygiene practices will serve to protect workers from the
risk of Cr(VI)-induced skin impairment.
VIII. Summary of the Final Economic and Regulatory Flexibility Analysis
A. Introduction
OSHA's Final Economic and Regulatory Flexibility Analysis (FEA)
addresses issues related to the costs, benefits, technological and
economic feasibility, and economic impacts (including small business
impacts) of the Agency's Occupational Exposure to Hexavalent Chromium
rule. The full Final Economic and Regulatory Flexibility Analysis has
been placed in the docket as Ex. 49. The analysis also evaluates
alternatives that were considered by the agency before adopting the final
rule. This rule is an economically significant rule under Section 3(f)(1)
of Executive Order 12866 and has been reviewed by the Office of Information and
Regulatory Affairs in the Office of Management and Budget, as required
by executive order. The purpose of this Final Economic and Regulatory
Flexibility Analysis is to:
Identify the establishments and industries potentially
affected by the final rule;
Estimate current exposures and the technologically
feasible methods of controlling these exposures;
Estimate the benefits of the rule in terms of the
reduction in lung cancer and dermatoses employers will achieve by
coming into compliance with the standard;
Evaluate the costs and economic impacts that
establishments in the regulated community will incur to achieve
compliance with the final standard;
Assess the economic feasibility of the rule for affected
industries; and
Evaluate the principal regulatory alternatives to the
final rule that OSHA has considered.
The full Final Economic Analysis contains the following chapters:
Chapter I. Introduction
Chapter II. Industrial Profile
Chapter III. Technological Feasibility
Chapter IV. Costs of Compliance
Chapter V. Economic Impacts
Chapter VI. Benefits and Net Benefits
Chapter VII. Final Regulatory Flexibility Analysis
Chapter VIII. Environmental Impacts
Chapter IX. Assessing the Need for Regulation.
These chapters are summarized in sections B to H of this Preamble
summary.
B. Introduction and Industrial Profile (Chapters I and II)
The final standard for occupational exposure to hexavalent chromium
was developed by OSHA in response to evidence that occupational
exposure to Cr(VI) poses a significant risk of lung cancer, nasal
septum ulcerations and perforations, and dermatoses. Exposure to Cr(VI)
may also lead to asthma. To protect exposed workers from these effects,
OSHA has set a Permissible Exposure Limit (PEL) of 5 [mu]g/m\3\
measured as an 8-hour time weighted average. OSHA also examined
alternative PELs ranging from 20 [mu]g/m\3\ to 0.25 [mu]g/m\3\ measured
as 8-hour time weighted averages.
OSHA's final standards for occupational exposure to Cr(VI) are
similar in format and content to other OSHA health standards
promulgated under Section 6(b)(5) of the Act. In addition to setting
PELs, the final rule requires employers to:
Monitor the exposure of employees (though allowing a
performance-oriented approach to monitoring);
Establish regulated areas when exposures may reasonably be
expected to exceed the PEL (except in shipyards and construction);
Implement engineering and work practice controls to reduce
employee exposures to Cr(VI);
Provide respiratory protection to supplement engineering
and work practice controls where those controls are not feasible, where
such controls are insufficient to meet the PEL, or in emergencies;
Provide other protective clothing and equipment as
necessary for dermal protection;
Make industrial hygiene facilities (hand washing stations)
available in some situations;
Provide medical surveillance when employees are exposed
above the action level for 30 days or more;
Train workers about the hazards of Cr(VI) (including
elements already required by OSHA's Hazard Communication Standard); and
Keep records related to the standard.
The contents of the standards, and the reasons for issuing separate
standards for general industry, construction and shipyard employment,
are more fully discussed in the Summary and Explanation section of this
Preamble.
Chapter II of the full FEA describes the uses of Cr(VI) and the
industries in which such uses occur. Employee exposures are defined in
terms of "application groups," i.e., groups of firms where employees
are exposed to Cr(VI) when performing a particular function. This
methodology is appropriate to exposure to Cr(VI) where a widely used
chemical like chromium may lead to exposures in many kinds of firms in
many industries but the processes used, exposures generated, and
controls needed to achieve compliance may be the same. For example,
because a given type of welding produces Cr(VI) exposures that are
essentially the same regardless of whether the welding occurs in a
ship, on a construction site, as part of a manufacturing process, or as
part of a repair process, it is appropriate to analyze such processes
as a group. However, OSHA's analyses of costs and economic feasibility
reflect the fact that baseline controls, ease of implementing ancillary
provisions, and the economic situation of the employer may differ
within different industries in an application group.
The most common sources of occupational exposure to Cr(VI), in
addition to the production and use of chromium metal and chromium metal
alloys, are chromium electroplating; welding of metals containing
chromium, particularly stainless steel or other high-chromium steels,
or with chromium coatings; and the production and use of Cr(VI)-
containing compounds, particularly Cr(VI) pigments, but also Cr(VI)
catalysts, chromic acid, and the production of chromium-containing
pesticides.
Some industries are seeing a sharp decline in chromium use.
However, many of the industries that are seeing a sharp decline have
either a small number of employees or have low exposure levels (e.g.,
wood working, printing ink manufacturers, and printing). In the case of
lead chromate in pigment production, OSHA's sources indicate that there
is no longer domestic output containing lead chromates. Therefore, this
trend has been recognized in the FEA. Painting activities in general
industry primarily involve the application of strontium chromate
coatings to aerospace parts; these exposures are likely to continue
into the foreseeable future. Similarly, removal of lead chromate paints
in construction and maritime is likely to present occupational risks
for many years.
In application groups where exposures are particularly significant,
both in terms of workforce size and exposure levels--notably in
electroplating and welding--OSHA anticipates very little decline in
exposures to hexavalent chromium due to the low potential for
substitution in the foreseeable future.
OSHA has made a number of changes to the industrial profile of the
application groups as a result of comments on the proposed rule. Among
the most important are:
Additions to the electroplating application group to
include such processes as chrome conversion, which were not considered
at the time of the proposal;
Additions to the painting application group to cover
downstream users, particularly automobile repair shops and construction
traffic painting;
Additions to glass manufacturing to cover fiberglass, flat
glass, and container glass industries;
Addition of the forging industry;
Addition of the ready mixed concrete industry;
Additions to the welding application group to include
welding on low-chromium steel and increase the estimated number of
exposed workers in the maritime sector; and
More careful division of the many different industries in
which electroplating, welding and painting may appear as applications.
Table VIII-1 shows the application groups analyzed in OSHA's FEA,
as well as the industries in each application group, and for each
provides the number of establishments affected, the number of employees
working in those establishments, the number of entities (firms or
governments) fitting SBA's small business criteria for the industry,
and the number of employees in those firms. (The table shows data for
both establishments and entities--defined as firms or governments. An
entity may own more than one establishment.) The table also shows the
revenues of affected establishment and entities, updated to reflect
2002 data. (This table provides the latest available data at the time
this analysis was produced.) As shown in the table, there are a total
of 52,000 establishments affected by the final standard.
Various types of welding applications account for the greatest
number of establishments and number of employees affected by the final
standard.
BILLING CODE 4510-26-P















BILLING CODE 4510-26-C
Table VIII-2 shows the current exposures to Cr(VI) by application
group. The exposure data relied on by OSHA in developing the exposure
profile and evaluating technological feasibility were compiled in a
database of exposures taken from OSHA compliance officers, site visits
by OSHA contractors and the National Institute for Occupational Safety
and Health (NIOSH), the U.S. Navy, published literature, commenters on
the proposed rule and other interested parties.
It is also important to note that Table VIII-2 and OSHA's cost and
feasibility analyses reflect the full range of exposures occurring in
each application group, not the median exposures. Some commenters
(e.g., Ex. 47-27-1) misunderstood this and believed OSHA determined
that only employers with median exposures above the PEL would incur
costs for engineering and work practice controls. OSHA did not use
exposure medians to assign compliance costs in this rulemaking. OSHA
made limited use of exposure medians for only a few purposes. The first
was in the analysis of baseline controls, described in the
technological feasibility discussion below. Where both exposure data
and information on the controls in place were available, OSHA used the
median exposure level experienced in the presence of a specific type of
control to assign an effectiveness level to the control. Second, to
determine whether to assume baseline controls were already in place in
cases where OSHA only had exposure data available, it compared median
exposure levels to the median exposure levels previously assigned to
baseline controls.
BILLING CODE 4510-26-P










BILLING CODE 4510-26-C
In all sectors OSHA has used the best available information to
determine baseline exposures and technological feasibility. Throughout
the rulemaking process OSHA requested industry-specific information.
These requests included site visits, discussions with industry experts
and trade associations, the 2002 Request for Information (RFI), and the
SBREFA process. These requests continued through the proposal and the
public hearing process where OSHA continued to request information.
OSHA reviewed all the data submitted to the record and where
appropriate updated the exposure profile. For exposure information to
be useful in the profile, only individual personal exposures
representing a full shift were used.
As noted earlier, OSHA used a variety of sources to obtain
information about exposures in each application group. These sources
include: NIOSH Health Hazard Evaluations (HHEs), OSHA's Integrated
Management Information System (IMIS) exposure data, data from other
government agencies, published literature, OSHA/NIOSH site visits,
discussions with industry experts and trade associations, and data
submitted to the OSHA record. In some instances OSHA's contractor had
difficulty obtaining permission to perform site visits in a specific
application group. For instance, OSHA's contractor could obtain
permission to conduct a site visit only at a steel mill that used the
teeming and primary rolling method--in contrast to continuous casting,
now used in approximately 95 percent of steel mills. In these few
cases, OSHA acknowledged these potential problems and OSHA (or its
contractor) discussed its concerns with industry experts and used their
professional judgment to determine technological feasibility.
In response to the exposure data submitted to the record OSHA has
made the following major changes to the exposure profile:
Electroplating--Revised the exposure distribution for hard
chrome electroplating to use only the more-detailed exposure data from
site visits and other NIOSH reports.
Welding--In construction, OSHA used exposure data from the
maritime sector for analogous operations to supplement the exposure
profile. Added additional exposure data to the profile as provided to
the record.
Painting--Revised the exposure profile to reflect the
additional aerospace exposure data submitted to the record.
Steel Mills--Revised the exposure profile to reflect
additional exposure data supplied to the record; welders were added
directly to this application group.
Chromium Catalyst Users--Revised the exposure profile
based on additional exposure data from a NIOSH HHE.
Wood working--Added information from the record.
Construction--Revised the exposure profile to reflect the
additional exposure information submitted to the record.
Detailed information on the changes made in the exposure profile
for each application group can be found in Chapter III of the Final
Economic Analysis.
OSHA's analysis of technological feasibility examined employee
exposures at the operation or task level to the extent that such data
were available. There are approximately 558,000 workers exposed to
Cr(VI), of which 352,000 are exposed above 0.25 micrograms per cubic
meter and 68,000 above the PEL of 5 micrograms per cubic meter.
C. Technological Feasibility
In Chapter III of OSHA's FEA, OSHA assesses the current exposures
and the technological feasibility of the final standard in all affected
industry sectors. The analysis presented in this chapter is organized
by application group and analyzes employee exposures at the operation
or task level to the extent that such data are available. Accordingly,
OSHA collected exposure data at the operation or task level to identify
the Cr(VI)-exposed workers or job operations that need to improve their
process controls to achieve exposures at or below the PEL. In the few
instances where there were insufficient exposure data, OSHA used
analogous operations to characterize these operations.
In general, OSHA considered the following kinds of controls that
could reduce employee exposures to Cr(VI): local exhaust ventilation
(LEV), which could include maintenance or upgrade of the current local
exhaust ventilation or installation of additional LEV; process
enclosures that would isolate the worker from the exposure; process
modifications that would reduce the generation of Cr(VI) dust or fume
in the work place; improved general dilution ventilation including
assuring that adequate make-up air is supplied to the work place;
improved housekeeping; improved work practices; and the supplemental
use of respiratory protection if engineering and work practice controls
were not sufficient to meet the PEL.
The technologies used in this analysis are commonly known, readily
available and are currently used to some extent in the affected
industries and processes. OSHA's assessment of feasible controls and
the exposure levels they can achieve is based on information collected
by Shaw Environmental, Inc. (Ex. 50), a consultant to OSHA, on the
current exposure levels associated with existing controls, on the
availability of additional controls needed to reduce employee
exposures, and on other evidence presented in the docket.
Through the above analysis, OSHA finds that a PEL of 5 [mu]g/m\3\
is technologically feasible for most operations in all affected
industries through the use of engineering and work practice controls.
As discussed further below, the final rule requires that when painting
of aircraft or large aircraft parts is performed in the aerospace
industry, the employer is only required to use engineering and work
practice controls to reduce employee exposures to Cr(VI) to or below 25
[mu]g/m\3\. The employer must then use respiratory protection to
achieve the PEL. Apart from this limited exception, all other
industries can achieve the PEL with only minimal reliance on
respiratory protection. Table VIII-3 shows OSHA's estimate of
respirator use by industry for each of the PELs that OSHA considered.
At the final PEL of 5 [mu]g/m\3\, only 3.5 percent of exposed employees
will be required to use respirators.
In only three sectors will respirator use be required for more than
5 percent of exposed employees. In two of these sectors, chromate
pigment producers and chromium dye producers, use of respirators will
be intermittent. The third sector, stainless steel welding, presents
technological challenges in certain environments such as confined
spaces. OSHA has concluded that, with a few limited exceptions which
are discussed below, employers will be able to reduce exposures to the
PEL through the use of engineering and work practice controls.
BILLING CODE 4510-26-P

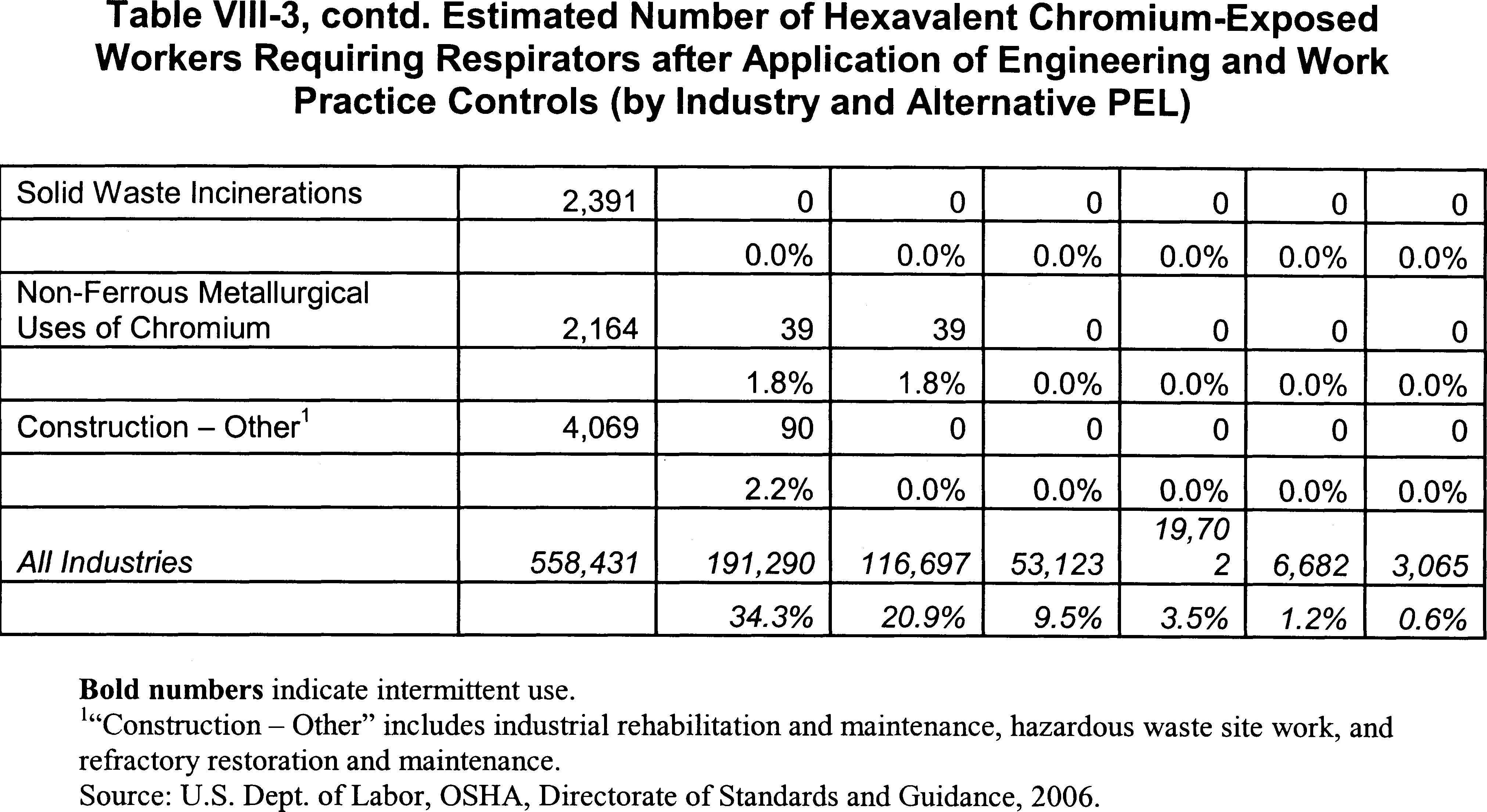
BILLING CODE 4510-26-C
In determining technological feasibility OSHA has used the median
to describe the exposure data. Since the median is a statistical term
indicating the central point of a sequence of numbers (50 percent below
and 50 percent above) it best describes exposures for most people. The
median is also a good substitute for the geometric mean for a log
normal distribution which often describes exposure data. As described
by the Color Pigments Manufacturers Association, Inc. (CPMA) in an
economic impact study by IES Engineers:
The exposure distribution (assuming it is log normal) can be
characterized by the geometric mean and standard deviation. The
median (not the average) is a reasonable estimate of the geometric
mean (Ex. 47-3, p. 54).
In contrast, the use of an arithmetic mean (or average) may tend to
misrepresent the exposure of most people. For example, if there are a
few workers with very high exposures due to poor engineering or work
practice controls, the arithmetic mean will be artificially high, not
representing realistic exposures for the workers.
The technological feasibility chapter of the FEA is broken down
into five main parts: Introduction, Exposure Profile, Baseline
Controls, Additional Controls and Substitution. The first part is an
introduction to the application group, which outlines the major changes
in the analysis between the Preliminary Economic Analysis and the Final
Economic Analysis and addresses comments specific to the application
group.
The next part of the technological feasibility analysis is the
exposure profile. The exposure profile describes the prevailing
exposures in each application group on a job-by-job basis. The exposure
profile represents exposure situations that may be well controlled or
poorly controlled. The data used to determine the current exposures
were obtained from any of the following sources: OSHA site visits; the
OSHA compliance database, Integrated Management Information System
(IMIS); NIOSH site visits; NIOSH control technology or health hazard
evaluation reports (HHE); information from the U.S. Navy; published
literature; submissions by individual companies or associations; or, in
a few cases, by consideration of analogous operations. While the
exposure profile was developed from current exposures and is not
intended to demonstrate feasibility, there were a few instances where
the exposure profile was used as ancillary support for technological
feasibility if there were a significant number of facilities already
meeting the PEL. An example of this case can be seen in the production
of colored glass, where over 90 percent of the exposure data were below
0.25 [mu]g/m3.
In the cases where analogous operations were used to determine
exposures, OSHA used data from industries or operations where materials
and exposure routes are similar. OSHA also tended to be conservative
(over-estimating exposures). For example, exposure data for the bagging
of pigments were used to estimate exposures for the bagging of plastic
colorants. In both cases the operation consists of bagging a pigmented
powder. However, exposures would tend to be higher for bagging pigments
due to the fact that in pigments there is a higher percentage of Cr(VI)
and the pigments tend to consist of finer particles than those in
plastic colorants where the Cr(VI) particles are diluted with other
ingredients. As Mr. Jeff Cox from Dominion Colour Corporation stated:
Exposure of packers in the pigment industry, who are making a
fine powder, is very much higher than packers in the plastics
colorants industry, who are basically packing pellets of
encapsulated product which are a few millimeters in diameter (Tr.
1710).
The use of operations that are more difficult to control to
estimate analogous operations would result in an overestimate of
exposures, subsequently resulting in an overestimate of the controls
needed to reduce the exposures to Cr(VI) in those analogous operations.
The next section of OSHA's analysis of technological feasibility in
the FEA describes the baseline controls. OSHA determined controls to be
"baseline" if OSHA believed that such controls are commonly used in
the application group. This should not be interpreted to mean that OSHA
believes that all firms use these controls, but rather that the
controls are common and widely available in the industry. Information on
the controls used in each specific application group was obtained from
several different sources such as: site visits, NIOSH HHEs, industry
experts, industry associations, published literature, submissions to the
docket, and published reports from other federal agencies. OSHA used the
median to estimate the exposure level associated with the baseline controls.
For the majority of the operations, the median was calculated using the
exposures directly associated with the baseline controls. However,
there were a few cases where the median was calculated from the
exposure profile and OSHA determined these exposures reflected the
baseline controls (e.g., fiberglass production).
The fourth section of the technological feasibility analysis
determined the need for additional controls. If the median exposure was
above the PEL with the use of baseline controls, OSHA would recommend
additional engineering or work practice controls that would reduce
exposures to or below the PEL. The final rule does not require an
employer to use these specific controls. The engineering controls or
work practices are, however, OSHA's suggestions for possible ways to
achieve the PEL. Through this process a few situations could arise when
the exposures with baseline exposures are above the PEL:
Engineering and work practice controls alone: OSHA
determined that additional controls would reduce worker's exposure
below the PEL if: 1) the proposed additional controls were already in
use at other facilities in the same application group and exposures
there were below the PEL, or 2) the additional controls were used in
analogous industries or operations and they were effective.
Respiratory protection required to meet the PEL: There
were a few instances where workers' exposures would remain above the
PEL even with the installation of additional controls. In these cases
OSHA indicated that the supplemental use of respirators may be needed
(e.g. enclosed spray-painting operations in aerospace).
Intermittent respiratory protection: There were instances
where a worker performs specific job-related activities that could
result in higher exposures above the PEL for limited periods of time.
In these cases OSHA noted that the supplemental use of respirators
during these activities may be necessary. For example, an employee who
works in pigment production generally, may need to use a respirator
only when entering the enclosure where the bagging operations take
place because the enclosure is the engineering control in this
operation.
The final component of the technological feasibility section in the
FEA is a discussion of substitution. Here, OSHA describes the options
available for eliminating or reducing the use of ingredients that
either contain or can produce Cr(VI) during processing. This is
primarily a discussion of the possibility of substitution. In some
cases there is no readily available substitute for either chromium
metal or Cr(VI) ingredients such as a non-Cr(VI) coating for corrosion
control in the aerospace industry. In other cases an application group
has been steadily reducing their use of Cr(VI), such as in the printing
industry. In some industries there are substitutes available for at
least some operations, such as the use of trivalent chromium in some
decorative electroplating operations. Finally, through hearing
testimony and docket submissions, OSHA received information regarding
new technologies that can be used to reduce some of the sources of
exposure to the workers.
In most cases OSHA does not rely on material substitution for
reducing exposures to Cr(VI) to determine technological feasibility.
For example, in the case of some welding operations, OSHA has
determined that the use of an alternate welding process that reduces
fume generation, such as the switching from shielded metal arc welding
(SMAW) to gas metal arc welding (GMAW), could be effective in reducing
a worker's exposure to hexavalent chromium to a level at or below the
PEL. Alternatively, experiments have also shown that elimination or
reduction of sodium and potassium in the flux reduces the production of
Cr(VI) in the welding fume (Ex. 50). However, this technology has yet
to be commercialized due to potential weld quality problems. Thus, OSHA
ultimately determined that material substitution was currently not
feasible for SMAW welding operations.
There were comments submitted to the record that did not agree with
certain aspects of OSHA's feasibility analysis. These comments
addressed:
OSHA's use of median values to describe exposure data and
failure to address costs for exposures above the PEL where the median
was below the PEL;
OSHA's use of the number of workers to determine the
number of facilities needing additional controls;
The use/validity of OSHA's analytical method; and
The lack of data/site visits to properly characterize an
application group.
Several commenters objected to OSHA's use of the median in the
technological feasibility analysis. The National Coil Coating
association stated:
It is inappropriate to use median exposure values to reach a
conclusion that no coil coating facility will be subject to
regulatory requirements associated with exceedances of the proposed
PEL. Of the 15 samples supplied, one sample exceeded the proposed
PEL and another one was equal to the proposed PEL (Ex. 39-72-1).
Collier Shannon Scott, representing the Specialty Steel Industry of
North America, stated:
OSHA conducted a technological feasibility analysis to determine
what engineering or administrative controls would be necessary to
achieve the proposed PEL only where the median exposure value for
any particular job category exceeded the proposed PEL. If correct,
this means that where the median exposure value fell below 1 ug/m3,
even though numerous of the exposure values for that job category
were above 1 ug/m3, OSHA's analysis does not recognize that controls
would have to be implemented for that job category at any facilities
where that job is conducted (Ex. 47-27-1).
OSHA believes that these commenters misunderstood OSHA's use of the
median value and the term "additional controls." As stated earlier,
OSHA used the median value to describe either the overall exposures or
the effectiveness of various controls. However, to estimate the cost of
controls, OSHA used the entire exposure profile. Thus, if any exposures
were over the PEL, then costs for engineering controls would be
assigned. If for a job category the "baseline controls" have been
determined to reduce employee exposures to below the PEL, then OSHA
would include costs for "baseline controls" for the percentage of the
facilities that had exposures over the PEL. However, if the
"baseline" controls would not be sufficient to reduce worker
exposures to below the PEL then OSHA would cost the "additional
controls."
Collier Shannon Scott, representing the Specialty Steel Industry of
North America also stated:
OSHA wrongly uses percentage distribution by job category to
estimate the number of facilities that would be required to install
engineering controls. This is a logical error. There is no
connection between the number of facilities that must install
controls and the percentage of employees above a given exposure
level (Ex. 47-27-1).
OSHA was also concerned about accurately using individual exposures
to represent the number of facilities that would need to implement either
baseline controls or additional controls. Thus, whenever exposure data
were associated with individual facilities, OSHA normalized the
exposure data by job category to the facility, with each facility
having a weighting factor of 1. However, if exposure data varied
significantly, OSHA accounted for this. For example, if fifty percent
of the exposure data for a job class in a facility was above the PEL
and fifty percent below the PEL, then OSHA counted this as representing
0.5 facilities above the PEL and 0.5 facilities below the PEL.
The use of this weighting system ensured that each facility
received the same weight so that one facility that supplied a large
amount of data would not overwhelm the exposure profile and skew the
distribution in an application group. This is particularly important
when there is a wide range of sizes of facilities and a large facility
could outweigh a smaller facility. OSHA then used this weighting system
to determine the percentage of facilities affected, so that the costs
were based on a per-facility versus a per-employee basis. However, in a
few instances OSHA could not use the weighting factor system because
certain exposure data were presented to OSHA as representing the
industry. For examples, in maritime welding and aerospace painting the
exposure data could not be attributed to individual facilities but were
presented to OSHA as representing a group of facilities.
There were comments about several different aspects of OSHA's
analytical method. The Policy Group, representing the Surface Finishing
Industry Council, was concerned about how OSHA interpreted the term
non-detect (ND):
Appropriate assessment of ND qualitative value would require
that the sample specific quantitation limit be lower than any
targeted analytical value, such as the new proposed AL and PEL.
According to a leading OSHA/NIOSH contract laboratory (DataChem
Laboratories) in the field of IH analyses, laboratories only report
to the lowest calibration standard. Thus, the lowest standard value
in the curve is the quantitation limit or reporting limit. This
limit is the minimum value the labs generally report, regardless of
any theoretical LOD value (Ex. 47-17-8).
OSHA agrees with The Policy Group's assessment and has updated the
exposure profiles to reflect non-detect samples as the Limit of
Quantification (LOQ) where the source of the data did not indicate the
limit of detection. This is discussed in more detail in the
electroplating section of the technological feasibility chapter in the
FEA.
Several comments questioned whether OSHA's analytical method truly
represents a worker's exposure (Ex. 38-216-1). Several other sources
indicate that OSHA's analytical method ID 215 is appropriate and it
accurately represents a worker's exposure. In a Journal of
Environmental Monitoring article the authors conclude:
* * * a field comparison of three recently developed or modified
CrVI sampling and analytical methods showed no statistically
significant differences among the means of the three methods based
on statistical analysis of variance. The overall performances of the
three CrVI methods were comparable in electroplating and spray
painting operations where soluble CrVI was present. Although the
findings reported herein are representative of workplace operations
utilizing soluble forms of CrVI, these analytical methods (using
identical sample preparation procedures) also have been shown to
quantitatively measure insoluble forms of CrVI in other occupational
settings. There were no significant differences observed among CrVI
concentrations measured by NIOSH 7605 and OSHA ID 215 (Ex. 40-10-5).
In addition URS Corporation stated:
The new OSHA method 215 was used to analyze samples collected
during the Site Visits for Company 1 and Company 18. This method is
far superior to the old OSHA method ID 103 and to other relative
older methods. The new method utilizes separations of the hexavalent
chromium from potential interferences prior to the analysis. It is
also designed to detect much lower CrVI concentrations levels and to
remove both positive and negative interferences at these lower
concentrations. Furthermore, this method has been fully validated in
the presence of interferences over a CrVI concentration range that
includes the proposed new AL and PEL values (Ex. 47-17-8).
OSHA's analytical method ID 215 is a fully validated analytical method
that can analyze Cr(VI) well below the PEL within the accuracy of
measurement as specified in the final standard.
Dr. Joel Barnhart, on behalf of the Chrome Coalition, questioned
how the samples were taken during the OSHA-sponsored site visits (Ex.
40-12-1). At all site visits conducted by OSHA's contractors, certified
industrial hygienists (CIHs) were responsible for either taking samples
or reviewing sampling data provided by the facility visited. All
samples were taken following procedures from either NIOSH or OSHA which
detail the type of sampler, filter and flow rates appropriate for the
analytical methods used. Full details about the samples, operations
they represent and engineering controls can be found in each site visit
report.
Several commenters mentioned that OSHA relied solely on one site
visit for an entire application group (Exs. 38-218; 38-205). While the
OSHA/NIOSH site visits were important to OSHA's understanding of the
processes used in the different application groups, the site visits
were not the sole source of information. OSHA, as stated earlier, used
many different sources to properly characterize an application group.
These sources included: OSHA site visits, OSHA's compliance data base
(IMIS), NIOSH site visits, NIOSH engineering control technology reports
or health hazard evaluation reports, published literature, submissions
by individual companies, as well as detailed discussions with industry
experts. In addition, throughout the rulemaking process OSHA has
requested information regarding processes, exposures, engineering
controls, substitutes and other information pertinent to Cr(VI)
application groups. These requests came in many forms such as
stakeholder meetings, site visits, OSHA's 2002 Request for Information,
and the SBREFA review. OSHA continued to update the technological
feasibility analysis based on information submitted to the docket
during the hearings and during the pre- and post-hearing comment
periods.
OSHA also received comments specific to application groups
regarding issues such as the number of employees potentially exposed,
additional exposure data, and the effectiveness of controls. Comments
that were application group-specific are addressed in the FEA in the
individual sections on those application groups.
The major changes made to the technological feasibility analysis
for the Final Economic Analysis are listed below:
Electroplating--The number of affected workers and
establishments was revised, the exposure distribution was revised for
hard chrome electroplating, and chromate conversion workers and
establishments were added.
Welding--The number of maritime welders was increased,
mild steel welding was added, and control technology for reducing
worker exposure was revised.
Painting--Auto body repair workers were added to general
industry and traffic painting was added to construction. Control
technology for reducing worker exposure was revised for aerospace spray
painting.
Chromium Catalyst Production--Control technology for
reducing worker exposure was revised.
Steel Mills--OSHA revised the distribution of steel
workers, carbon steel workers were added, and downstream users (e.g.
rolling mills and forging operations) were added to this application
group.
Glass Production--Fiber, flat, and container glass
production were added.
Producers of Pre-Cast Concrete Products--Ready mixed
concrete workers were added.
Throughout the analysis the exposure profiles were updated
to reflect additional exposure data submitted to the docket.
Technological Feasibility of the New PEL: There are over 558,000
workers exposed to Cr(VI). Table VIII-2 shows the current exposures to
Cr(VI) by application group. There are employers and some entire
application groups that already have nearly all exposures below the
PEL. However, many others will need to install or improve engineering
and work practice controls to achieve the PEL.
OSHA has determined that the primary controls most likely to be
effective in reducing employee exposure to Cr(VI) are local exhaust
ventilation (LEV), process enclosure, process modification, and
improving general dilution ventilation. In some cases, a firm may not
need to upgrade its local exhaust system, but instead must ensure that
the exhaust system is working to design specification throughout the
process. In other cases, employers will need to upgrade or install new
LEV. This includes installing duct work, a type of hood and/or a
collection system. OSHA estimates that process enclosures may be
necessary for difficult-to-control operations such as dusty operations.
These enclosures would isolate the employees from high exposure
processes and reduce the need for respirators. Process modifications
can also be effective in reducing exposures in some industries to a
level at or below the PEL.
Below are discussions of the types of engineering and work practice
controls that may be needed for the application groups where exposures
are more difficult to control.
Electroplating: OSHA has determined that the PEL of 5 [mu]g/m\3\ is
technologically feasible for all job categories through the use of a
combination of engineering controls. For decorative plating and
anodizing the vast majority (over 80 percent) of workers are already
below 5 [mu]g/m\3\. For the workers above the PEL, there are several
control options to reduce exposures, such as properly maintained
ventilation and the use of fume suppressants. Some firms may not need
to upgrade their local exhaust systems, but must ensure that their
current exhaust systems are working according to design specification.
For example, in hard chrome electroplating (where Cr(VI) exposures are
highest) nearly 100 percent of hard chrome electroplating baths have
LEV at the tank; however, none of the systems inspected during site
visits and for NIOSH reports were operating at the designed
capabilities. Many had disconnected supply lines or holes in the hoods
and were working at 40 percent of their design capabilities. In such
cases, OSHA recommends that these facilities perform the proper
maintenance necessary to bring the system back to its initial
parameters. Even with these deficiencies in engineering controls, over
75 percent of workers are below 5 [mu]g/m\3\.
In addition to improving LEV, the use of fume suppressants can
further reduce the volume of Cr(VI) fumes released from the plating
bath. However, OSHA was unable to conclude, based on the evidence in
the record, that the proposed PEL of 1 [mu]g/m\3\ would have been
technologically feasible for all hard chrome electroplating operations.
In particular, OSHA has significant concerns about the technological
feasibility of the proposed PEL for hard chrome electroplating
operations in which fume suppressants cannot be used to control
exposures to Cr(VI) because they would interfere with product
specifications and render the resulting product unusable.
Welding: The welding operations OSHA expects to trigger
requirements under the new Cr(VI) rule are those performed on stainless
steel, as well as those performed on high-chrome-content carbon steel
and those performed on carbon steel in confined and enclosed spaces. At
the time of the proposal, OSHA believed that carbon steel contained
only trace amounts of chromium and therefore that welding on carbon
steel would not be affected by the standard. Comments and evidence
received during the rulemaking, however, led OSHA to conclude that 10
percent of carbon steel contains chromium in more than trace amounts;
OSHA adjusted its analysis accordingly. See Tr. 581-82.
OSHA has determined that the PEL of 5 [mu]g/m\3\ is technologically
feasible for all affected welding job categories on carbon steel. OSHA
has concluded that no carbon steel welders are exposed to Cr(VI) above
5 [mu]g/m\3\, with the exception of a small portion of workers welding
on carbon steel in enclosed and confined spaces. Furthermore, OSHA has
determined that engineering and work practice controls are available to
permit the vast majority (over 95 percent) of welding operations on
carbon steel in enclosed and confined spaces to comply with a PEL of 5
[mu]g/m\3\.
Although stainless steel welding generally results in higher
exposures than carbon steel welding, OSHA has determined that the PEL
of 5 [mu]g/m\3\ is also technologically feasible for all affected
welding job categories on stainless steel. Many welding processes, such
as tungsten-arc welding (TIG) and submerged arc welding (SAW), already
achieve Cr(VI) exposures below the PEL because they inherently generate
lower fume volumes. However, the two most common welding processes,
shielded metal arc welding (SMAW) and gas metal arc welding (GMAW),
generate greater exposures and may require the installation or
improvement of LEV (defined to include portable LEV systems such as
fume extraction guns (FEG)).
OSHA has found process substitution to be the most effective method
of reducing Cr(VI) exposures. For example, the generation of Cr(VI) in
GMAW welding fume is approximately 4 percent of the total Cr content,
compared to upwards of 50 percent for SMAW. In the proposal, OSHA
estimated that all SMAW workers outside of confined spaces (over 90
percent of the welders) could switch welding processes. However,
hearing testimony and comments indicated that switching to GMAW is not
feasible to the extent that OSHA had originally estimated.
Some comments indicated that this conversion has already taken
place where possible. For example, Atlantic Marine stated they have
already "greatly reduced the use of SMAW and replaced it with GMAW
over the last several years' (Ex. 39-60). Other comments indicated it
is still an ongoing process. For instance, General Dynamics stated,
"There are ongoing efforts to reduce the use of SMAW and replace it
with GMAW for both efficiency and health reasons" (Ex. 38-214). In
addition, some comments expressed concerns about the quality of the
weld if GMAW is used instead of SMAW. (Ex. 39-70).
In view of these concerns OSHA has revised its estimate of the
percentage of SMAW welders that can switch to GMAW from 90 percent to
60 percent. This estimate is consistent with the estimate made by
Edison Welding Institute in a report for the Department of Defense on
Cr(VI) exposures which "identifies engineering controls that can be
effective in reducing worker exposure for many applications in the
shipbuilding and repair industry" (Ex. 35-410).
For those stainless steel SMAW operations that cannot switch to
GMAW, and even for some GMAW operations, the installation or
improvement of LEV may be needed and can be used to reduce exposures.
OSHA has found that LEV would permit most SMAW and GMAW operations to
comply with a PEL of 5 [mu]g/m\3\. OSHA recognizes that the
supplemental use of respirators may still be necessary in some
situations. A significant portion of the welders who may need
supplemental respiratory protection are working in confined spaces or
other enclosed areas, where the use of engineering controls may be
limited due to space constraints. However, respirator use in those
circumstances will not be extensive and does not undermine OSHA's
finding that the PEL of 5 [mu]g/m\3\ is technologically feasible.
For a more detailed explanation of OSHA's technological feasibility
analysis for all welding operations, see Chapter III of the FEA.
Aerospace: OSHA has determined that most operations in the
aerospace industry can achieve a PEL of 5 [mu]g/m\3\. These operations
include sanding Cr(VI) coated parts, assembly, and two-thirds of the
spray painting operations. Field studies have shown that use of LEV at
the sanding source can reduce exposures by close to 90 percent, with
workers exposures well below the final PEL of 5 [mu]g/m\3\. Exposure
data provided to the docket show that the spray painting operations in
paint booths or paint rooms using optimum engineering controls can
achieve worker exposures below the final PEL of 5 [mu]g/m\3\ (excluding
large parts, whole planes, or the interior of the fuselage)
OSHA recognizes that there are certain instances where the
supplemental use of respirators may be necessary because engineering
and work practice controls are not sufficient to reduce exposures below
the PEL. For example, when spray painting large parts or entire planes
in hangars, engineering controls become less effective because of the
large area needing ventilation and the constantly changing position of
workers in relationship to these controls. As a result, OSHA estimates
that engineering and work practice controls can limit exposures to
approximately 25 [mu]g/m\3\ under the conditions described above and
supplemental use of respirators will be needed to achieve the PEL of 5
[mu]g/m\3\. Accordingly, OSHA has adopted a provision for the painting
of whole aircrafts (interior or exterior) and large aircraft parts that
requires employers to reduce exposures to 25 [mu]g/m\3\ with
engineering and work practice controls and supplement these controls
with the use of respiratory protection to achieve the PEL. For a more
detailed explanation of OSHA's technological feasibility analysis for
aerospace painting, see Chapter III of the FEA.
Other Industries: Other application groups that generate fine dusts
such as chromate pigment production, chromium catalyst production, and
chromium dye production may require new or improved ventilation to
achieve the PEL of 5 [mu]g/m\3\. Housekeeping measures are also
important for controlling Cr(VI) exposures in these industries. General
housekeeping and the use of HEPA vacuums instead of dry sweeping will
minimize background exposures for most job categories. For a more
detailed explanation of OSHA's technological feasibility analysis for
chromate pigment producers, chromium catalyst producers, and chromium
dye producers, see Chapter III of the FEA.
Apart from the aerospace painting operations discussed above, OSHA
recognizes that there are a few limited operations where the
supplemental use of respirators may be necessary to achieve the PEL of
5 [mu]g/m\3\. However, OSHA believes that the final PEL can be achieved
in most operations most of the time with engineering and work practice
controls. As noted previously, Table VIII-3 shows OSHA's estimate of
respirator use by industry for each of the PELs that OSHA considered.
Technological Feasibility of the Proposed PEL: As discussed more
thoroughly in paragraph (c) of the Summary and Explanation of the
Standard and in Chapter III of the FEA, OSHA has determined that the
proposed PEL of 1 [mu]g/m\3\ is not feasible across all industries
because it cannot be achieved using engineering and work practice
controls in a substantial number of industries and operations employing
a large number of workers covered by the standard (in particular, see
"Technological Feasibility of the Proposed 1 [mu]g/m\3\ 8-Hour TWA
PEL" in Chapter III of the FEA). Specifically, OSHA has determined
that a PEL of 1 [mu]g/m\3\ is not feasible for welding, which affects
the largest number of establishments and employees.
A PEL of 1 [mu]g/m\3\ is also technologically infeasible for
aerospace painting, where two-thirds of all spray painting operations
cannot reduce exposures to at or below 1 [mu]g/m\3\ using engineering
and work practice controls. Finally, OSHA was unable to conclude that
the proposed PEL was technologically feasible for existing facilities
in several other industries or operations, such as pigment production,
catalyst production, and some hard chrome electroplating operations,
where a PEL of 1 [mu]g/m\3\ would significantly increase the number of
workers requiring respiratory protection.
D. Costs
The costs employers are expected to incur to comply with the final
standard are $282 million per year. In addition, OSHA estimates that
employers will incur $110 million per year to comply with the personal
protective equipment and hygiene requirements already present in
existing generic standards. The final requirements to provide
protective clothing and equipment and hygiene areas are closely aligned
with the requirements of OSHA's current generic PPE and sanitation
standards (e.g., 1910.132 and 1926.95 for PPE and 1910.142 and 1926.51
for the hygiene requirements). Therefore, OSHA estimates that the
marginal cost of complying with the new PPE and sanitation requirements
of the Cr(VI) standard was lower for firms currently subject to and in
compliance with existing generic standards. OSHA's research on these
current standards, however, uncovered some noncompliance. The baseline
chosen for the Cr(VI) regulatory impact analysis reflects this non-
compliance with current requirements. Although OSHA estimates that
employers would need to spend an additional $110 million per year to
bring themselves into compliance with the personal protective equipment
and hygiene requirements already prescribed in existing generic
standards, this additional expenditure is not attributable to the
Cr(VI) rulemaking. However, the rule does require employers to pay for
PPE. In some cases where employers do not now pay for PPE, employers
will incur costs they did not previously have. However, because these
costs were previously borne by employees, this change does not
represent a net cost to the country. OSHA estimates that employers
would be essentially transferring a benefit to employees of $6 million
per year, the value of the portion of the total expense now paid by
employees.
All costs are measured in 2003 dollars. Any one-time costs are
annualized over a ten-year period, and all costs are annualized at a
discount rate of 7 percent. (A sensitivity analysis using a discount
rate of 3 percent is presented in the discussion of net benefits.) The
derivation of these costs is presented in Chapter IV of the full FEA.
Table VIII-4 provides the annualized costs by provision and by
industry. Engineering control costs represent 41 percent of the costs
of the new provisions of the final standard, and respiratory protection costs
represent 25 percent of the costs of the new provisions of the final
standard. Costs for the new provisions for general industry are $192
million per year, costs for constructions are $67 million per year, and
costs for the shipyard sector are $23 million per year. In developing
the costs for construction, OSHA assumed that all work by construction
firms would be covered by the construction standard. However, in
practice some work by construction firms takes the form of maintenance
operations that would be covered by the general industry standard.
(OSHA sought comment on this issue but received none.)
BILLING CODE 4510-26-P


BILLING CODE 4510-26-C
Table VIII-4 also shows the costs by application group. The various
types of welding represent the most expensive application group,
accounting for 51 percent of the total costs.
Table VIII-5 presents OSHA's final total annualized costs by cost
category for each of the alternative PELs considered by OSHA in the
proposed rule. At a discount rate of 7 percent, total costs range from
$112 million for a PEL of 20 [mu]g/m\3\ to $1.8 billion for a PEL of
0.25 [mu]g/m\3\.
OSHA also presents, in Table VIII-6, the distribution of compliance
costs at the time they are imposed. Because firms will have the choice
of whether to finance expenditures in a single year, or spread them out
over four years, OSHA considers it unlikely that a firm would be
impacted in an amount equal to the entire startup cost in the year that
the initial requirements are imposed. On the other hand, capital
markets are not perfectly liquid and particular firms may face
additional lending constraints, therefore OSHA believes that
identifying startup costs, in addition to the annualized costs, is
relevant when exploring the question of economic feasibility and the
overall impact of this rulemaking.
BILLING CODE 4510-26-P

BILLING CODE 4510-26-C
E. Economic Impacts
To determine whether the final rule's projected costs of compliance
would raise issues of economic feasibility for employers in affected
industries, i.e., would adversely alter the competitive structure of
the industry, OSHA first compared compliance costs to industry revenues
and profits. OSHA then examined specific factors affecting individual
industries where compliance costs represent a significant share of
revenue, or where the record contains other evidence that the standard
could have significant impact on the competitive structure of the
industry.
OSHA compared the baseline financial data with total annualized
incremental costs of compliance by computing compliance costs as a
percentage of revenues and profits. This impact assessment for all
firms is presented in Table VIII-7. This table is considered a
screening analysis and is the first step in OSHA's analysis of whether
the compliance costs potentially associated with the standard would
lead to significant impacts on establishments in the affected
industries. The actual impact of the standard on the viability of
establishments in a given industry, in a static world, depends, to a
significant degree, on the price elasticity of demand for the services
sold by establishments in that industry.
BILLING CODE 4510-26-P









BILLING CODE 4510-26-C
Price elasticity refers to the relationship between the price
charged for a service and the demand for that service; that is, the
more elastic the relationship, the less able is an establishment to
pass the costs of compliance through to its customers in the form of a
price increase and the more it will have to absorb the costs of
compliance from its profits. When demand is inelastic, establishments
can recover most of the costs of compliance by raising the prices they
charge for that service; under this scenario, profit rates are largely
unchanged and the industry remains largely unaffected. Any impacts are
primarily on those using the relevant services. On the other hand, when
demand is elastic, establishments cannot recover all the costs simply
by passing the cost increase through in the form of a price increase;
instead, they must absorb some of the increase from their profits.
Commonly, this will mean both reductions in the quantity of goods and
services produced and in total profits, though the profit rate may
remain unchanged. In general, "when an industry is subject to a higher
cost, it does not simply swallow it, it raises its price and reduces
its output, and in this way shifts a part of the cost to its consumers
and a part to its suppliers," in the words of the court in American
Dental Association v. Secretary of Labor (984 F.2d 823, 829 (7th Cir.
1993)).
The Court's summary is in accordance with micro-economic theory. In
the long run, firms can only remain in business if their profits are
adequate to provide a return on investment that assures that investment
in the industry will continue. Over time, because of rising real
incomes and productivity, firms in most industries are able to assure
an adequate profit. As technology and costs change, however, the long
run demand for some products naturally increases and the long run
demand for other products naturally decreases. In the face of rising
external costs, firms that otherwise have a profitable line of business
may have to increase prices to stay viable. Commonly, increases in
prices result in reduced demand, but rarely eliminate all demand for
the product. Whether this decrease in the total production of the
product results in smaller production for each establishment within the
industry, or the closure of some plants within the industry, or a
combination of the two, is dependent on the cost and profit structure
of individual firms within the industry.
If demand is completely inelastic (i.e., price elasticity is 0),
then the impact of compliance costs that are 1 percent of revenues for
each firm in the industry would result in a 1 percent increase in the
price of the product or service, with no decline in quantity demanded.
Such a situation represents an extreme case, but might be correct in
situations in which there are few if any substitutes for the product or
service in question, or if the products or services of the affected
sector account for only a very small portion of the income of its
consumers.
If the demand is perfectly elastic (i.e., the price elasticity is
infinitely large), then no increase in price is possible and before-tax
profits would be reduced by an amount equal to the costs of compliance
(minus any savings resulting from improved employee health and/or
reduced insurance costs) if the industry attempted to keep producing
the same amount of goods and services as previously. Under this
scenario, if the costs of compliance are such a large percentage of
profits that some or all plants in the industry can no longer invest in
the industry with hope of an adequate return on investment, then some
or all of the firms in the industry will close. This scenario is highly
unlikely to occur, however, because it can only arise when there are
other goods and services that are, in the eyes of the consumer, perfect
substitutes for the goods and services the affected establishments
produce.
A common intermediate case would be a price elasticity of one. In
this situation, if the costs of compliance amount to 1 percent of
revenues, then production would decline by 1 percent and prices would
rise by 1 percent. In this case, the industry revenues would stay the
same, with somewhat lower production, but similar profit rates (in most
situations where the marginal costs of production net of regulatory
costs would fall as well). Consumers would, however, get less of the
product or the service for their expenditures, and producers would
collect lower total profits; this, as the court described in ADA v.
Secretary of Labor, is the more typical case.
If there is a price elasticity of one, the question of economic
feasibility is complicated. On the one hand, the industry will
certainly not be "eliminated" with the level of costs found in this
rulemaking, since under these assumptions the change in total profits
is somewhat less than the costs imposed by the regulation. But there is
still the question of whether the industry's competitive structure will
be significantly altered. For example, given a 20 percent increase in
costs, and an elasticity of one, the industry will not be eliminated.
However, if the increase in costs is such that all small firms in an
industry will have to close, this could reasonably be concluded to have
altered its competitive structure. For this reason, when costs are a
significant percentage of revenues, OSHA examines the differential
costs by size of firm, and other classifications that may be important.
Some commenters (Ex. 38-265; Ex. 38-202; Ex. 40-12) questioned the
screening analysis approach for several reasons: (1) It fails to
provide for a facility-by-facility analysis; (2) it fails to consider
that, in some plants, there may be product lines that do not involve
hexavalent chromium; and (3) the concept of cost pass-through is
largely negated by foreign competition. It should be noted that almost
all commenters arguing for the inadequacy of screening analysis also
argued for much higher costs than those estimated by OSHA (criticisms
of costs were examined in Chapter 4). No one in the record presented an
argument as to why costs representing less than one percent of revenues
would be economically infeasible.
First, some commenters (Ex. 38-265; Ex. 40-12; Ex. 47-5) argued
that industry ratios of costs to profits or costs to revenues cannot
adequately determine economic feasibility--instead the analysis must be
conducted on a facility-by-facility basis. OSHA rejects this argument
for two reasons. First, the judicial definition of economic feasibility
notes that a regulation may be economically feasible and yet cause some
marginal facilities to close. (American Textile Mfrs. Institute, Inc.
v. Donovan 452 U.S. 490, 530-532 (1981))
OSHA's obligation is not to determine whether any plants will
close, or whether some marginal plants may close earlier than they
otherwise might have, but whether the regulation will eliminate or
alter the competitive structure of an industry. OSHA has an obligation
to examine industries, and to consider its industry definitions
carefully, so that they compare like with like. However, OSHA does not
have an obligation to conduct facility-by-facility analysis of the
thousands of facilities in the dozens of industries covered by a major
standard. OSHA criteria can be examined through examination of industry
ratios, particularly when the costs represent a very small percentage
of revenues. Again, it must be noted that almost all commenters arguing
for the inadequacy of screening analysis also argued for much higher
costs than those estimated by OSHA, and while not agreeing with the
need for facility-by-facility analysis, OSHA agrees that as costs
become high as a percentage of revenues, something more than industry
ratio analysis may be needed.
Second, some commenters argued that some facilities and industries
have some lines of production involving hexavalent chromium, and some
that do not, and, in such cases, OSHA should analyze only the revenues
and profits associated with the lines using hexavalent chromium. Even
if this were desirable, the data for such an analysis is simply not
publicly available. No
government data source collects data in a way that could be used for
this purpose, and there is little privately collected data that could
be used for this purpose. Even if such data were available, there are
reasons to produce a product line even if it has profits lower than
other product lines, and the data to examine this issue is even more
unavailable. Further, OSHA's mandates, as interpreted by the courts,
focus on the effect of a standard on industries, not on product lines
within those industries. (American Iron & Steel Institute v. OSHA, 939
F.2d 975, 986 (D.C. Cir, 1991))
Finally, some commenters (SFIC, Ex. 38-265; SSINA, Ex. 40-12, Ex.
47-5; Engelhard, Ex. 38-202) questioned the above analysis by bringing
up the issue of foreign competition, and some presented the argument
that foreign competition made price increases impossible.
While foreign competition is an important issue to consider in
analyzing economic feasibility, the presence of foreign competition
does not mean that price increases are impossible. In economic terms,
the case that foreign competition makes price increases impossible
would be an argument that foreign competition puts all firms into the
situation of having infinite elasticity of domestic demand, because
foreign firms are not subject to the regulation, and, as a result can
underprice American firms and drive them out of business.
Is this the case? Both theory and history suggest that it is not.
From a theoretical viewpoint, the ability to sell to a consumer is
determined by the price at the site, plus the cost of transportation,
plus or minus intangible factors (such as quality or timeliness). Under
these circumstances, a specific establishment can be competitive even
if its cost of production is greater than that of foreign competitors--
if the U.S. producer has other advantages.
From a practical viewpoint, econometric studies typically talk
about the elasticity of domestic production with respect to foreign
prices. No one assumes that a lower foreign price simply and totally
assures that the domestic industry will be eliminated. Foreign
competition has been a fact for decades--this does not mean that any
domestic regulation assures that the domestic industry will be
eliminated.
However, foreign competition does mean the elasticity of demand for
domestic production will be greater than the total elasticity of demand
for the product in question. Thus foreign competition is a factor that
can result in greater elasticity of demand for domestic firms, and that
needs to be considered in the context of the overall feasibility
analysis, just as other factors such as the presence or absence of good
substitutes need to be considered in the analysis.
A different problem with the formulation in terms of demand
elasticity given above is that it ignores other things besides the
regulatory costs that may act to shift either the costs of the
production or demand for a product or service. In the normal course of
events, neither demand nor supply is static. Costs of inputs needed
commonly increase (at least in nominal terms). Productivity may
increase or decrease as technology changes. Increases in income or GDP
normally serve to increase demand for a good or service from year to
year (for the majority of goods with positive income elasticity). In a
typical year for most manufacturing industries, some costs will rise,
productivity will also improve, and increases in GDP will increase
demand. Adjusting to cost increases is thus a part of the normal
economic scene. Even a real cost increase brought about by a regulation
may be partially offset by productivity improvement. Finally, even real
price increases may not decrease the quantities sold (and thus force
employers to close) if the price increases are offset by income-driven
increased demand for the good or service. A real price increase caused
by the costs of a regulation will mean that the quantity sold will be
lower than it otherwise would have been, but does not imply that actual
quantity sold for the product will decline as compared to past years.
Table VIII-7 provides costs as percentage of revenues and profits
for all affected establishments. OSHA believes that this is the best
starting point for fulfilling its statutory responsibility to determine
whether the standard affects the viability of an industry as a whole.
Table VIII-8 shows costs as a percentage of profits and revenues
for firms classified as small by the Small Business Administration and
Table VIII-9 shows costs as a percentage of revenues and profits for
establishments with fewer than 20 employees. (These tables use costs
with a discount rate of 7 percent.) These small-business tables show
greater potential impacts, especially for small electroplating
establishments. Based on these results, OSHA has prepared a Final
Regulatory Flexibility Analysis (see Chapter VII of the FEA) to examine
the impacts on small businesses and how they can be alleviated. (Tables
V-5, V-6, and V-7 in the FEA show the same information using a discount
rate of 3 percent.)
BILLING CODE 4510-26-P
















BILLING CODE 4510-26-C
Economic Feasibility for Many Industries With Low Potential Impacts
To determine whether a rule is economically feasible, OSHA
evaluates evidence from a number of sources. And while there is no hard
and fast rule, in the absence of evidence to the contrary OSHA
generally considers a standard economically feasible when the costs of
compliance are less than one percent of revenues. Common-sense considerations
indicate that potential impacts of such a small magnitude are unlikely to
eliminate an industry or significantly alter its competitive structure
particularly since most industries have at least some ability to raise
prices to reflect increased costs. Of course, OSHA recognizes that even
when costs are within this range, there could be unusual circumstances
requiring further analysis. In addition, as a second check, OSHA also
looks to see whether even such low costs may represent more than ten
percent of the profit in a particular industry. If either of these
factors is present, or if there is other evidence of industry demise or
potential disruption in an industry's competitive structure because of
the standard, OSHA examines the effect of the rule on that industry
more closely. Finally, OSHA reviews the record for any other unusual
circumstances, such as excellent substitutes of equal cost that might
make an industry particularly sensitive to price change. In this case,
the only argument of this kind that OSHA noted was an argument by one
commenter that trivalent chromium plating might be substituted in some
applications for hexavalent chromium. However, even if this is the case
(some in the record did not agree), a plating operation could switch to
trivalent plating with minimal capital investment and thus remain in
business.
OSHA believes that a potential one percent revenue effect is an
appropriate way to begin the analysis in light of the fact that the
United States has a dynamic and constantly changing economy. There is
an enormous variety of year-to-year events that could cause a one
percent increase in a business's costs, e.g., increasing fuel costs, an
unusual one-time expense, changes in costs of materials, increased
rents, increased taxes, etc. Table V-8, which shows year to year
changes in prices for a number of industries affected by the standard,
reflects this phenomenon.
Changes in profits are also subject to the dynamics of the economy.
A recession, or a downturn in a particular industry, will typically
cause profit declines in excess of ten percent for several years in
succession. Table V-9, which shows annual profits for several years in
succession, illustrates this phenomenon. While a permanent loss of
profits presents a greater problem than a temporary loss, these year-
to-year variations do serve to show that small changes in profits are
quite normal without affecting the viability of industries.
The potential impacts of this regulation on the affected employers,
for the most part, are within the range of normal year-to-year
variation that firms and industries expect and survive. Table V-8 in
the FEA shows year-to-year price variations for selected industries
with hexavalent chromium exposure, and Table V-9 (in the FEA) shows
year-to-year profit variations for selected industries with hexavalent
chromium exposures. Table V-8 serves the purpose of showing that, for
many industries, annual price changes of one percent or more are
commonplace without affecting the viability of the industry. Table V-9
serves to show that temporary profit swings of significantly more than
ten percent are also well within the boundaries of normal year-to-year
change.
Because a permanent decrease in profits is much more significant
than a temporary swing of the same magnitude, OSHA has also used the
fact that a very large short term decline can be compared in effect to
a smaller long-term decrease in profits to calculate the extent to
which the temporary changes shown in Table V-9 may demonstrate an
industry's ability to withstand a long-term change. For example, using
a 7 percent discount rate, and the assumption that profits return to
the long term average following a temporary decline, the following
short term declines are approximately equivalent to a 10 percent long-
term decline:
50 percent decline for one year;
30 percent decline for two years;
20 percent decline for three years.
Looking at profits of the average corporation for the period of
1990 to 2002, events of one of the above magnitudes have occurred twice
in that 12-year period without threatening industrial viability. (Based
on corporate profit rate data from IRS, Statistics of Income: Corporate
Income Tax Returns, as Reported in U.S. Department of Commerce, U.S.
Statistical Abstract 2006). And since, as discussed below, demand is
not perfectly elastic in any of the affected industries, it is unlikely
that the actual effect on profits will be as high as indicated in Table
VIII-7.
The record does not contain evidence that any of the affected
industries for which OSHA found that the costs of complying with the
standard will be less than both one percent of prior revenue and ten
percent of prior profits will in fact be threatened by the standard.
Although some industry representatives asserted that compliance would
threaten their existence, these assertions (with one exception,
discussed below) were not supported by empirical evidence that even the
proposed PEL of 1 would be economically infeasible. As noted above,
cost changes of less than one percent are routinely passed on and
impacts that are less than 10 percent of profits have not been shown to
be likely to affect the viability or competitive structure of any of
the industries affected by this standard.
Economic Feasibility for Industries With Higher Potential Impacts
In Table VIII-7, OSHA found that there were 9 industries in three
application groups in which costs were greater than 1 percent of
revenues, and an additional 22 industries in six application groups in
which costs were greater than 10 percent of profits.
However, this number of industries is somewhat misleading. Seven of
the industries in which costs exceed one percent of revenues, and an
additional twelve of those in which costs exceeded 10 percent of
profits (without exceeding 1 percent of revenues) are industries in the
plating and welding application groups in which plating or welding are
exceedingly rare, such as electroplating in the performing arts,
spectator sports and related industries (NAICS 711) and welding in
religious, governmental, civil, and professional organizations (NAICS
813). In both cases, only one establishment in the entire industry
reported engaging in either welding or plating. It is difficult to
determine whether reports of welding or plating in such industries
represent an extremely unusual situation or, perhaps, simply someone
inadvertently checking the wrong box on a survey. In either case, OSHA
concludes that if such establishments do indeed engage in welding or
plating, they could maintain their primary line of business, as almost
everyone else in their industries does, by dropping welding or plating
operations if such operations represented any threat whatsoever to the
viability of their businesses.
The same is true of the other industries that are in the general
category of extremely rare and unusual users of plating operations:
Specialty trade contractors (NAICS 238); wholesale trade and durable
goods (NAICS 423); motor vehicle and parts dealers (NAICS 441);
furniture and home furnishing stores (NAICS 442); electronics and
appliance stores (NAICS 443); building materials and garden equipment
dealers (NAICS 444); health and personal care stores (NAICS 446);
miscellaneous store retailers (NAICS 453); nonstore retailers (NAICS
454); information services and data processing service (NAICS 519);
rental and leasing services (NAICS 532); professional, scientific and
technical services (NAICS 541); performing arts, spectator sports and
related industries (NAICS 711); and personal and laundry services
(NAICS 812). In the welding application groups, the industries in this
category are: gasoline stations (NAICS 447); nursing and residential
care (NAICS 623); social assistance (NAICS 624); food services and
drinking places (NAICS 722); and religious, governmental, civil, and
professional organizations (NAICS 813).
The remainder of this section examines those industries with higher
potential impacts where their businesses may be dependent on Cr (VI)
applications.
Electroplating Job Shops: Electroplating job shops (NAICS 332813:
electroplating, plating, polishing anodizing and coloring services) are
a service industry for the manufacturing sector, and, to a lesser
extent, to those maintaining, restoring, or customizing objects with
metal parts. At a PEL of 5, job shops have costs as a percentage of
profits of 30 percent and costs as a percentage of revenues of 1.24
percent. These firms sell a service rather than a product. (Firms that
directly sell the products they plate end up in other NAICS codes.) As
a result, plating firms are primarily affected by foreign competition
through the loss of other manufacturing in the United States, rather
than through their customers sending products or their component parts
abroad for electroplating. However, some commenters noted that there
may be cases of sending products abroad for the sole purpose of
electroplating. This seems unlikely to be commonplace however, because
of the shipping times and costs for a process that normally represents
a very small part of the value added for the ultimate product. In
addition, because electroplating is essential to the manufacture of
most plated products, the ultimate demand for plating services is
unlikely to decrease significantly.
Finally, independent electroplating shops have been subject to
annual profit changes larger in magnitude than those associated with
this standard. Table V-9 in the FEA shows that, over the past ten
years, profits in this industry have risen and fallen as much as 49
percent in one year without affecting the viability of the industry.
Although these kinds of temporary changes would not have the effect of
permanent decline of profits by 30 percent, OSHA believes that all of
the factors discussed above indicate that there is sufficient price
elasticity and other flexibility in this industry to absorb these
costs.
The price increase of 1.24 percent required to fully restore
profits at a PEL of five is significantly less than the average annual
increase in price of electroplating services, as shown by Table V-8 in
the FEA. Further, during the period shown in Table V-8, the industry
successfully survived, without any real price increase, the regulatory
costs imposed by EPA's Chrome MACT standard. The costs of that standard
are somewhat uncertain. Some commenters argued that that standard could
be quite expensive. One commenter suggested that one facility had
incurred costs of $80,000 per year to meet that standard, and that such
high costs were not atypical. (Tr. 2003) Another commenter noted,
however, that "the effect of the MACT Standard was minimized when
people realized that the combination of a mist suppressant and the
development of a mist suppressant that would work in a hard chrome
installation along with the use of mesh pads puts you below the MACT
standard." (Tr. 2203) The commenter apparently felt that, in the
latter case, the costs would not have been significant. Nevertheless,
in either event, probably due to productivity improvement in other
aspects of the industry, there was no real price increase or massive
dislocation in the industry.
SFIC (Ex. 38-265) also argued that it was difficult to pass on
costs in electroplating based on an EPA study that estimated a cost
pass through elasticity of 0.58. This study was based on pre-1996 data,
and found a statistical relationship between nominal price increases
and increases in a nominal cost index. Whatever the difficulties in
passing increased costs to its customers the industry might have had
before 1996, since that time nominal prices have increased in ways that
did not have the effects on profit predicted by the EPA study.
Even in the event of a real price increase, we believe that demand
for electroplating services is relatively inelastic. For most products
that are plated, plating is basically essential to the function of the
product. The EPA study for the MACT standard found that products
incorporating electroplating had relatively inelastic demand, on the
order of less than 0.5, and the cost of plating represented a very
small percentage of the total costs of the products in question. In
this situation, the chief danger associated with a real cost increase
of less than 1 percent is that there would be some increased foreign
penetration of U.S. markets. However, the small size of the change, and
the difficulty of sending products abroad solely for plating services,
assures that the price change in question would not eliminate the
industry, and is unlikely to alter the competitive structure of the
industry.
However, OSHA is concerned about the economic feasibility of the
standard for electroplating at a PEL of 1. At this lower PEL, costs of
the standard represent 2.7 percent of revenues and 65 percent of
profits. In almost all OSHA health standards in which this figure was
developed, the costs for the most affected industry have been less than
2 percent of revenues. (The major exception was brass and bronze
foundries, where the lead standard PEL was found economically
infeasible with the use of engineering controls.) Further, in standards
where the costs might have been in excess of 2 percent of revenues,
OSHA has sought ways to lower the cost through long term phase-ins of
engineering controls. OSHA examined this possibility for job-shop
electroplaters, and found that even allowing the use of respirators
rather than engineering controls would not significantly lower the
costs as percentage of revenues. OSHA also examined the issue of
whether there were particular types of platers that might have
unusually high or low costs, and found that even quite different
plating shop configurations with respect to the type of plating done
would have approximately equal average costs.
Given the high level of costs as a percentage of revenues and
profits, and the inability to alleviate those impacts without a higher
PEL, OSHA further examined the economic feasibility of the standard at
a PEL of 1. It seems unlikely that a price increase of 2.7 percent,
although significantly larger than the average nominal price increases
in recent years, would eliminate the industry entirely. OSHA has
concluded, however, that the costs associated with such a PEL could
alter the competitive structure of the industry. OSHA has concluded
this because these costs substantially exceed the average nominal price
increases in the industry, and the reasons for these nominal price
increases--increases in the cost of labor and energy, for example--will
continue. Thus a price increase that would assure continued
profitability for the entire industry would require almost tripling the
annual nominal price increase. (The long term average price increase
for plating, as shown in Table V-9, is 1.6 percent per year. Assuming
this continues to be needed, an increase that would leave profits
unchanged would require a cost increase of 4.2 percent (1.6 plus 2.6),
almost three times as much.) That would represent a significant real
price increase that might not be passed forward, particularly by older
and less profitable segments of the industry.
Welding (Stainless Steel) in Construction: OSHA calculated that the
costs of the standard could equal 22.3 percent of profits in this
industry, but only 0.92 percent of revenues. The maximum price
increases required to fully restore profits (0.92 percent) is unlikely
to significantly alter the demand for construction welding services
which are essential for many projects and not subject to foreign
competition. Further, costs of using stainless steel (the chief source
of welding exposure) already vary significantly from year to year, and
often from month to month. Table V-10 shows the producer price index
for steel prices. Prices of steel have changed by more than 10 percent
within a single year a number of times in the past ten years without
affecting the viability of the use of stainless steel in construction.
Welding in General Industry: There are a significant number of
establishments engaged in welding in repair and maintenance (NAICS 811)
and in personal and laundry services (NAICS 812). For repair and
maintenance services, the costs as a percentage of revenues are 0.40
percent and the costs as a percentage of profits are 10.5 percent. For
personal and laundry services the costs as a percentage of revenues are
0.67 percent and costs as a percentage of profits are 13 percent. (All
costs include the costs of any respirators welders will need to use.)
These two sectors conduct maintenance and repair welding. Even if costs
cannot be passed on, the resulting declines in profits are unlikely to
affect the viability of an otherwise viable employer. Further,
businesses of this kind are more likely to be able to increase costs
because of the absence of foreign competition. While some loss of
revenue is possible with a price increase, it is unlikely that the
quantity of routine repairs would be significantly affected by price
increases of this magnitude.
Painting and Corrosion Protection: Four sectors in the painting
application groups have costs as a percentage of revenues in excess of
one percent or costs as a percentage of profits in excess of 10
percent. These are motor vehicle body and trailer manufacturing (NAICS
3362) with costs of 0.51 percent and 20 percent; military armored
vehicle and tank manufacturers (NAICS 336992) with costs of 0.25
percent and 10 percent; used car dealers (NAICS 44112) with costs of
0.41 percent and 34 percent; and automotive body, paint and interior
repair (NAICS 81121) with costs of 1.5 percent and 39 percent. These
costs are incurred in part for the use of hexavalent chromium pigments,
but largely for using hexavalent chromium coating (applied like paint)
as undercoats for corrosion protection. In the case of the first two
NAICS codes, these are part of manufacturing processes. For both of
these manufacturing industries, while the costs of hexavalent chromium
coatings may be significant in the establishments where they are
applied, the costs of Hexavalent chromium coatings represent an
insignificant percentage of the costs of a car or a tank. While
manufacturers may seek substitutes for hexavalent chromium coatings,
additional expenses for such coatings are unlikely to affect the
ultimate demand for cars or tanks. The latter two affected industries
involve repair and refurbishing of existing automobiles. The cost
analysis assumes all firms who currently use hexavalent chromium in
these industries will continue to do so. In each case, there are
choices that would avoid the costs in question. One choice would be to
use non-hexavalent chromium pigments or non-hexavalent chromium
corrosion protection. A variety of substitutes have been developed, and
the use of hexavalent chromium based coatings for these purposes is
already banned in California. (Tr. 1913) Although these substitutes
have not yet been subject to long term use and their protectiveness is
currently less certain than that of hexavalent chromium, it is likely
that products that are equivalent to hexavalent chromium will be
developed, particularly if demand for such products increases as a
result of the standard. In addition, applying hexavalent chromium
coatings represents a very small portion of the business of either auto
body repair shops or used car dealers. A firm whose viability was
seriously threatened as a result of this standard could retain most of
its core businesses without continuing to use hexavalent chromium.
In addition, it is also reasonable to suppose that both used cars
and auto body repair do not have highly elastic demand, such that a
small change in prices would result in a very large drop in the number
of cars repaired. As a result, the required increases in price can be
accommodated without such significant losses as to alter the
competitive structure of the industries.
Chromium Catalyst Producers (0.8 percent; 27 percent) and Service
Companies (0.44 percent; 12 percent): Chromium catalyst production and
service companies are also unlikely to be affected by costs of the
relative magnitude found here. Most companies are locked into the use
of specific catalysts without major new investments. As a result, while
there may be some small long-term shift away from the use of chromium
catalysts, a price change of one percent is unlikely to immediately
prompt such a change. This also means that the market for chrome
catalyst services is likely to be maintained. Further, faced with a new
regulation, companies are more rather than less likely to turn to a
service company to handle chromium products. Based on these
considerations, OSHA determined that the standard is economically
feasible in these sectors.
Iron and Steel Foundries: Iron and steel foundries (NAICS 3315)
have costs that are 0.42 percent of revenues and 15 percent of profits.
An oddity of the estimated costs for this industry is that 44 percent
of the costs are associated with monitoring costs. In this cost
estimate, OSHA assumes that iron and steel foundries will use scheduled
periodic monitoring rather than adopting the option of performance-
based monitoring. Adopting a performance-based monitoring approach
rather than scheduled monitoring might well reduce costs as a
percentage of profits to less than 10 percent of profits. As noted
above, cost changes of less than one percent are routinely passed on
and impacts that are less than 10 percent of profits have not been
shown to be likely to affect the viability or competitive structure of
any of the industries affected by this standard.
Even if costs are not reduced, the industry has demonstrated its
ability to survive real cost increases by remaining viable in the face
of a 32 percent increase in the price of its basic input, steel, over
the last two years. Based on these considerations, OSHA concludes the
standard is feasible for this sector.
F. Benefits and Net Benefits
OSHA estimated the benefits associated with alternative PELs for
Cr(VI) by applying the dose-response relationship developed in the risk
assessment to current exposure levels. OSHA determined current exposure
levels by first developing an exposure profile for industries with
Cr(VI) exposures using OSHA inspection and site visit data, and then
applying this profile to the total current worker population. The
industry-by-industry exposure profile was given in Table VIII-2 above.
By applying the dose-response relationship to estimates of current
exposure levels across industries, it is possible to project the number
of lung cancers expected to occur in the worker
population given current exposures (the "baseline"), and the number
of these cases that would be avoided under alternative, lower PELs.
OSHA assumed that exposures below the limit of detection (LOD) are
equivalent to no exposure to Cr(VI), thus assigning no baseline or
avoided lung cancers (and hence, no benefits) to these exposures. For
exposures above the current PEL and for purposes of determining the
benefit of reducing the PEL, OSHA assumed exposure at exactly the PEL.
Consequently, the benefits computed below are attributable only to
a change in the PEL. No benefits are assigned to the effect of a new
standard increasing compliance with the current PEL. OSHA estimates
that between 3,167 and 12,514 lung cancers attributable to Cr(VI)
exposure will occur during the working lifetime of the current worker
population. Table VIII-10 shows the number of avoided lung cancers by
PEL. At the final PEL of 5 [mu]g/m3, an estimated 1,782 to
6,546 lung cancers would be prevented over the working lifetime of the
current worker population.
Note that the Agency based these estimates on a worker who is
employed in a Cr(VI)-exposed occupation for his entire working life,
from age 20 to 65. The calculation also does not allow workers to enter
or exit Cr(VI) jobs, nor switch to other exposure groups during their
working lives. While the assumptions of 45 years of exposure and no
mobility among exposure groups may seem restrictive, these assumptions
actually are likely to yield somewhat conservative (lower) estimates of
the number of avoided cancers, given the nature of the risk assessment
model.
BILLING CODE 4510-26-P

BILLING CODE 4510-26-C
For example, consider the case of job covered by five workers, each
working nine years rather than one worker for 45 years. The former
situation will likely yield a slightly higher rate of lung cancers,
since more workers are exposed to the carcinogen (albeit for a shorter
period of time) and the average age of the workers exposed is likely to
decrease. This is due to: (1) The linearity of the estimated dose-
response relationship, and (2) once an individual accumulates a dose, the
increase in relative risk persists for the remainder of his lifetime.
For example, a worker exposed from age 20 to 30 will have a constant
increased relative risk for about 50 or so years (from age 30 on,
assuming no lag between exposure and increased risk and death at age
80), whereas a person exposed from age 40 to 50 will have only about 30
years of increased risk (again assuming no lag and death at age 80).
The persistence of the increased relative risk for a lifetime follows
directly from the risk assessment and is typical of life table
analysis.
For informational purposes only, OSHA has estimated the monetary
value of the benefits associated with the final rule. These estimates
are informational because OSHA cannot use benefit-cost analysis as a
basis for determining the PEL for a health standard. In order to
estimate monetary values for the benefits associated with the final
rule, OSHA reviewed the approaches taken by other regulatory agencies
for similar regulatory actions. OSHA found that occupational illnesses
are analogous to the types of illnesses targeted by EPA regulations and
has thus used them in this analysis.
OSHA is adopting EPA's approach, applying a value of $6.8 million
to each premature fatality avoided. The $6.8 million value represents
individuals' willingness-to-pay (WTP) to reduce the risk of premature
death.
Nonfatal cases of lung cancer can be valued using a cost of illness
(COI) approach, using data on associated medical costs. The EPA Cost of
Illness Handbook (Ex.35-333) reports that the medical costs for a
nonfatal case of lung cancer are, on average, $136,460. Updating the
EPA figure to 2003 dollars yields the value of $160,030. Including
values for lost productivity, the total COI which is applied to the
OSHA estimate of nonfatal cases of lung cancer is $188,502.
An important limitation of the COI approach is that it does not
measure individuals' WTP to avoid the risk of contracting nonfatal
cancers or illnesses. As an alternative approach, nonfatal cancer
benefits may be estimated by adjusting the value of lives saved
estimates. In its Stage 2 Disinfection and Disinfection Byproducts
water rule, EPA used studies on the WTP to avoid nonfatal lymphoma and
chronic bronchitis as a basis for valuing nonfatal cancers. In sum, EPA
valued nonfatal cancers at 58.3 percent of the value of a fatal cancer.
Using WTP information would yield a higher estimate of the benefits
associated with the reduction in nonfatal lung cancers, as the nonfatal
cancers would be valued at $4 million rather than $188,502 per case.
These values represent the upper and lower bound values for nonfatal
cases of lung cancer avoided.
Using these assumptions, latency periods of 15, 20, 25, and 30
years--and adjustments to the value of statistical life to today--OSHA
estimated the total annual benefits of the standard at various PELS in
Table VIII-11, considering the benefits from preventing both fatal and
non-fatal cases of lung cancer.
Occupational exposure to Cr(VI) has also been linked to a multitude
of other health effects, including irritated and perforated nasal
septum, skin ulceration, asthma, and dermatitis. Current data on Cr(VI)
exposure and health effects are insufficient to quantify the precise
extent to which many of these ailments occur. However, it is possible
to provide an upper bound estimate of the number of cases of dermatitis
that occur annually and an upper estimate of the number that will be
prevented by a standard. This estimate is an upper bound because it
uses data on incidence of dermatitis among cement workers, where
dermatitis is more common than it would be for other exposures to
Cr(VI). It is important to note that if OSHA were able to quantify all
Cr(VI)-related health effects, the quantified benefits would be
somewhat higher than the benefits presented in this analysis.
BILLING CODE 4510-26-P

BILLING CODE 4510-26-C
Using National Institute for Occupational Safety and Health (NIOSH)
data, Ruttenberg and Associates (Ex. 35-332) estimate that the
incidence of dermatitis among concrete workers is between 0.2 and 1
percent. Applying the 0.2 percent-1 percent incidence rate indicates
that there are presently 418-2,089 cases of dermatitis occurring
annually. This approach represents an overestimate for cases of
dermatitis in other application groups, since some dermatitis among
cement workers is caused by other known factors, such as the high
alkalinity of cement. If the measures in this final standard are 50
percent effective in preventing dermatitis, then there would be an
estimated 209-1,045 cases of Cr(VI) dermatitis avoided annually.
To assign values to the cases of avoided dermatitis OSHA applied
the COI approach. Ruttenberg and Associates computed that, on average,
the medical costs associated with a case of dermatitis are $119 (in
2003 dollars) and the indirect and lost productivity costs are $1,239
(Ex. 35-332). These estimates were based on an analysis of BLS data on
lost time associated with cases of dermatitis, updated to current
dollars. Based on the Ruttenberg values, OSHA estimates that a Cr(VI)
standard will yield $0.3 million to $1.4 million in annual benefits due
to reduced incidence of dermatitis.
Occupational exposure to Cr(VI) can lead to nasal septum
ulcerations and nasal septum perforations. As with cases of dermatitis,
the data were insufficient to conduct a formal quantitative risk
assessment to relate exposures and incidence. However, previous studies
provide a basis for developing an approximate estimate of the number of
nasal perforations expected under the current PEL as well as PELs of
0.25 [mu]g/m3, 0.5 [mu]g/m3, 1.0 [mu]g/
m3, 5.0 [mu]g/m3, 10.0 [mu]g/m3 and
20.0 [mu]g/m3. Cases of nasal perforations were computed
only for workers in electroplating and chrome production. The
percentage of workers with nasal tissue damage is expected to be over
50 percent for those regularly exposed above approximately 20 [mu]g/
m3. Less than 25 percent of workers could reasonably be
expected to experience nasal tissue damage if Cr(VI) exposure was kept
below an 8-hour TWA of 5 [mu]g/m3 and regular short-term
exposures (e.g. an hour or so) were below 10 [mu]g/m3. Less
than 10 percent of workers could reasonably be expected to experience
nasal tissue damage at a TWA Cr(VI) below 2 [mu]g/m3 [and
short-term exposures below 10 [mu]g/m3]. It appears likely
that nasal damage might be avoided completely if all Cr(VI) exposures
were kept below 1 [mu]g/m3.
OSHA estimates that 1,728 nasal perforations/ulcerations occur
annually under current exposure levels. OSHA estimates that 1,140 of
these would be prevented under the final PEL of 5 [mu]g/m3.
Due to insufficient data, it was not possible to monetize the benefits.
Thus, the benefits associated with a reduction in nasal perforations/
ulcerations are excluded from the net benefits analysis presented
below.
Finally, for informational purposes, OSHA examined the net benefits
of the standard, based on the benefits and costs presented above, and
the costs per case of cancer avoided, as shown in Table VIII-12.
As noted above, the OSH Act requires OSHA to set standards based on
eliminating significant risk to the extent feasible. That criterion or
a criterion of maximizing net (monetary) benefits may result in very
different regulatory outcomes. Thus, these analyses of net benefits
cannot be used as the basis for a decision concerning the choice of a
PEL for a Cr(VI) standard.
BILLING CODE 4510-26-P

BILLING CODE 4510-26-C
Nevertheless, the Agency agrees that additional information
concerning the circumstances in which monetary benefits exceed costs
would be a useful addition to the above table. OSHA found the following
conditions key to determining whether benefits exceed costs:
If the risk is at the lowest end of the range considered,
then benefits do not exceed costs no matter what other variables are
used.
If the risk is at the high end of the range, and a
discount rate of 7 percent is used, then benefits exceed costs for PELs
of 1 and 20 if the latency period is less than 20 years, and for PELs
of 5 and 10 if the latency period is less than 25 years.
If the risk is at the high end of the range, and a
discount rate of 3 percent is used, then benefits exceed costs for a
PEL of 0.5 if the latency period is twenty years or less, and benefits
exceed costs for all latency periods for all higher PELs.
Incremental costs and benefits are those that are associated with
increasing stringency of the standard. Comparison of incremental
benefits and costs provides an indication of the relative efficiency of
the various PELs. OSHA cannot use this information in selecting a PEL,
but it has conducted these calculations for informational purposes.
Incremental costs, benefits, net benefits and cost per cancer avoided
are presented in Table VIII-13.
In addition to examining alternative PELs, OSHA also examined
alternatives to other provisions of the standard. These alternatives
are discussed in the summary of the Final Regulatory Flexibility
Analysis in the next section.

G. Summary of the Final Regulatory Flexibility Analysis
The full final regulatory flexibility analysis is presented in
Chapter VII of the FEA. Many of the topics discussed there, such as the
legal authority for the rule; the reasons OSHA is going forward with
the rule; and economic impacts on small business have been presented in
detail elsewhere in the Preamble. As a result, this section focuses on
two issues: duplicative, overlapping, or conflicting rules; and
alternatives OSHA considered.
Federal Rules That May Duplicate, Overlap, or Conflict With the Final
Rules
OSHA's SBREFA panel for this rule suggested that OSHA address a
number of possible overlapping or conflicting rules: EPA's Maximum
Achievable Control Technology (MACT) standard for chromium
electroplaters; EPA's standards under the Federal Insecticide,
Fungicide, and Rodenticide Act (FIFRA) for Chromium Copper Arsenate
(CCA) applicators; and state use of OSHA PELs for setting fence line
air quality standards. The Panel was also concerned that, in some
cases, other OSHA standards might overlap and be sufficient to assure
that a new final standard would not be needed, or that some of the
final standard's provisions might not be needed.
OSHA has thoroughly studied the provisions of EPA's MACT standard
and has also consulted with EPA. The standards are neither duplicative
nor conflicting. The rules are not duplicative because they have
different goals--environmental protection and protection against
occupation exposure. It is quite possible, as many electroplaters are
now doing, to achieve environmental protection goals without achieving
occupational protection goals. The regulations are not conflicting
because there exist controls that can achieve both goals without
interfering with one another. However, it is possible that meeting the
final OSHA standard would cause someone to incur additional costs for
the MACT standard. If an employer has to make major changes to install
LEV, this could result in significant expenses to meet EPA requirements
not accounted for in OSHA's cost analysis. In its final cost estimates,
OSHA has included costs for additional MACT testing in cases where it
may be needed. OSHA has also allowed all facilities four years to
install engineering controls, with the result that electroplaters can
better coordinate their EPA and OSHA requirements and avoid the need
for extra testing.
OSHA examined the potential problem of overlapping jurisdiction for
CCA applicators, and found that there would indeed be overlapping
jurisdiction. As a result, OSHA had excluded CCA applicators from the
scope of the coverage of the rule. OSHA has been unable to find a case
where a state, as a matter of law, bases fence line standards on OSHA
PELs. OSHA notes that the OSHA PEL is designed to address the risks
associated with life long occupational exposure only.
OSHA has also examined other OSHA standards, and where standards
are overlapping, referred to them by reference in the final standard in
order to eliminate the possibility of overlapping, duplicative or conflicting
standards. Existing OSHA standards that may duplicate the final provisions
in some respect include the standards addressing respiratory protection (29 CFR
1910.134); hazard communication (29 CFR 1910.1200); access to medical
and exposure records (29 CFR 1910.1020); general requirements for
personal protective equipment in general industry (29 CFR 1910.132),
construction (29 CFR 1926.95), and shipyards (29 CFR 1915.152); and
sanitation in general industry (29 CFR 1910.141), construction (29 CFR
1926.51), and shipyards (29 CFR 1915.97).
Regulatory Alternatives
This section discusses various alternatives to the final standard
that OSHA considered, with an emphasis on those suggested by the SBREFA
Panel as potentially alleviating impacts on small firms. (A discussion
on the costs of some of these alternatives to OSHA's final regulatory
requirements for the hexavalent chromium standard can be found in
Section III.3 Costs of Regulatory Alternatives in the final report by
OSHA's contractor, Shaw (Shaw, 2006). In the Shaw report, costs are
analyzed by regulatory alternative and major industry sector at
discount rates of 7 percent and 3 percent.)
Scope: The proposed standard covered exposure to all types of
Cr(VI) compounds in general industry, construction, and shipyard.
Cement work in construction was excluded.
OSHA considered the Panel recommendation that sectors where there
is little or no known exposure to Cr(VI) be excluded from the scope of
the standard. OSHA decided against this option. The costs for such
sectors are relatively small--probably even smaller than OSHA has
estimated because OSHA did not assume that any industry would use
objective data to demonstrate that initial assessment was not needed.
However, it is possible that changes in technology and production
processes could change the exposure of employees in what are currently
low exposure industries. If this happens, OSHA would need to issue a
new standard to address the situation. As a result, OSHA is reluctant
to exempt industries from the scope of the standard.
However, OSHA has rewritten the scope of the standard for the final
rule so that it exempts from the scope of the standard any employer who
can demonstrate that a material containing Cr(VI) or a specific
process, operation, or activity involving Cr(VI) will not result in
concentrations at or above 0.5 [mu]g/m\3\ under any condition of use.
As a result, industries are exempted from all provisions of the
standard and all costs if the industry can demonstrate that exposure is
always at relatively low levels. This approach seems the best way to
minimize the costs for the standard for industries where exposure is
currently minimal, but could change in the future.
As stated above, the final standard does not cover exposures to
hexavalent chromium resulting solely from exposure to portland cement.
OSHA's assessment of the data indicates that the primary exposure to
cement workers is dermal contact that can lead to irritant or contact
allergic dermatitis. Current information indicates that the exposures
in cement work are well below 0.25 [mu]g/m\3\. Moreover, unlike other
exposures in construction, general industry or shipyards, exposures
from cement are most likely to be solely from dermal contact. There is
little potential for airborne exposures and unlikely to be any in the
future, as Cr(VI) appears in cement in only minute quantities
naturally. Given these factors, the final standard excludes cement from
the scope of the standard. OSHA has determined that addressing the
dermal hazards from these exposures to Cr(VI) through guidance
materials and enforcement of existing personal protective equipment and
hygiene standards may be a more effective approach. Such guidance
materials would include recommendations for specific work practices and
personal protective equipment for cement work in construction.
OSHA's analysis suggests that there are 2,093 to 10,463 cases of
dermatitis among cement workers annually. Using a cost of illness (COI)
approach, avoiding 95 percent of these dermatoses would be valued at
$2.5 million to $12.6 million annually, and avoiding 50 percent of
these dermatoses would be valued $1.3 million to $6.6 million annually.
The costs of including cement would depend on what requirements
were applied to wet cement workers. OSHA estimates that the costs
associated with existing standards (e.g., requirements for PPE and
hygiene practices) could range from $80 million to $300 million per
year. Placing wet cement within the scope of the standard would cost an
additional $33 million per year for compliance with such provisions as
initial monitoring; those costs would be incurred even if the employer
has no airborne exposures.
PELS: Section F of this preamble summary presented data on the
costs and benefits of alternative PELS for all industries. The full FEA
contains detailed data on the impacts on small firms at each PEL.
The SBREFA Panel also suggested alternatives to a uniform PEL
across all industries and exposures. The Panel recommended that OSHA
consider alternative approaches to industries that are intermittent
users of Cr(VI). OSHA has adopted the concept of permitting employers
with intermittent exposures to meet the requirements of the standard
using respirators rather than engineering controls. This approach has
been used in other standards and does not require workers to routinely
wear respirators.
The SBREFA Panel also recommended considering Separate Engineering
Control Airborne Limits (SECALs). OSHA has adopted this approach for
applications in the aerospace industry. OSHA considered a SECAL for
electroplating when the Agency was considering setting PELs lower than
5, but found a SECAL would not significantly lower costs because
respirator use would be almost as expensive as using engineering
controls. The expense of respirator use would also be a problem with
SECALs for this sector at any PEL. OSHA's reasons for not using the
SECAL approach in other sectors are provided in the Summary and
Explanation. The SBREFA Panel also suggested that OSHA consider
different PELs for different Cr(VI) compounds leading to exposure to
Cr(VI). This issue is fully discussed in VI. Quantitative Risk
Assessment. Here, it will only be noted that this would result in lower
PELs than OSHA is setting in at least some industries, and thus
potentially increase impacts on some small businesses.
Special Approaches to the Shipyard and Construction Industries: The
SBREFA Panel was concerned that changing work conditions in the
shipyard and construction industry would make it difficult to apply
some of the provisions that OSHA suggested at the time of the Panel.
OSHA has decided to change its approach in these sectors. OSHA is
proposing three separate standards, one for general industry, one for
construction, and one for shipyards. OSHA initially proposed that, in
shipyards and construction, medical surveillance would be required only
for persons with signs and symptoms, and regulated areas would not be
required. In the final standard, OSHA has provided for the same medical
surveillance standard in all sectors. The reasons for doing this are
discussed in the Summary and Explanation section of the Preamble.
However, employers must still meet the PEL with engineering controls
and work practices where feasible. OSHA's proposed rule did not require
exposure monitoring in the construction and maritime sectors. In light
of comments, OSHA has shifted from this approach to requiring all sectors
to conduct exposure monitoring, but allowing a performance-oriented option
to exposure monitoring.
Timing of the Standard: The SBREFA Panel also recommended
considering a multi-year phase-in of the standard. OSHA has solicited
comment and examined the comments on this issue. OSHA has decided to
allow employers four years (rather than two years) to comply with the
engineering control provisions of the standard. This expanded phase-in
of engineering controls has several advantages from a viewpoint of
impacts on small businesses. First, it reduces the one-time initial
costs of the standard by spreading them out over time. This would be
particularly useful for small businesses that have trouble borrowing
large amounts of capital in a single year. A phase-in is also useful in
the electroplating sector by allowing employers to coordinate their
environmental and occupational safety and health control strategies to
minimize potential costs. See the Summary and Explanation section of
this Preamble for further discussion of this issue.
SBREFA Panel
Table VIII-14 lists all of the SBREFA Panel recommendations and
notes OSHA responses to these recommendations.












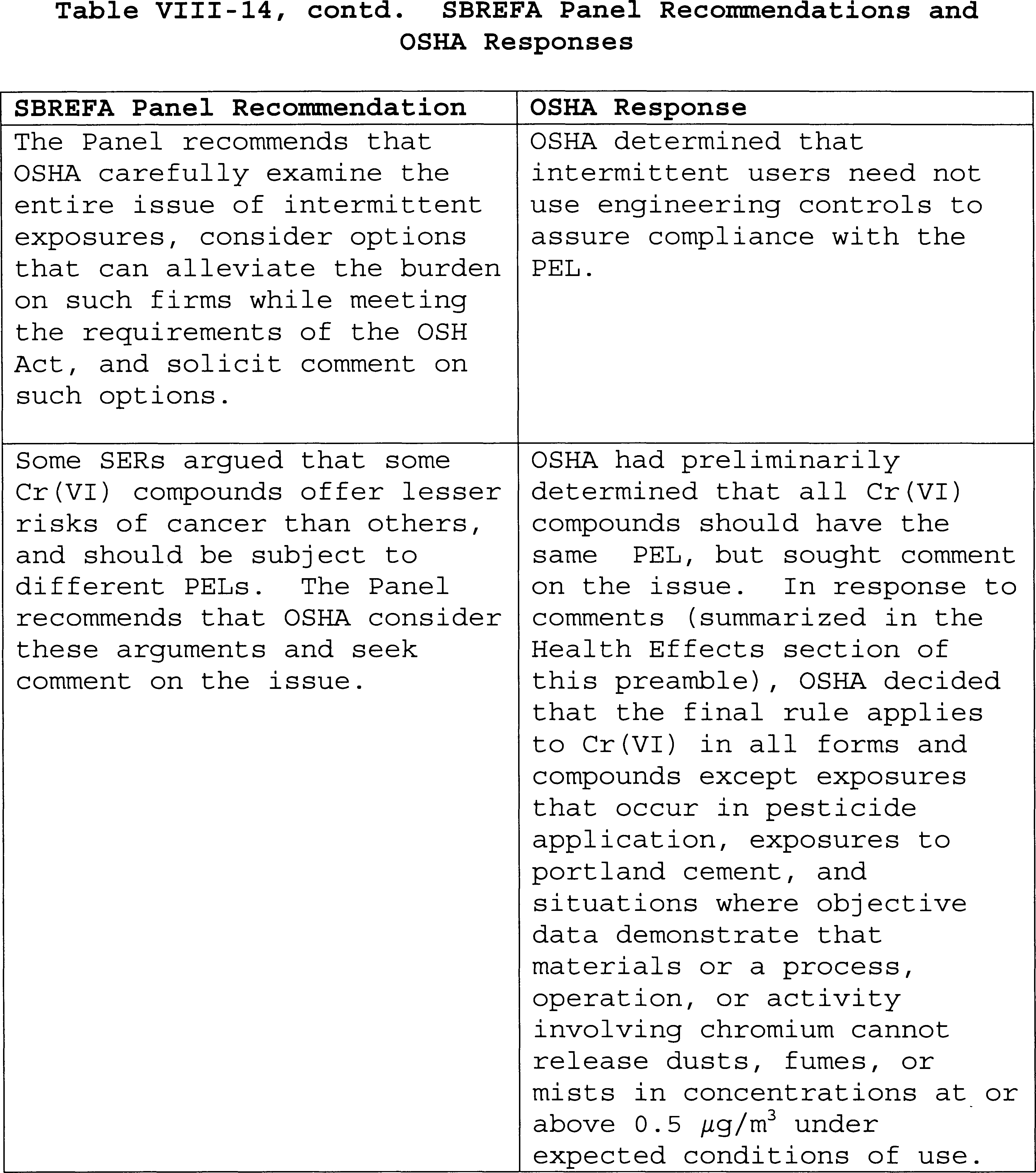


BILLING CODE 5410-26-C
H. Need for Regulation
Employees in work environments addressed by the final standards are
exposed to a variety of significant hazards that can and do cause
serious injury and death. The risks to employees are excessively large
due to the existence of market failures, and existing and alternative
methods of alleviating these negative consequences have been shown to
be insufficient. After carefully weighing the various potential
advantages and disadvantages of using a regulatory approach to improve
upon the current situation, OSHA concludes that in this case the final
mandatory standards represent the best choice for reducing the risks to
employees. In addition, rulemaking is necessary in this case in order
to replace older existing standards with updated, clear, and consistent
health standards.
IX. OMB Review Under the Paperwork Reduction Act of 1995
The final Cr(VI) rule contains collection of information
(paperwork) requirements that are subject to review by the Office of
Management and Budget (OMB) under the Paperwork Reduction Act of 1995
(PRA-95), 44 U.S.C. 3501 et seq., and OMB's regulations at 5 CFR part
1320. The Paperwork Reduction Act defines "collection of information"
as "the obtaining, causing to be obtained, soliciting, or requiring
the disclosure to third parties or the public of facts or opinions by
or for an agency regardless of form or format * * * " (44 U.S.C.
3502(3)(A)). The collection of information requirements (paperwork)
associated with the proposed Cr(VI) rule were submitted to OMB on
October 1, 2004. On November 30, 2004 OMB did not approve the Cr(VI)
paperwork requirements, and instructed OSHA to examine "public comment
in response to the NPRM, including paperwork requirements," and
address any public comments on the paperwork in the preamble. OMB
assigned the control number 1218-0252 for the Agency to use in future
submissions.
The major information collection requirements in the Standard
include conducting employee exposure assessment (Sec. Sec. 1910.1026
(d)(1)-(3), 1915.1026 (d)(1)-(3), and 1926.1126 (d)(1)-(3)), notifying
employees of their Cr(VI)exposures when employee exposures exceed the
PEL (Sec. Sec. 1910.1026 (d)(4), 1915.1026 (d)(4), and 1926.1126
(d)(4)), providing respiratory protection (Sec. Sec. 1910.1026 (g),
1915.1026 (f), and 1926.1126 (f)), labeling bags or containers of
contaminated protective clothing or equipment (Sec. Sec. 1910.1026
(h)(2), 1915.1026 (g)(2), and 1926.1126 (g)(2)), informing persons who
launder or cleans protective clothing or equipment contaminated with
Cr(VI) of the potential harmful effects (Sec. Sec. 1910.1026 (h)(3),
1915.1026 (g)(3), and 1926.1126 (g)(3)), implementing medical-
surveillance of employees (Sec. Sec. 1910.1026 (k), 1915.1026 (i), and
1926.1126 (i)), providing physician or other licensed health care
professional (PLHCP) with information (Sec. Sec. 1910.1026 (k)(4),
1915.1026 (i)(4), and 1926.1126 (i)(4)), ensuring that employees
receive a copy of their medical-surveillance results (Sec. Sec.
1910.1026 (k)(5), 1915.1026 (i)(5), and 1926.1126 (i)(5)), maintaining
employees' exposure-monitoring and medical-surveillance records for
specific periods, and maintaining historical monitoring and objective
data (Sec. Sec. 1910.1026 (m), 1915.1026 (k), and 1926.1126 (k)). The
collection of information requirements in the rule are needed to assist
employers in identifying and controlling exposures to Cr(VI) in the
workplace, and to address Cr(VI)-related adverse health effects. OSHA
will also use records developed in response to this standard to
determine compliance.
The final rule imposes new information collection requirements for
purposes of the PRA. In response to comments on the proposed rule, OSHA
has revised provisions of the final rule that affect collection of
information requirements. These revisions include:
The final rule exempts exposures to portland cement in
general industry and shipyards;
An exemption is included in the final rule where the
employer can demonstrate that Cr(VI) exposures will not exceed 0.5
[mu]g/m\3\ under any expected conditions;
The final PEL of 5 [mu]g/m\3\ has been revised from the
proposed 1 [mu]g/m\3\;
Requirements for exposure determination have been added to
the construction and shipyard standards, and a performance-oriented
option for exposure determination is included in the standards for each
sector (general industry, construction, and shipyards);
Medical surveillance must be provided to employees exposed
to Cr(VI) above the action level (rather than the PEL) for 30 or more
days per year in general industry, construction, and shipyards;
Requirements to maintain records used for exposure
determination have been added to the construction and shipyard
standards, while requirements for training records have been removed
for all sectors.
OSHA has revised the paperwork package to reflect these changes,
and estimates the total burden hours associated with the collection of
information to be approximately 940,000 and estimates the cost for
maintenance and operation to be approximately $126 million.
Potential respondents are not required to comply with the
information collection requirements until they have been approved by
OMB. OMB is currently reviewing OSHA's request for approval of the
final rule's paperwork requirements. OSHA will publish a subsequent
Federal Register document when OMB takes further action on the
information collection requirements in the Cr(VI) rule.
X. Federalism
The Agency reviewed the final Cr(VI) standard according to the most
recent Executive Order on Federalism (Executive Order 13132, 64 FR
43225, August 10, 1999). This Executive Order requires that federal
agencies, to the extent possible, refrain from limiting state policy
options, consult with states before taking actions that restrict their
policy options, and take such actions only when clear constitutional
authority exists and the problem is of national scope. The Executive
Order allows federal agencies to preempt state law only with the
expressed consent of Congress; in such cases, federal agencies must
limit preemption of state law to the extent possible. Under section 18
of the Occupational Safety and Health Act (the "Act" or "OSH Act"),
Congress expressly provides that OSHA preempt state occupational safety
and health standards to the extent that the Agency promulgates a
federal standard under section 6 of the Act. Accordingly, under section
18 of the Act OSHA preempts state promulgation and enforcement of
requirements dealing with occupational safety and health issues covered
by OSHA standards unless the state has an OSHA approved occupational
safety and health plan (i.e., is a state-plan state) [see Gade v.
National Solid Wastes Management Association, 112 S. Ct. 2374 (1992)].
Therefore, with respect to states that do not have OSHA-approved plans,
the Agency concludes that this final rule falls under the preemption
provisions of the Act. Additionally, section 18 of the Act prohibits
states without approved plans from issuing citations for violations of
OSHA standards; the Agency finds that this final rulemaking does not
expand this limitation. OSHA has authority under Executive Order 13132
to promulgate a Cr(VI) standard because the problems addressed by these
requirements are national in scope.
As explained in section VII of this preamble, employees face a
significant risk from exposure to Cr(VI) in the workplace. These
employees are exposed to Cr(VI) in general industry, construction, and
shipyards. Accordingly, the final rule would establish requirements for
employers in every state to protect their employees from the risks of
exposure to Cr(VI). However, section 18(c)(2) of the Act permits state-
plan states to develop their own requirements to deal with any special
workplace problems or conditions, provided these requirements are at
least as effective as the requirements in this final rule.
XI. State Plans
The 26 states and territories with their own OSHA-approved
occupational safety and health plans must adopt comparable provisions
within six months of the publication date of the final hexavalent
chromium standard. These states and territories are: Alaska, Arizona,
California, Hawaii, Indiana, Iowa, Kentucky, Maryland, Michigan,
Minnesota, Nevada, New Mexico, North Carolina, Oregon, Puerto Rico,
South Carolina, Tennessee, Utah, Vermont, Virginia, Virgin Islands,
Washington, and Wyoming. Connecticut, New Jersey and New York have OSHA
approved State Plans that apply to state and local government employees
only. Until a state-plan state promulgates its own comparable
provisions, Federal OSHA will provide the state with interim
enforcement assistance, as appropriate.
XII. Unfunded Mandates
The Agency reviewed the final Cr(VI) standard according to the
Unfunded Mandates Reform Act of 1995 (UMRA) (2 U.S.C. 1501 et seq.) and
Executive Order 12875. As discussed in section VIII of this preamble,
OSHA estimates that compliance with this final rule would require
private-sector employers to expend about $288 million each year.
However, while this final rule establishes a federal mandate in the
private sector, it is not a significant regulatory action within the
meaning of section 202 of the UMRA (2 U.S.C. 1532). OSHA standards do
not apply to state and local governments, except in states that have
voluntarily elected to adopt an OSHA-approved state occupational safety
and health plan. Consequently, the provisions of the final rule do not
meet the definition of a "Federal intergovernmental mandate" [see
section 421(5) of the UMRA (2 U.S.C. 658(5))]. Therefore, based on a
review of the rulemaking record, the Agency believes that few, if any,
of the employers affected by the final rule are state, local, or tribal
governments. Therefore, the Cr(VI) requirements promulgated herein do
not impose unfunded mandates on state, local, or tribal governments.
XIII. Protecting Children From Environmental Health and Safety Risks
Executive Order 13045 requires that Federal agencies submitting
covered regulatory actions to OMB's Office of Information and
Regulatory Affairs (OIRA) for review pursuant to Executive Order 12866
must provide OIRA with (1) an evaluation of the environmental health or
safety effects that the planned regulation may have on children, and
(2) an explanation of why the planned regulation is preferable to other
potentially effective and reasonably feasible alternatives considered
by the agency. Executive Order 13045 defines "covered regulatory
actions" as rules that may (1) be economically significant under
Executive Order 12866 (i.e., a rulemaking that has an annual effect on
the economy of $100 million or more, or would adversely affect in a
material way the economy, a sector of the economy, productivity,
competition, jobs, the environment, public health or safety, or state,
local, or tribal governments or communities, and (2) concern an
environmental health risk or safety risk that an agency has reason to
believe may disproportionately affect children. In this context, the
term "environmental health risks and safety risks" means risks to
health or safety that are attributable to products or substances that
children are likely to come in contact with or ingest (e.g., through
air, food, water, soil, product use). The final Cr(VI) standard is
economically significant under Executive Order 12866 (see section VIII
of this preamble). However, after reviewing the final
Cr(VI) standard, OSHA has determined that the standard would not impose
environmental health or safety risks to children as set forth in
Executive Order 13045. The final standard requires employers to limit
employee exposure to Cr(VI) and take other precautions to protect
employees from adverse health effects associated with exposure to
Cr(VI). To the best of OSHA's knowledge, no employees under 18 years of
age work under conditions that involve exposure to Cr(VI). However, if
such conditions exist, children who are exposed to Cr(VI) in the
workplace would be better protected from exposure to Cr(VI) under the
final rule than they are currently. Based on this determination, OSHA
believes that the final Cr(VI) standard does not constitute a covered
regulatory action as defined by Executive Order 13045.
XIV. Environmental Impacts
The Agency reviewed the final Cr(VI) standard according to the
National Environmental Policy Act (NEPA) of 1969 (42 U.S.C. 4321 et
seq.), the regulations of the Council on Environmental Quality (40 CFR
part 1500), and the Department of Labor's NEPA procedures (29 CFR part
11).
As a result of this review, OSHA has made a final determination
that the final Cr(VI) standard will have no impact on air, water, or
soil quality; plant or animal life; the use of land or aspects of the
external environment. Therefore, OSHA concludes that the final Cr(VI)
standard will have no significant environmental impacts.
XV. Summary and Explanation of the Standards
(a) Scope
OSHA is issuing separate standards addressing hexavalent chromium
(also referred to as chromium (VI) or Cr(VI)) exposure in general
industry, construction, and shipyards. The standard for shipyards also
applies to marine terminals and longshoring. The standards for
construction and shipyards are very similar to each other, but differ
in some respects from the standard for general industry. OSHA believes
that certain conditions in these two sectors warrant requirements that
are somewhat different than those that apply to general industry. This
summary and explanation will describe the final rule for general
industry and will note differences between it and the standards for
construction and shipyards.
Commenters were generally supportive of OSHA's decision to propose
separate standards for general industry, construction, and shipyards
(e.g., Exs. 38-199-1; 38-212; 38-214; 38-220-1; 38-236; 38-244; 39-19),
although one commenter believed that a single standard should apply to
all sectors (Ex. 39-51). Where concerns were expressed about the
establishment of separate standards, they focused on the provisions of
the standards and their application, rather than the concept of
establishing separate standards. Some commenters argued that certain
activities or industries should be covered by the construction standard
rather than the general industry standard (e.g., Exs. 38-203; 38-228-1,
p. 18; 39-52-2; 39-56); others considered the proposed construction and
shipyard standards to be less protective than the proposed general
industry standard (Exs. 38-222; 39-71; 47-23, pp. 16-17; 47-28).
OSHA has long recognized a distinction between the construction and
general industry sectors, and has issued standards specifically
applicable to construction work under 29 CFR Part 1926. The Agency has
provided a definition of the term "construction work" at 29 CFR
1910.12(b), has explained the terms used in that definition at 29 CFR
1926.13, and has issued numerous interpretations over the years
explaining the classification of activities as either general industry
or construction. OSHA recognizes that in some circumstances, general
industry activities and conditions in workplaces where general industry
tasks are performed may be comparable to those found in construction.
However, the Agency believes the longstanding delineation between
sectors is appropriate. The distinction between sectors is generally
well understood by both OSHA enforcement personnel and the regulated
community, and any attempt to create exceptions or to provide different
criteria in this final rule would not improve upon the current criteria
but would rather cause confusion.
OSHA is issuing the construction and shipyard standards to account
for the particular conditions found in those sectors. The Agency
intends to ensure that Cr(VI)-exposed workers in construction and
shipyards are provided protection that, to the extent feasible, is
comparable to the protection afforded workers in general industry. OSHA
believes that concerns raised about differences between the Cr(VI)
proposed standard for general industry and the proposed standards for
construction and shipyards will be lessened because the final standards
are more consistent with one another than as originally proposed.
Specifically, OSHA proposed explicit exposure assessment requirements
for general industry, but not for construction and shipyard workplaces.
The requirements of the final rule for exposure determination are
nearly identical for all sectors (see discussion of exposure
determination under paragraph (d) of this section). In addition, OSHA
proposed a requirement for periodic medical examinations in general
industry, but not in construction and shipyards. The final rule
includes requirements for periodic medical examinations in all sectors
(see discussion of medical surveillance requirements under paragraph
(k) of this section). The final standards for construction and
shipyards provide the most adequate protection within the constraints
of feasibility.
The final rule applies to occupational exposures to Cr(VI), that
is, any chromium species with a valence of positive six, regardless of
form or compound. Examples of Cr(VI) compounds include chromium oxide
(CrO2), ammonium dichromate
((NH4)2Cr2O7), calcium
chromate (CaCrO4), chromium trioxide (CrO3), lead
chromate (PbCrO4), potassium chromate
(K2CrO4), potassium dichromate
(K2Cr2O7), sodium chromate
(Na2CrO4), strontium chromate
(SrCrO4), and zinc chromate (ZnCrO4).
Some commenters supported the proposal to include all chromium
compounds within the scope of the new rule. (See, e.g., Exs. 38-214;
39-60). Other commenters, however, contended that specific Cr(VI)
compounds should be excluded from the scope of the final rule. Notably,
the Color Pigments Manufacturers Association and Dominion Colour
Corporation argued that differences in the bioavailability and toxicity
of lead chromate pigments when compared to other Cr(VI) compounds
warrant unique treatment (Exs. 38-201; 38-205). The Boeing Company also
argued that OSHA should consider the bioavailability of different
Cr(VI) compounds (Ex. 38-106). Boeing indicated that exposures to
strontium chromate and zinc chromate used in aerospace manufacturing
are not equivalent to Cr(VI) exposures in other industries.
OSHA considers all Cr(VI) compounds to be carcinogenic. This
conclusion is based upon careful consideration of the epidemiological,
animal, and mechanistic evidence in the rulemaking record, and is
discussed in section V, "Health Effects," of this preamble. OSHA's
conclusion that all Cr(VI) compounds are carcinogenic is consistent
with the findings of IARC, NTP, and NIOSH. These organizations have
each found Cr(VI) compounds to be carcinogenic, without exception.
OSHA therefore sees no reason to exempt any Cr(VI) compounds from the final rule.
Several commenters argued that existing standards provide adequate
protection for employees exposed to Cr(VI), citing in particular OSHA's
current welding and lead standards (Exs. 38-203; 38-254; 38-124; 39-19;
39-47; 39-48; 39-52, p. 22; 39-54; 39-56). However, none of these
standards provide the full range of protections afforded by the Cr(VI)
rule. For example, OSHA's welding requirements (29 CFR Subpart Q for
general industry; 1926 Subpart J for construction; 1915 Subpart D for
shipyards) include provisions for ventilation, but do not address other
aspects of worker protection included in the Cr(VI) rule such as
exposure determination or medical surveillance. OSHA's lead standards
(29 CFR 1910.1025 for general industry; 29 CFR 1926.62 for
construction) have a PEL of 50 [mu]g/m\3\, which effectively limits
Cr(VI) exposure from lead chromate to 12.5 [mu]g/m\3\; however, this
value is more than double the PEL in the Cr(VI) rule. Other standards
therefore do not provide protection equivalent to the final Cr(VI)
rule. Moreover, even though other requirements may affect Cr(VI)
occupational exposure, Cr(VI) exposure in the current workplace still
results in a significant risk that can be substantially reduced in a
feasible manner by the requirements of this final rule.
Portland Cement
The final rule does not cover exposure to Cr(VI) in portland
cement. OSHA proposed to exclude exposure to portland cement in
construction; the final rule extends this exclusion to all sectors. In
the proposal, OSHA identified two general industry application groups
where all employee exposure to Cr(VI) is from portland cement: Portland
Cement Producers and Precast Concrete Products. (A third application
group, Ready-Mixed Concrete, was later identified.) OSHA proposed to
cover exposures to portland cement in general industry because the
Agency's preliminary exposure profile indicated that some employees in
these application groups were exposed to Cr(VI) levels associated with
a significant risk of lung cancer. However, evidence in the record
indicating the low Cr(VI) content of portland cement has led OSHA to
conclude that the current PEL for portland cement effectively limits
inhalation exposures from work with portland cement.
Cement ingredients (clay, gypsum, and chalk), chrome steel grinders
used to crush ingredients, refractory bricks lining the cement kiln,
and ash may serve as sources of chromium that may be converted to
Cr(VI) during kiln heating, leaving trace amounts of Cr(VI) in the
finished product (Ex. 35-317, p. 148). The amount of Cr(VI) in American
portland cement is generally less than 20 g Cr(VI)/g cement (Exs. 9-57;
9-22; 35-417). Because the Cr(VI) concentration in portland cement is
so low, OSHA's current PEL for portland cement (15 mg/m\3\ for total
dust, 29 CFR 1910.1000) effectively limits the Cr(VI) inhalation
exposure from cement to levels below the new Cr(VI) PEL and Action
Level (i.e., if an employee is exposed at the PEL for portland cement
and the Cr(VI) concentration in that cement is below 20 [mu]g/g, the
employee's exposure to Cr(VI) will be below 0.3 [mu]g/m\3\ ). Because
the evidence in the record demonstrates that current requirements for
portland cement are as protective as the new PEL with regard to Cr(VI)
inhalation exposures, OSHA considers it reasonable to exclude portland
cement from the scope of the final rule. This position was supported by
a number of commenters (e.g., Exs. 38-127; 38-217; 38-227; 38-229; 38-
235).
A number of other commenters, including over 200 laborers,
requested that portland cement be covered under the scope of the final
rule (e.g., Exs. 38-10; 38-35; 38-50; 38-110; 38-222). These comments
generally, but not exclusively, focused on dermal hazards associated
with exposure to portland cement. For example, the Building and
Construction Trades Department, AFL-CIO (BCTD) stated:
To provide construction employees with protection from
predictable exposures to hexavalent chromium, the construction
standard must include portland cement within its scope. Portland
cement represents both a dermal and inhalation hazard in
construction, and reduction of exposures would greatly benefit
construction employees (Ex. 38-219).
Commenters favoring coverage of portland cement in the final rule
argued that a number of the proposal's provisions would serve to
protect cement workers, such as requirements for appropriate protective
clothing (Exs. 47-26, pp. 26-27; 35-332, pp. 22-23; 40-4-2, p. 20),
hygiene facilities (particularly washing facilities)(Exs. 38-219-1, p.
14; 47-26, pp. 26-27; 35-332, p. 19; 40-4-2, p. 19), and training and
education (Exs. 47-26, pp. 26-27; 35-332, p. 19; 40-4-2, p. 19). Some
commenters also favored medical surveillance requirements for workers
exposed to portland cement (38-219-1, p. 18; 47-26, pp. 26-27) and
requirements to reduce the Cr(VI) content of portland cement through
the addition of ferrous sulfate (Exs. 38-199-1, p. 43; 38-219-1, p. 14-
15; 38-222; 35-332, p. 23-24). Some noted that OSHA's Advisory
Committee on Construction Safety and Health had recommended that the
Agency apply certain provisions of the Cr(VI) rule to portland cement
exposures in construction (Ex. 38-199-1, p. 30).
The primary intent of this rule is to protect workers from lung
cancer resulting from inhalation of Cr(VI). The Agency has established
that exposure to Cr(VI) at the previous PEL results in a significant
risk of lung cancer among exposed workers, and compliance with the new
PEL will substantially reduce that risk. As indicated previously, the
existing PEL for portland cement protects employees against inhalation
of Cr(VI) that is present in portland cement as a trace contaminant.
Therefore, OSHA does not believe further requirements addressing
inhalation exposure to Cr(VI) in portland cement are warranted.
The Agency does recognize, however, that in addition to respiratory
effects resulting from Cr(VI) inhalation, Cr(VI) is also capable of
causing serious dermal effects (see discussion in section V of this
preamble). In previous chemical-specific health standards, OSHA
typically has addressed serious health effects associated with exposure
to a chemical, even if those effects are not the focus of the rule. For
example, OSHA issued a standard for cadmium primarily based on lung
cancer and kidney damage associated with inhalation exposures to
cadmium; however, contact with cadmium can also cause irritation of the
skin and OSHA included a provision in the final cadmium rule addressing
protective clothing and equipment to prevent skin irritation. OSHA has
followed a similar approach in the Cr(VI) rule, incorporating
provisions for protective clothing and equipment that will address
potential dermal hazards, and including consideration of dermal effects
in medical surveillance requirements. The Agency believes this is a
reasonable approach to protecting workers when a chemical causes a
variety of adverse health effects.
The dermal hazards from contact with portland cement, however, are
not related solely to the Cr(VI) content of cement. Portland cement is
alkaline, abrasive, and hygroscopic (water-absorbing). Cement
dermatitis may be irritant contact dermatitis induced by these
properties, allergic contact dermatitis elicited by an immunological
reaction to Cr(VI), or a combination of the two (Exs. 35-317; 46-74).
Although reports vary, the weight of the evidence indicates that the vast
majority of cement dermatitis cases do not involve Cr(VI) sensitization
(Ex. 46-74). Dermatitis associated with exposure to portland cement is
thus substantially, perhaps even primarily, related to factors other
than Cr(VI) exposure.
Moreover, OSHA believes that appropriate requirements are already
in place elsewhere in OSHA standards, to protect workers from dermal
effects associated with exposure to portland cement. The Agency has
existing requirements for the provision and use of personal protective
equipment (PPE) (29 CFR 1910.132 for general industry; 29 CFR 1915.152
for shipyards; 29 CFR 1926.95 for construction). These requirements are
essentially equivalent to the requirements of the final Cr(VI) rule
with respect to provision of protective clothing and equipment.
OSHA also has existing requirements for washing facilities that are
comparable to those found in the final Cr(VI) rule (29 CFR 1910.141(d)
for general industry and shipyards; 29 CFR 1926.51(f) for
construction). For example, in operations where contaminants may be
harmful to employees, the Sanitation standard for construction requires
employers to provide adequate washing facilities in near proximity to
the worksite. With only limited exceptions for mobile crews and
normally unattended worksites, lavatories with running water, hand soap
or similar cleansing agents, and towels or warm air blowers must be
made available in all places of employment covered by the standard. The
Sanitation requirements that apply to general industry and shipyards
provide equivalent protections.
OSHA's Hazard Communication standard (29 CFR 1910.1200) requires
training for all employees potentially exposed to hazardous chemicals,
including mixtures such as portland cement. This training must cover
the physical and health hazards of the chemicals and measures employees
can take to protect themselves from these hazards, such as appropriate
work practices, emergency procedures, and personal protective equipment
to be used.
Concerns raised in the record with regard to protective clothing,
washing facilities, and training on cement dermatitis hazards appear to
relate to lack of compliance with these existing requirements, rather
than any inadequacy in the requirements themselves. For example, BCTD
representatives indicated that in spite of current requirements,
washing facilities are rarely provided on construction sites (Tr. 1464,
1470-1471, 1474, 1479-1480). By covering portland cement in the final
Cr(VI) rule, BCTD argued that compliance would improve (Tr. 1519-1522).
OSHA recognizes that reiterating the requirements of generic rules
such as the Sanitation standard in a chemical-specific standard like
the Cr(VI) rule can be useful in some instances by providing employers
with a comprehensive reference of applicable requirements. However, the
Agency does not consider the Code of Federal Regulations to be the best
tool for raising awareness about existing standards. Rather, OSHA
believes guidance documents, compliance assistance efforts, and
enforcement of existing requirements are the best mechanisms for
accomplishing this objective.
Some commenters argued that requirements not included in the
generic standards were needed to protect employees working with
portland cement. The International Brotherhood of Teamsters (IBT)
stated that absent coverage under the standard, portland cement workers
would be responsible for purchasing and maintaining their own PPE. If
there is no requirement for an employer to purchase and provide
required PPE, IBT argued, most employees would elect not to purchase it
(Ex. 38-199-1, p. 30). Of course many employers choose to pay for the
PPE so that they can be sure of its effectiveness. The important
factors are that the PPE must be suitable for the job and must be used
correctly. Moreover, even when employees provide their own protective
equipment, OSHA's PPE standards specify that the employer is
responsible for ensuring its adequacy, including proper maintenance and
sanitation (see 29 CFR 1910.132(b); 29 CFR 1926.95(b)).
Other commenters believed that medical surveillance was needed for
employees exposed to portland cement (Exs. 38-219-1, p. 18; 47-26, pp.
26-27). However, irritant contact dermatitis and allergic contact
dermatitis present the same clinical appearance, and it is difficult to
determine if an employee with dermatitis is sensitized to Cr(VI).
Because cement dermatitis is often related to the irritant properties
of cement rather than Cr(VI), medical surveillance requirements for
portland cement would necessarily involve covering health effects not
solely, or even primarily, attributable to Cr(VI) exposure. OSHA
therefore does not consider a requirement for medical surveillance for
portland cement workers to be appropriate within the context of the
Cr(VI) rule.
Ferrous Sulfate
Finally, some commenters suggested it would be appropriate to
require the addition of ferrous sulfate to portland cement (Exs. 38-
199-1, p. 43; 38-219-1, pp. 14-15; 38-222; 35-332, pp. 23-24; 47-26, p.
8). Cr(VI) concentrations in portland cement can be lowered by the
addition of ferrous sulfate, which reduces Cr(VI) to Cr(III). Residual
Cr(VI) concentrations of less than 2 ppm are typical. As discussed in
section V of this preamble, reports from two researchers suggest that
the addition of ferrous sulfate to cement in Scandinavian countries
reduces the incidence of Cr(VI)-related allergic contact dermatitis in
cement workers (Exs. 9-131; 48-8).
It is reasonable to believe that a reduction in the Cr(VI)
concentration of portland cement would reduce the potential for Cr(VI)-
induced allergic contact dermatitis. However, the lack of available
information regarding a dose-response relationship between Cr(VI)
exposure and allergic contact dermatitis makes it impossible to
estimate how substantial that reduction might be. For instance, a
portion of cement samples already have relatively low Cr(VI)
concentrations. Analyses of 42 samples of American portland cement
reported by Perone et al. indicated that 33 of the samples had Cr(VI)
concentrations below 2 ppm (Ex. 9-57); the benefit of adding ferrous
sulfate to cement with already low Cr(VI) concentrations is unclear.
Moreover, it is not clear that the addition of ferrous sulfate to
cement would be successful in reducing Cr(VI) to Cr(III) under
conditions found in the U.S. Attempts in the U.S. to reduce Cr(VI) in
cement to Cr(III) with ferrous sulfate have been unsuccessful, due to
oxidation of the ferrous sulfate in the production process (Ex. 35-
417). Methods used to handle and store cement have also been shown to
influence the effectiveness of ferrous sulfate in reducing Cr(VI). When
cement is exposed to moisture during storage, the ferrous sulfate in it
is likely to be oxidized, and as a result, the Cr(VI) will not be
reduced to Cr(III) when the cement is mixed with water (Ex. 9-91).
Handling and storage of cement in silos can have this effect (Tr.
1363). Because a substantial amount of cement in the U.S. is produced
in winter and stored for use during warmer weather, ferrous sulfate
added to the cement at the time of production could be oxidized during
that time, rendering it ineffective (Tr. 1363).
Considering this evidence, OSHA does not believe the record
demonstrates that the addition of ferrous sulfate to portland cement in
the U.S. would necessarily result in a reduction in the incidence of
Cr(VI)-induced allergic contact dermatitis. Therefore, OSHA does not
believe that requiring the addition of ferrous sulfate to cement is warranted.
In any event, even if ferrous sulfate was completely effective in
eliminating the potential for Cr(VI)-induced allergic contact
dermatitis from portland cement, the potential for portland cement to
induce irritant contact dermatitis would not be affected. (See section
V(D) of this preamble for additional discussion.) Therefore,
appropriate protective clothing, good hygiene practices, and training
on hazards and control methods would still be necessary and these are
adequately covered by OSHA's generic standards.
Pesticides
The final rule does not cover exposures to Cr(VI) that occur in the
application of pesticides. Some Cr(VI)-containing chemicals, such as
chromated copper arsenate (CCA) and acid copper chromate (ACC), are
used for wood treatment and are regulated by EPA as pesticides. Section
4(b)(1) of the OSH Act precludes OSHA from regulating working
conditions of employees where other Federal agencies exercise statutory
authority to prescribe or enforce standards or regulations affecting
occupational safety or health. Therefore, OSHA specifically excludes
those exposures to Cr(VI) resulting from the application of a pesticide
regulated by EPA from coverage under the final rule.
The exception for exposures that occur in the application of
pesticides was limited to the proposed standard for general industry.
At the time, OSHA was not aware of exposures to Cr(VI) from application
of pesticides in other sectors. Exposures to Cr(VI) from pesticide
application outside of general industry were brought to OSHA's
attention during the public comment period (Exs. 39-47, p. 9; 39-48, p.
4; 39-52). This provision excluding coverage or exposures occurring in
the application of pesticides has therefore been added to the standards
for construction and shipyards as well.
The exemption pertains to the application of pesticides only. The
manufacture of pesticides containing Cr(VI) is not considered pesticide
application, and is covered under the final rule. The use of wood
treated with pesticides containing Cr(VI) is also covered. In this
respect, the Cr(VI) standard differs from OSHA's Inorganic Arsenic
standard (29 CFR 1910.1018). The Inorganic Arsenic standard explicitly
exempts the use of wood treated with arsenic. When the Inorganic
Arsenic standard was issued in 1978, OSHA found that the evidence in
the record indicated "the arsenic in the preserved wood is bound
tightly to the wood sugars, exhibits substantial chemical differences
from other pentavalent arsenicals after reaction, and appears not to
leach out in substantial amounts" (43 FR 19584, 19613 (5/5/78)). Based
on the record in that rulemaking, OSHA did not consider it appropriate
to regulate the use of preserved wood. A number of commenters argued
that a similar exception should be included in the final rule for use
of wood preserved with Cr(VI) compounds (Exs. 38-208; 38-231; 38-244;
43-28). However, OSHA's exposure profile indicates that work with wood
treated with pesticides containing Cr(VI) can involve Cr(VI) exposures
above the new PEL (see FEA, Chapter III). OSHA therefore considers a
blanket exception from the scope of the final rule for use of wood
treated with Cr(VI) to be unjustified.
Other Requested Exemptions
In addition to those who maintained that Cr(VI)-treated wood should
be exempted from the final rule, a number of commenters requested
exemptions from the final rule for other operations or industries
(e.g., welding, electric utilities, Cr(VI) pigment production,
residential construction, and telecommunications (Exs. 38-124; 38-203;
38-205; 38-211; 38-230; 38-244; 38-254; 39-14; 39-15; 39-47; 47-25; 47-
37). OSHA does not believe that the evidence in the record supports a
blanket exception from the final rule for these operations and
industries. In no case have commenters submitted data demonstrating
that the operations or industries for which an exception was requested
do not involve exposures to Cr(VI) that present significant risk to the
health of employees. Rather, the data presented in Chapter III of the
FEA indicate that exposures in these sectors can and do involve
exposures at levels that entail significant risk to workers, and may
exceed the new PEL. OSHA therefore has not included exceptions for
these operations or industries in the final rule.
One commenter argued that the provisions of the standard, including
the new PEL, should apply only where Cr(VI) exposures occur on more
than 30 days per year (Ex. 38-233, pp. 43-44). However, exposures of 30
or fewer days per year may involve cumulative exposures associated with
significant risk of lung cancer. For example, if an employee was
exposed to 50 [mu]g/m3 Cr(VI) for 30 days during a year,
that employee s cumulative exposure for the year would exceed that of
an employee exposed at the new PEL of 5 [mu]g/m3 working
five days a week through the entire year. Therefore, OSHA does not
believe such an exemption is appropriate because it would deny workers
exposed to relatively high levels of Cr(VI) for 30 or fewer days per
year the protections afforded by the Cr(VI) rule. The Agency does
include exceptions from certain requirements of the rule for exposures
occurring on fewer than 30 days per year (e.g., with regard to
requirements for engineering controls and periodic medical
surveillance). However, these exceptions are related to the practical
aspects of implementing protective measures, and not to an absence of
risk for exposures occurring on fewer than 30 days per year.
Other commenters suggested that materials or substances containing
trace amounts of Cr(VI) (e.g., less than 0.1% or 1%) be exempted from
the final rule (Exs. 38-203; 38-254; 39-19; 39-47; 39-48; 39-52; 39-54;
39-56). In particular, some utilities argued that fly ash produced by
the incineration of coal contains trace amounts of Cr(VI) that are so
low as to be insignificant, and that an exclusion from the final rule
for coal ash was warranted (Ex. 39-40). Edison Electric Institute
supported this argument by submitting sampling data and material safety
data sheets that indicated the Cr(VI) concentrations in ash by-products
of the coal combustion process (Exs. 47-25-1; 47-25-2; 47-25-3; 47-25-
4; 47-25-5; 47-25-6; 47-25-7).
OSHA does not believe that it would be appropriate to establish a
threshold Cr(VI) concentration for coverage of substances under the
scope of this final rule. The evidence in the rulemaking record is not
sufficient to lead OSHA to conclude that the suggested concentration
thresholds would be protective of employee health. While OSHA has
recognized that the Cr(VI) content of portland cement is sufficiently
low to warrant an exception from the standard, a threshold
concentration of 0.1% for Cr(VI) would be more than 50-fold higher than
Cr(VI) levels typically found in portland cement (< 0.002%). See above
discussion of the extremely low Cr(VI) concentration in portland cement
(< 20 [mu]g/g).
Although evidence submitted to the record indicates that Cr(VI)
levels in coal ash may be comparable to levels in portland cement, OSHA
does not believe that the evidence is sufficient to establish that all
coal ash from allsources will necessarily have comparable Cr(VI) content.
A threshold concentration is also not reasonable because many
operations where Cr(VI) exposures occur are the result of work with
materials that do not contain any Cr(VI). Welders, who represent nearly
half of the workers covered by this final rule, do not ordinarily work
with materials that contain Cr(VI). Rather, the high temperatures
created by welding oxidize chromium in steel to the hexavalent state.
An exception based on a specified Cr(VI) concentration could be
interpreted to exclude these workers from the scope of the standard.
This would be particularly inappropriate in view of the fact that data
in the record show that many welders have significant Cr(VI) exposures.
OSHA does, however, appreciate the concerns of commenters regarding
situations where they believe exposures are minimal and represent very
little threat to the health of workers. The Agency believes that a
reasonable approach is to have an exception based on Cr(VI) exposure
level. OSHA is therefore including in the final rule an exception for
those circumstances where the employer has objective data demonstrating
that a material containing chromium or a specific process, operation,
or activity involving chromium cannot release dusts, fumes, or mists of
chromium (VI) in concentrations at or above 0.5 [mu]g/m3 as
an 8-hour TWA under any expected conditions of use.
OSHA believes this approach is sensible because it provides an
exception for situations where airborne exposures are not likely to
present significant risk and thus allows employers to focus resources
on the exposures of greatest occupational health concern. The Agency
has added a definition for "objective data" (discussed with regard to
paragraph (b) of the final rule) to clarify what information and data
can be used to satisfy the obligation to demonstrate that Cr(VI)
exposures will be below 0.5 [mu]g/m3.
Other standards which have included similar exceptions (e.g.,
Acryolitrile, 29 CFR 1019.1045; Ethylene Oxide, 29 CFR 1910.1047; 1,3-
Butadiene, 29 CFR 1910.1051) have generally relied upon the action
level as an exposure threshold. A threshold lower than the action level
has been selected for the Cr(VI) rule because OSHA believes this to be
more protective of worker health given the existing significant risk at
the action level. Although OSHA understands the difficulties of
developing objective data to demonstrate that exposures will be below a
given level, the Agency believes that the 0.5 [mu]g/m3
coverage threshold represents an exposure level where it is still
reasonably possible to develop objective data to take advantage of this
exception if Cr(VI) exposure levels are minimal. For instance,
variation in exposures even in well controlled workplaces requires that
typical exposures be below 0.25 [mu]g/m3 in order for an
employer to be reasonably sure that exposures will consistently be
below 0.5 [mu]g/m3 (see Exs. 46-79; 46-80; 46-81). Where
typical exposures are below 0.25 [mu]g/m3, an industry
survey might be used to show that exposures for a given operation would
be below 0.5 [mu]g/m3 under any expected conditions of use.
When using the phrase "any expected conditions of use" OSHA is
referring to situations that can reasonably be foreseen. The criteria
are not intended to be so circumscribed that it is impossible to meet
them. OSHA acknowledges that a constellation of unforeseen
circumstances can occur that might lead to exposures above 0.5 [mu]g/
m3 even when the objective data demonstration has been
correctly made, but believes that such occurrences will be extremely
rare.
(b) Definitions
"Action level" is defined as an airborne concentration of Cr(VI)
of 2.5 micrograms per cubic meter of air (2.5 [mu]g/m3)
calculated as an eight-hour time-weighted average (TWA). The action
level triggers requirements for exposure monitoring and medical
surveillance.
Because employee exposures to airborne concentrations of Cr(VI) are
variable, workers may sometimes be exposed above the PEL even if
exposure samples (which are not conducted on a daily basis) are
generally below the PEL. Maintaining exposures below the action level
provides increased assurance that employees will not be exposed to
Cr(VI) at levels above the PEL on days when no exposure measurements
are made in the workplace. Periodic exposure measurements made when the
action level is exceeded provide the employer with a degree of
confidence in the results of the exposure monitoring. The importance of
the action level is explained in greater detail in the exposure
determination and medical surveillance discussions of this section
(paragraphs (d) and (k) respectively).
As in other standards, the action level has been set at one-half of
the PEL. The Agency has had successful experience with an action level
of one-half the PEL in other standards, including those for inorganic
arsenic (29 CFR 1910.1018), ethylene oxide (29 CFR 1910.1047), benzene
(29 CFR 1910.1028), and methylene chloride (29 CFR 1910.1052).
Following the publication of the proposed rule, which included a
proposed action level of 0.5 [mu]g/m3 (\1/2\ the proposed
PEL of 1 [mu]g/m3), OSHA received several comments
pertaining to the definition of the action level. Commenters such as
the International Brotherhood of Teamsters (IBT) supported OSHA s
preliminary determination that the action level should be set at one-
half the permissible exposure limit (Exs. 38-199-1, p. 9; 38-219, p.
16-17; 38-228-1; 40-10-2). The IBT stated that the action level set at
one-half the PEL has been successful historically in OSHA's standards
such as inorganic arsenic, cadmium, benzene, ethylene oxide,
methylenedianiline, and methylene chloride (Ex. 38-199-1, pp. 9, 44).
NIOSH also supported OSHA's approach, stating that the action level of
one-half the PEL is the appropriate level to indicate sufficient
probability that an employee's exposure does not exceed the PEL on
other days (Ex. 40-10-2, p. 17). The North American Insulation
Manufacturer's Association (NAIMA) agreed that an action level of one-
half the PEL is appropriate (in conjunction with a higher PEL than that
proposed) (Ex. 38-228-1, pp. 23-24).
Previous standards have recognized a statistical basis for using an
action level of one-half the PEL (see, e.g., acrylonitrile, 29 CFR
1910.1045; ethylene oxide, 29 CFR 1910.1047). In brief, OSHA previously
determined (based in part on research conducted by Leidel et al.) that
where exposure measurements are above one-half the PEL, the employer
cannot be reasonably confident that the employee is not exposed above
the PEL on days when no measurements are taken (Ex. 46-80).
Following the publication of the proposed rule, the United
Automobile, Aerospace, and Agricultural Implement Workers of America
(UAW) requested an action level of one-tenth of the permissible
exposure limit (PEL) (Tr. 791; Exs. 39-73; 39-73-2, pp. 3, 10; 40-19-
1). The UAW argued that the lower action level is appropriate because
variability in exposures is greater than was previously believed in
some occupational settings. While OSHA previously assumed a geometric
standard deviation (GSD) of 1.4, the UAW stated that a GSD of 2 should
be assumed as a matter of policy. They concluded that this GSD implies
an action level of one-tenth the PEL to minimize the frequency of
exposures above the PEL on days when measurements are not taken (Ex.
39-73-2, p. 12).
If the variability of workplace exposures is typically as high as
the UAW suggests, an action level less than one-half the PEL would be
required to give employers a high degree of confidence that employees'
exposures are below the PEL on most workdays. Leidel et al., calculated
that for exposures with a GSD of 2.0, an action level of 0.115 times
the PEL would be required to limit to 5% the probability that 5% or
more of an employee's unmeasured daily exposure averages will exceed
the PEL (Ex. 46-80, p. 29). However, the evidence in the record is
insufficient to permit OSHA to conclude that a GSD of 2.0 is typical of
workplace Cr(VI) exposures. Furthermore, while OSHA recognizes the
value of high (95%) confidence that exposures exceed the PEL very
infrequently (< 5%), the Agency believes that the action level should
be set at a value that effectively encourages employers to reduce
exposures below the action level while still providing reasonable
(though possibly < 95%) assurance that workers' exposures are typically
below the PEL. OSHA's experience with past rules and the comments and
testimony of NIOSH and other union representatives indicate that
reasonable assurance of day-to-day compliance with the PEL is achieved
with an action level of one-half the PEL (Exs. 40-10-2, p. 17; 199-1,
pp. 9, 44).
The Agency's experience with previous standards also indicates that
an action limit of one-half the PEL effectively encourages employers,
where feasible, to reduce exposures below the action level to avoid the
added costs of required compliance with provisions triggered by the
action level. Where there is continuing significant risk at the PEL,
the decision in the Asbestos case (Building and Construction Trades
Department, AFL-CIO v. Brock, 838 F. 2d 1258 (D.C. Cir 1988)) indicates
that OSHA should use its legal authority to impose additional
requirements on employers to further reduce risk when those
requirements will result in a greater than de minimus incremental
benefit to workers' health. OSHA believes that the action level will
result in a very real and necessary further reduction in risk beyond
that provided by the PEL alone.
The action level improves employee protection while increasing the
cost-effectiveness and performance orientation of the standard. The
action level will encourage employers who can, in a cost-effective
manner, identify approaches or innovative methods to reduce their
employees' exposures to levels below the action level, because this
will eliminate the costs associated with exposure monitoring and
medical surveillance. The employees of such employers will have greater
protection against adverse health effects because their exposures to
Cr(VI) will be less than half of those permitted by the permissible
exposure limit. Employees of those employers who are not able to lower
exposures below the action level will have the additional protection
provided by medical surveillance, exposure monitoring, and the other
provisions of the standard that are triggered by the action level.
"Chromium (VI) [hexavalent chromium or Cr(VI)]" means chromium
with a valence of positive six, in any form or chemical compound in
which it occurs. This term includes Cr(VI) in all states of matter, in
any solution or other mixture, even if encapsulated by another or
several other substances. The term also includes Cr(VI) when created by
an industrial process, such as when welding of stainless steel
generates Cr(VI) fume.
For regulatory purposes, OSHA is treating Cr(VI) generically,
instead of addressing specific compounds individually. This is based on
OSHA's determination that the toxicological effect on the human body is
similar from Cr(VI) in any of the substances covered under the scope of
this standard, regardless of the form or compound in which it occurs.
As discussed in Section V of this preamble, some variation in potency
may result due to differences in the solubility of compounds. Other
factors, such as encapsulation, may have some effect on the
bioavailability of Cr(VI). However, OSHA believes that these factors do
not result in differences that merit separate provisions for different
Cr(VI) compounds. OSHA considers it appropriate to apply the
requirements of the standard uniformly to all Cr(VI) compounds.
"Emergency" means any occurrence that results, or is likely to
result, in an uncontrolled release of Cr(VI), such as, but not limited
to, equipment failure, rupture of containers, or failure of control
equipment. To constitute an emergency, the exposure to Cr(VI) must be
unexpected and significant. If an incidental release of chromium (VI)
can be controlled at the time of release by employees in the immediate
release area, or by maintenance personnel, it is not an emergency.
Similarly, if an incidental release of Cr(VI) may be safely cleaned up
by employees at the time of release, it is not considered to be an
emergency situation for the purposes of this section. Those instances
that constitute an emergency trigger certain requirements in this
standard (e.g., medical surveillance) that are discussed later in this
section.
In comments submitted to OSHA following the publication of the
proposed Cr(VI) rule, the International Brotherhood of Teamsters (IBT)
disagreed with OSHA's definition of "emergency". IBT stated that all
spills and leaks involving Cr(VI) are unexpected and significant, and
should be considered emergencies (Ex. 38-199-1, pp. 20-21).
OSHA does not agree with the IBT's position that every spill or
leak should be considered an emergency. Not all spills and leaks are
significant; the particular circumstances of the release, such as the
quantity involved, confined space considerations, and the adequacy of
ventilation will have an impact on the amount of Cr(VI) to which
employees are exposed when a spill or leak occurs. For example, a minor
spill that can be quickly cleaned up by an employee with minimal
airborne or dermal exposure to Cr(VI) is clearly not an emergency. In
addition, factors such as the personal protective equipment available,
pre-established standard operating procedures for responding to
releases, and engineering controls that employees can activate to
assist them in controlling and stopping the release are all factors
that must be considered in determining whether a release is incidental
or an emergency.
The IBT also stated that the person who determines whether a spill
or leak constitutes an emergency situation should be qualified with
specific training, knowledge, and experience regarding the hazards
associated with exposure to Cr(VI) and the appropriate response
measures that must be implemented to prevent Cr(VI) exposures during
the spill or leak remediation (Ex. 38-199-1, pp. 20-21). OSHA believes
that the provisions of the Hazard Communication standard adequately
address the IBT's concern (29 CFR 1910.1200). Paragraph (h)(3) of that
standard directs employers to provide employees who are exposed or
potentially exposed to a hazardous chemical (such as Cr(VI)) with
training on the physical and health hazards of the chemical and
[t]he measures employees can take to protect themselves from these
hazards, including specific procedures the employer has implemented
to protect employees from exposure to hazardous chemicals, such as
appropriate work practices, emergency procedures, and personal
protective equipment to be used * * * (29 CFR 1910.1200
(h)(3)(iii)).
The Agency expects that employers and employees equipped with the
training required by the Hazard Communication
standard will be sufficiently knowledgable to determine whether an
emergency has occurred, and that it is not necessary to mandate
additional specialized training for this purpose.
"Employee exposure" means exposure to airborne Cr(VI) that would
occur if the employee were not using a respirator. This definition is
included to clarify the fact that employee exposure is measured outside
any respiratory protection worn. It is consistent with OSHA's previous
use of the term in other standards.
"Historical monitoring data" means data from chromium (VI)
monitoring conducted prior to May 30, 2006, obtained during work
operations conducted under workplace conditions closely resembling the
processes, types of material, control methods, work practices, and
environmental conditions in the employer's current operations. To
demonstrate employees' exposures, historical monitoring data must
satisfy all exposure monitoring requirements of this section (e.g.,
accuracy and confidence requirements).
"Objective data" means information other than employee monitoring
that demonstrates the expected employee exposure to chromium (VI)
associated with a particular product or material or a specific process,
operation, or activity. Types of information that may serve as
objective data include, but are not limited to, air monitoring data
from industry-wide surveys; data collected by a trade association from
its members; or calculations based on the composition or chemical and
physical properties of a material.
"Physician or other licensed health care professional" [PLHCP] is
an individual whose legally permitted scope of practice (i.e., license,
registration, or certification) allows him or her to independently
provide or be delegated the responsibility to provide some or all of
the particular health care services required by the medical
surveillance provisions of this final rule. This definition is
consistent with several recent OSHA standards, including the
respiratory protection standard (29 CFR 1910.134), the bloodborne
pathogens standard (29 CFR 1910.1030), and the methylene chloride
standard (29 CFR 1910.1052). In these standards, the Agency determined
that any professional licensed by state law to do so may perform the
medical evaluation procedures required by the standard. OSHA recognizes
that the personnel qualified to provide the required medical evaluation
may vary from state to state, depending on state licensing laws.
At the public hearing, the 3M Company (3M) expressed concern with
OSHA's interpretation of licensing requirements for PLHCPs. In the
recent standards discussed above, OSHA has interpreted the requirements
to mean that PLHCPs must be licensed in the states of residence for the
employees they evaluate. This interpretation is based on OSHA's
recognition of state licensing laws that require PHLCP's to be licensed
in the state in which they practice. 3M encouraged OSHA to adopt an
expanded definition of PLHCP for the Cr(VI) standard, allowing PLHCPs
licensed in any U.S. state to evaluate employees residing in that or
any other state, arguing that other federal agencies such as the
Department of Transportation permitted similar allowances. 3M argued
that this arrangement " * * * would permit one medical director to
oversee the program in several states" where a company has operations
(Tr. 1592, Ex. 47-36). Moreover, 3M added that OSHA has no authority to
enforce state licensing requirements.
Despite the concerns raised by 3M, OSHA continues to believe that
it is appropriate to establish PLHCP requirements consistent with state
requirements for medical practice. OSHA's goal is that the medical
surveillance provisions of the final Cr(VI) rule be conducted by or
under the supervision of a health care professional who is
appropriately licensed to perform those provisions and is therefore
operating under his or her legal scope of practice. OSHA also continues
to believe that issues regarding a PLCHP's legal scope of practice
reside most appropriately with state licensing boards. While OSHA does
not enforce state licensing requirements (e.g., fining an individual
PHCLP for operating outside their legal state license), OSHA can cite,
using the Cr(VI) standard, an employer for using a health care
professional who is not operating under his or her legal scope of
practice. Thus, the Agency believes that the proposed definition for
PHLCP is reasonable, and has retained it in the final rule. OSHA's
experience with other standards using this definition supports the
Agency's determination in this matter.
"Regulated area" means an area, demarcated by the employer, where
an employee's exposure to airborne concentrations of Cr(VI) exceeds, or
can reasonably be expected to exceed the PEL. This definition is
consistent with the use of the term in other standards, including those
for cadmium (29 CFR 1910.1027), butadiene (29 CFR 1910.1051), and
methylene chloride (29 CFR 1910.1052).
OSHA has not included a requirement for regulated areas in
construction and shipyards. This definition is therefore not included
in the standards for construction and shipyards.
The definitions for "Assistant Secretary", "Director", "High-
efficiency particulate air [HEPA] filter", and "This section" are
consistent with OSHA's previous use of these terms found in other
health standards.
(c) Permissible Exposure Limit (PEL)
Introduction
Paragraph (c) of the final rule establishes an 8-hour time-weighted
average (TWA) exposure limit of 5 micrograms of Cr(VI) per cubic meter
of air (5 [mu]g/m3). This limit means that over the course
of any 8-hour work shift, the average exposure to Cr(VI) cannot exceed
5 [mu]g/m3. The new limit applies to Cr(VI), as opposed to
the previous PEL which was measured as CrO3. The previous
PEL of 1 milligram per 10 cubic meters of air (1 mg/10m3, or
100 [mu]g/m3) reported as CrO3 is equivalent to a
limit of 52 [mu]g/m3 as Cr(VI).
OSHA proposed a PEL of 1 [mu]g/m\3\ for Cr(VI). This PEL was
proposed because the Agency made a preliminary determination that
occupational exposure to Cr(VI) at the previous PEL resulted in a
significant risk of lung cancer among exposed workers, and compliance
with the proposed PEL was expected to substantially reduce that risk.
Based on the information available to OSHA at the time, a PEL of 1
[mu]g/m\3\ was believed to be economically and technologically feasible
for affected industries.
The PEL was a focus of comment in the rulemaking process, revealing
sharply divided opinion on the justification for a PEL of 1 [mu]g/m\3\.
Some support was expressed for the proposed PEL (Exs. 38-199-1, p. 42;
38-219-1, p. 2; 39-73-1). The vast majority of commenters, however, did
not believe the proposed PEL was appropriate. Some maintained that a
higher PEL was warranted, arguing that the proposed limit was
infeasible or was not justified by the health and risk evidence (e.g.,
Exs. 38-205; 38-215; 38-231; 38-228; 38-233). Several commenters
suggested alternative PELs that they considered appropriate, such as 10
[mu]g/m\3\ (Exs. 38-134; 38-135; 38-195; 38-203; 38-212; 38-250; 38-
254), 20 [mu]g/m\3\ (Ex. 38-204), 23 [mu]g/m\3\ (e.g., Exs. 38-7; 43-
22; 43-23; 43-25; 43-39), or 26 [mu]g/m\3\ (Ex. 38-263). Others
maintained that the remaining risk at the proposed PEL was excessive
and believed OSHA should adopt a lower PEL, suggesting 0.2 or 0.25
[mu]g/m\3\ (Exs. 39-71; 40-10-2; 47-23; 47-28).
After careful consideration of the evidence in the rulemaking
record, OSHA has established a final PEL of 5 [mu]g/m\3\. OSHA s
examination of the health effects evidence, discussed in section V of
this preamble, reaffirms the Agency's preliminary conclusion that
exposure to Cr(VI) causes lung cancer, as well as other serious adverse
health effects. OSHA's quantitative risk assessment, presented in
section VI, indicates that the most reliable lifetime estimate of risk
from exposure to Cr(VI) at the previous PEL is 101 to 351 excess lung
cancer deaths per 1000 workers. As discussed in section VII, this
clearly represents a significant risk of material impairment of health.
OSHA believes that lowering the PEL to 5 [mu]g/m\3\ will substantially
reduce this risk. OSHA estimates the lifetime excess risk of death from
lung cancer at the new PEL to be between 10 and 45 per 1000 workers.
The Agency considers the level of risk remaining at the new PEL to
be significant. However, based on evidence evaluated during the
rulemaking process, OSHA has concluded that a uniform PEL of 5 [mu]g/
m\3\ is appropriate. The new PEL is technologically and economically
feasible for all industry sectors. In only two operations within one of
those sectors, the painting of aircraft and large aircraft parts in the
aerospace industry, is a PEL of 5 [mu]g/m\3\ infeasible. In accordance
with section 6(b)(5) of the OSH Act, OSHA has determined that the new
PEL is the lowest limit that employers can generally achieve,
consistent with feasibility constraints. Additional requirements are
included in the final rule to further reduce any remaining risk. OSHA
anticipates that these ancillary provisions will reduce the risk beyond
the reduction that will be achieved by the new PEL alone.
OSHA's rationale for adopting a uniform PEL of 5 [mu]g/m\3\ is set
forth in greater detail below. The discussion is organized around the
issues of primary importance to commenters: (a) Whether a uniform PEL
is appropriate for all chromium compounds, (b) the technologic and
economic feasibility of various PELs, (c) the requirement of section
6(b)(5) to promulgate the most protective standard consistent with
feasibility, and (d) whether there is a need for a short-term exposure
limit.
A Uniform PEL Is Appropriate for All Chromium Compounds
OSHA believes that it is appropriate to establish a single PEL that
applies to all Cr(VI) compounds. OSHA's preferred estimates of risk are
derived from two cohorts of chromate production workers that were
predominantly exposed to sodium chromate and sodium dichromate. A
number of commenters argued that risk estimates from these cohorts were
not applicable to certain other Cr(VI) compounds (Exs. 38-106; 38-201-
1; 38-205; 38-215-2).
After carefully evaluating the epidemiological, animal and
mechanistic evidence in the rulemaking record, OSHA considers all
Cr(VI) compounds to be carcinogenic. (For additional discussion see
section V of this preamble.) OSHA has determined that the risk
estimates developed from the chromate production cohorts are reasonably
representative of the risks expected from equivalent exposures to
different Cr(VI) compounds in other industries. OSHA finds that the
risks estimated from the Gibb and Luippold cohorts of chrome production
workers adequately represent the risks to workers in other industries
who are exposed to equivalent levels of Cr(VI) compounds. (The
rationale supporting these conclusions is discussed in detail in
sections V and VI of this preamble. In particular, see Section VI(H) of
the Quantitative Risk Assessment.) Because OSHA's estimates of risk are
reasonably representative of all occupational Cr(VI) exposures, the
Agency considers it appropriate to establish a single PEL applicable to
all Cr(VI) compounds. A number of rulemaking participants supported
this approach (Exs. 38-214; 38-220; 39-20; 39-60; 40-10; 40-19). See
also, e.g., Color Pigments Mfr. Ass'n, Inc. v. OSHA, 16 F.3d 1157, 1161
(11th Cir. 1994):
Given the absence of definiteness on the issue, the volume of
evidence that points at least implicitly to the dangers of cadmium
pigments, and the serious potential health risks present if cadmium
exposure is as great in pigment form as in other compounds, we
believe that OSHA was justified in choosing to include cadmium
pigments in the PEL * * * ;
Asarco, Inc. v. OSHA, 746 F.2d 483, 495 (9th Cir. 1984) (permissible
for OSHA to "use trivalent arsenic studies and conclusions to support
inclusion of pentavalent arsenic in the standard").
The Final PEL of 5 [mu]g/m\3\ Is Technologically and Economically
Feasible for all Affected Industries; the Proposed PEL Is Not
OSHA has concluded that a PEL of 5 [mu]g/m\3\ is economically and
technologically feasible for all the affected industries. OSHA has also
concluded, based on the comments and evidence submitted to the record,
that the proposed PEL of 1 [mu]g/m\3\ is not feasible in all
industries. OSHA's feasibility determinations are explained below.
Technologic feasibility of the final PEL. In making its
determination of technological feasibility, OSHA relied upon guidance
provided by the courts that have reviewed previous standards. In
particular, the decision of the U.S. Court of Appeals for the District
of Columbia on OSHA's Lead standard (United Steelworkers of America v.
Marshall, 647 F.2d 1189 (D.C. Cir. 1981)) established a benchmark that
the Agency has relied on for evaluating technological feasibility. The
court explained that OSHA has "great discretion * * * in determining
the feasibility of a chosen PEL." 647 F.2d at 1309. Both technological
and economic feasibility are "to be tested industry-by-industry." 647
F.2d at 1301. In order to establish that a standard is technologically
feasible, "OSHA must prove a reasonable possibility that the typical
firm will be able to develop and install engineering and work practice
controls that can meet the PEL in most of its operations." 647 F.2d at
1272. The court allowed that "insufficient proof of technological
feasibility for a few isolated operations within an industry, or even
OSHA's concession that respirators will be necessary in a few such
operations, will not undermine" OSHA's finding of technological
feasibility. Id.
Applying this definition of feasibility, OSHA has evaluated each
affected industry and has concluded that a PEL of 5 [mu]g/m\3\ can be
achieved through engineering and work practice controls, with only
limited respirator use, in every industry. The primary evidentiary
support for this conclusion is the report of Shaw Environmental, Inc.,
discussed in depth in the Final Economic and Regulatory Flexibility
Analysis (FEA). Based on the data collected by Shaw, OSHA concludes
that engineering controls, such as local exhaust ventilation (LEV),
process control, and process modification or substitution can be used
to control exposures in most operations.
OSHA recognizes that there are certain instances in which
supplemental respirator use will be required because engineering and
work practice controls are not always sufficient to reduce airborne
exposures below the PEL. Summary information regarding the extent of
respirator usage expected at various potential PELs is presented in
Table VIII-3 (see section VIII, summary of the FEA). Considering this
information together with other data and analysis presented in the FEA,
OSHA has concluded that a PEL of 5 [mu]g/m\3\ is technologically feasible
in all affected industry sectors and in virtually all operations, with the
limited exception of some aerospace painting operations discussed more fully
below. In only three sectors would respirator use be required by more than
5% of exposed employees. In two of these sectors, chromate pigment producers and
chromium dye producers, use of respirators will be intermittent. The
third sector, stainless steel welding, presents technological
challenges in certain operations. However, the new PEL can clearly be
achieved in most operations with engineering and work practice
controls.
OSHA recognizes that for two distinct operations within the
aerospace industry, painting aircraft and painting large aircraft
parts, engineering and work practice controls cannot control exposures
below 25 [mu]g/m\3\ and respirators would be required for most
employees performing these operations. (See additional discussion of
aerospace painting below.) For that reason OSHA is adopting a provision
for those specific operations requiring employers to use engineering
and work practice controls to limit employee exposures to 25 [mu]g/
m\3\. Respiratory protection must then be used to achieve the PEL.
OSHA did not set the PEL at 25 [mu]g/m\3\, a level achievable in
every operation in every industry with engineering and work practice
controls alone. That approach is inappropriate because it would leave
the vast majority of affected employees exposed to Cr(VI) levels above
those that could feasibly be achieved in most industries and
operations. As discussed above, the lower PEL of 5 [mu]g/m\3\ is
feasible within the meaning of the case law, although it will result in
limited use of respirators in some industries and significant
respirator use in two painting operations in the aerospace industry.
The two aerospace painting operations with significant respirator use
are covered by the provision discussed above. For those operations,
OSHA weighed the added protection provided by respirators against the
negative aspects of respiratory protection requirements, and decided
that the additional respirator use was acceptable.
Technological feasibility of the proposed PEL. OSHA concludes that
the proposed PEL of 1 [mu]g/m\3\ is not technologically feasible for
all industries under the criteria in the D.C. Circuit's Lead decision.
The court's definition of technological feasibility recognizes that for
a standard based on a hierarchy of controls, a particular PEL is not
technologically feasible simply because it can be achieved through the
widespread use of respirators. 647 F.2d at 1272. This is consistent
with OSHA's long-held view that it is prudent to avoid requirements
that will result in extensive respirator use.
In its post-hearing brief, Public Citizen argued that a PEL should
be considered technologically feasible if respirator use would be
necessary to achieve compliance in a significant number of operations
within an industry, or even if the PEL could only be achieved through
use of respirators alone (Ex. 47-23, pp. 12-15). That position is
inconsistent with the established test for feasibility for standards
based on the hierarchy of controls. Moreover, as discussed in the
preamble explanation of paragraph (f) on methods of compliance, use of
respirators in the workplace presents a number of independent safety
and health concerns. The vision of workers wearing respirators may be
diminished, and respirators can impair the ability of employees to
communicate with one another. Respirators can impose physiological
burdens on employees due to the weight of the respirator and increased
breathing resistance experienced during operation. The level of
physical work effort required, the use of protective clothing, and
environmental factors such as temperature extremes and high humidity
can interact with respirator use to increase the physiological strain
on employees. Inability to cope with this strain as a result of medical
conditions such as cardiovascular and respiratory diseases, reduced
pulmonary function, neurological or musculoskeletal disorders, impaired
sensory function, or psychological conditions can place employees at
increased risk of illness, injury, and even death. Routine use of
respirators for extended periods of time is regarded by the Agency to
be of greater significance than intermittent use for short time
periods.
OSHA also believes that respirators are inherently less reliable
than engineering and work practice controls. To consistently provide
adequate protection, respirators must be appropriately selected and
fitted, properly used, and properly maintained. Because these
conditions can be difficult to attain, and are subject to human error,
OSHA does not believe respirators provide the same degree of protection
as do engineering and work practice controls.
Based on evidence and comment submitted in response to the
proposal, OSHA finds that a PEL of 1 [mu]g/m\3\ is not technologically
feasible for a substantial number of industries and operations
employing a large number of the workers covered by the standard. The
record shows that a PEL of 1 [mu]g/m\3\ is technologically infeasible
for welding and aerospace painting because engineering and work
practice controls cannot reduce exposures below 1 [mu]g/m\3\ for many
operations. OSHA also finds that the record contains insufficient
evidence to establish the technologic feasibility of the proposed PEL
for four other industries: chromate pigment producers, chromium
catalyst producers, chromium dye producers and some hard chrome
electroplaters. OSHA's findings on the technologic feasibility of the
proposed PEL are summarized below, and are discussed more extensively
in Chapter III of the FEA (in particular, see section titled:
"Technological Feasibility of the Proposed 1 [mu]g/m\3\ 8-Hour TWA
PEL.").
Welding. OSHA has concluded that a PEL of 1 [mu]g/m\3\ is not
technologically feasible for shielded metal arc welding (SMAW) on
stainless steel because engineering and work practice controls cannot
generally reduce employee exposures to below 1 [mu]g/m\3\. Almost one
third (29%) of all stainless steel SMAW operations would need to use
respirators at a PEL of 1 [mu]g/m\3\. In general industry alone, more
than half (52%) of stainless steel SMAW processes would be unable to
use engineering or work practice controls to reduce Cr(VI) exposures
below 1 [mu]g/m\3\. Notably, stainless steel welding is widespread
throughout the economy; it occurs in over 20,000 establishments
employing approximately 127,000 workers in over sixty-five 3-digit
NAICS codes. SMAW is the most common type of stainless steel welding
and is performed by more than 67,000 employees--more than half of the
total number of stainless steel welders and one quarter of all welders
covered by the standard.
OSHA initially recommended the substitution of gas metal arc
welding (GMAW) for SMAW as the cheapest and most effective method to
reduce Cr(VI) exposures. GMAW, like SMAW, is a common type of welding,
but GMAW tends to produce lower exposures than SMAW. However, based on
hearing testimony and evidence submitted to the record, OSHA now
believes that only 60% of SMAW operations can switch to GMAW (Exs. 38-
220-1, p. 8; 39-60, p. 3; 39-70, p. 2; 35-410, p. 4). Moreover, even
among the SMAW operations with current exposures above 1 [mu]g/m\3\
that can switch to GMAW, only a portion (40% in general industry and
59% in construction and maritime)
would be able to achieve a PEL of 1 [mu]g/m\3\ without respirators.
OSHA has also determined that a PEL of 1 [mu]g/m\3\ is
technologically infeasible for stainless steel welding that is
performed in confined or enclosed spaces due to limitations on the
availability of ventilation. Because engineering and work practice
controls cannot consistently reduce exposures to below 1 [mu]g/m\3\, a
large percentage of stainless steel welding operations in confined or
enclosed spaces would require respirators at a PEL of 1 [mu]g/m\3\. In
general industry, for example, 60% of welding tasks done on stainless
steel in confined spaces would be unable to comply with the proposed
PEL by using engineering or work practice controls.
In sum, OSHA has concluded that it is infeasible for some of the
most common welding operations to achieve a PEL of 1 [mu]g/m\3\. For a
more detailed explanation of OSHA's technological feasibility analysis
for welding operations, see Chapter III of the FEA. OSHA has also
decided that although it may be feasible for some of the less common
types of welding operations to achieve a PEL of 1 [mu]g/m\3\ with
engineering and work practice controls, the ubiquitous nature of
welding necessitates a finding that a PEL of 1 [mu]g/m\3\ is generally
infeasible for all welding operations. In particular, OSHA believes
that the proposed PEL is infeasible for welding operations generally
because welding is not easily separated into high and low exposure
operations. Welders may perform different types of welding in the same
day, making it difficult or impossible for employers to monitor them on
an operation by operation basis. See, e.g., Ex. 39-22. In addition,
because workers doing different types of welding often work alongside
one another, what is technologically feasible for a welding operation
considered in isolation may not be technologically feasible for that
operation when it is performed next to SMAW on stainless steel or
another operation for which a PEL of 1 [mu]g/m\3\ is technologically
infeasible.
Welding occurs in over 40,000 establishments spanning sixty-five
different 3-digit NAICS codes. Welding is done in a variety of sites
throughout many diverse workplaces (Ex. 38-8, p. 5). Stainless steel
SMAW is commonly done in close proximity to other welding or cutting
operations, which could expose nearby workers to the higher exposures
generated by the SMAW welder (Ex. 38-214, p. 7). The Specialty Steel
Industry of North America commented that, "workers in job categories
other than those evaluated by OSHA may spend significant time in areas
of potential exposure" (Ex. 38-233, p. 10). The Integrated Waste
Services Association similarly indicated that inspectors, scaffold
workers, laborers, pipe fitters, and refractory workers may pass
through areas with potential Cr(VI) exposure during nickel chrome alloy
overlay (Ex. 38-258, p. 2). The Building and Construction Trades
Department of the AFL-CIO also stated that "workers may be exposed to
hazards even if they are not directly performing tasks associated with
Cr VI exposure via close proximity exposure" (Ex. 31-6-1).
Moreover, OSHA is aware that welders sometimes weld in many
different environments on a variety of types of base metal using
different welding methods in the course of a project or even during a
single work shift (Exs. 34-10, 38-235). In those situations, the
employee's overall exposure levels are inevitably influenced by the
variety of exposures present during the various welding tasks he or she
performs. Therefore, depending on how much time the employee spends
doing welding operations for which a PEL of 5 [mu]g/m\3\ is the lowest
feasible level, even the use of engineering and work practice controls
to comply with a PEL of 1 [mu]g/m\3\ in the other welding operations
would not necessarily reduce the employee's overall exposure levels
below that mark.
Because of these factors, welding is not easily separated into high
and low exposure operations in the real work site. For these reasons,
OSHA believes the record demonstrates that the proposed PEL of 1 [mu]g/
m\3\ is infeasible for welding operations generally. Almost 270,000 of
the employees covered by the new standard engage in these welding
operations (Table VIII-2).
Aerospace painting. There are approximately 8300 exposed employees
in aerospace painting (Table VIII-2). A PEL of 1 [mu]g/m\3\ is not
feasible for approximately two thirds of all aerospace painting
operations. At a PEL of 5 [mu]g/m\3\, only \1/3\ of aerospace painting
operations would require substantial respirator use.
Exposures in aerospace painting are controlled by enclosing the
operations in painting booths or dedicated rooms with LEV. This is
feasible for small parts, but as the size of the parts increases it
becomes more difficult to control exposures. For example, when painting
most small parts, exposures below 1 [mu]g/m\3\ are achievable, but for
larger parts exposures can only be reduced to between 1 [mu]g/m\3\ and
5 [mu]g/m\3\ using engineering and work practice controls. This group
that can achieve levels between 1 [mu]g/m\3\ and 5 [mu]g/m\3\
(approximately \1/3\ of total aerospace painting operations) can use
LEV, but as the size of the part increases it becomes increasingly
difficult to provide good air flow around the entire part, such as
underneath large horizontal structures. Moreover, as the size of the
part increases, it becomes increasingly difficult for the painter to
position him or herself to avoid being downstream of the paint
overspray due to the geometry of the parts.
When painting even larger parts, such as fuselages, wings or the
entire aircraft, exposures below 5 [mu]g/m\3\ are no longer achievable
without supplementary respiratory protection. Because these large parts
do not fit into enclosures or painting rooms, they must be painted in
oversized workspaces, typically hangers that can reach the size of a
football field (Ex. 38-106-2, p. 2). In oversized workspaces the
ventilation system becomes less effective and generally, the larger the
space, the more difficult it is to ventilate.
Moreover, when ventilation is put into such areas, the simple
solution of increasing air flow is not feasible because the amount of
air that is needed to dilute or diffuse the contaminated air can
adversely affect the quality of the job to the point where the paint or
coating is unacceptable for its purpose of protecting the part or plane
(Ex. 38-106, p. 38). Thus, simply increasing the air flow in these
sites and situations is not a viable alternative. As discussed above,
OSHA has established a provision to address the situation where
exposures cannot be brought below 25 [mu]g/m\3\ through engineering and
work practice controls alone. However, a PEL of 5 [mu]g/m\3\ can be
achieved using respiratory protection for these operations.
In short, OSHA believes a PEL of 5 [mu]g/m\3\ is feasible for
aerospace painting operations. Although one-third of those operations
will need to use respiratory protection to achieve the PEL, the
remainder can do so with engineering and work practice controls alone.
Half of that remaining group cannot achieve a PEL of 1 [mu]g/m\3\
because, even though they can take advantage of enclosures such as
paint rooms with LEV, the LEV becomes less effective as the part
becomes larger. For this reason lowering the PEL from 5 [mu]g/m\3\ to 1
[mu]g/m\3\ would result in the above-described substantial increase in
the number of employees required to wear respirators. OSHA has
therefore concluded that a PEL of 1 is not generally feasible for
aerospace painting. For a more detailed explanation of OSHA's
technological feasibility analysis for aerospace
painting operations, see Chapter III of the FEA.
Other industries. There are other major industries or applications
where OSHA is confident the PEL of 5 [mu]g/m\3\ can be met with
engineering and work practice controls, but the record does not
establish that a PEL of 1 [mu]g/m\3\ would be technologically feasible.
In particular, chromate pigment producers, chromium catalyst producers,
and chromium dye producers would have difficulty meeting the proposed
PEL. A significant portion of operations in these industries are
conducted in open and often large areas that are very dusty, making
exposures hard to control. Just as in aerospace painting above, the
primary control is to enclose the operation and then ventilate.
However, some of the operations cannot be enclosed because of the
physical configuration of the plant, especially in older facilities
(Ex. 47-3, p. 55). Moreover, because the medium containing the Cr(VI)
tends to be a fine powder, additional LEV in any worksite potentially
can result in significant and intolerable product loss. In other words,
the product could be drawn up through the ventilation system (Ex. 38-
12, pp. 12-14).
Thus, depending in large part on the number of facilities that can
accommodate enclosures, these operations could potentially require
extensive respirator use in order to meet a PEL of 1 [mu]g/m\3\; at 1
[mu]g/m\3\, OSHA expects that 44% of employees in these three
industries would need to wear respirators on at least an intermittent
basis. This number could be even higher if there are a large number of
facilities that cannot enclose troublesome operations.
To find the proposed PEL technologically feasible for an industry,
OSHA must "prove a reasonable possibility" that the typical firm can
meet it with engineering and work practice controls in most operations.
United Steelworkers, 647 F.2d at 1272. Table VIII-3 indicates that
intermittent respirator use would be required to reach the proposed PEL
of 1 [mu]g/m\3\ for chromate pigment producers, chromium catalyst
producers, and chromium dye producers. The extent of daily respirator
usage that would be required to meet the proposed PEL is not clear if
the recommended controls of enclosures and automation of the key
operations are not feasible for existing facilities, but could be
substantial depending upon the variables discussed above. On balance,
OSHA does not believe that the record establishes the likelihood that
the typical firm in these industries can meet the proposed PEL with
engineering and work practice controls. There are a total of
approximately 469 exposed employees in these three industries (Table
VIII-2). For a more detailed explanation of OSHA's technological
feasibility analysis for chromate pigment producers, chromium catalyst
producers, and chromium dye producers, see Chapter III of the FEA.
Technological feasibility is also an issue for hard chrome
electroplating operations where fume suppressants cannot be used to
control Cr(VI) exposures because they would interfere with the product
specifications, making the resulting product unusable.
In conclusion, OSHA has determined that while a PEL of 5 [mu]g/m\3\
is technologically feasible for all affected industries, the record
does not support the feasibility of the proposed PEL of 1 [mu]g/m\3\
for welding operations, aerospace painting, chromate pigment producers,
chromium catalyst producers, chromium dye producers, and some hard
chrome electroplating operations.
Economic feasibility of the final and proposed PELs. OSHA has also
evaluated the economic feasibility of the proposed and final PELs. With
regard to economic feasibility, OSHA must "provide a reasonable
assessment of the likely range of costs of its standard, and the likely
effects of those costs on the industry," so as to "demonstrate a
reasonable likelihood that these costs will not threaten the existence
or competitive structure of an industry, even if it does portend
disaster for some marginal firms." AFL-CIO v. OSHA, 965 F.2d 982 (11th
Cir. 1992). OSHA believes that the final PEL of 5 [mu]g/m\3\ is
feasible for all affected industries. (For a more detailed discussion
of OSHA's economic feasibility analysis, see Chapter VIII, Summary of
the Final Economic Analysis and Regulatory Flexibility Analysis,
Sections D and E.) In the majority of industries, costs will be less
than 1% of revenues. For fewer than 10 of the approximately 250 NAICS
(North American Industry Classification System) categories affected by
the rule, costs are estimated to exceed 1% of revenues. OSHA has
concluded that all affected industries will be able to absorb these
costs without threatening their existence or competitive structure.
Accordingly, OSHA has concluded that the new standard is economically
feasible for all industries.
By contrast, the proposed PEL of 1 [mu]g/m\3\ would not be
economically feasible for a significant industry-electroplating job
shops (NAICS 332813; electroplating, plating, polishing anodizing and
coloring services). Electroplating establishments can be broadly
classified into two categories: (1) Job shops and (2) captive shops,
with roughly half of establishments falling into each category. Job
shops perform electroplating services for others, while captive shops
provide plating services to the facility of which they are part.
A PEL of 1 [mu]g/m\3\ would result in costs exceeding 2.7% of
revenues and 65% of profits for electroplating job shops. As explained
further in section VIII of this preamble, and in the FEA, OSHA does not
believe that options for reducing impacts (e.g., phase-ins or allowing
use of respirators) would significantly alleviate the burden of the
proposed PEL. OSHA is concerned that these costs could alter the
competitive structure of the industry. Approximately 33,400 workers are
employed in electroplating job shops.
Summary of the technological and economic feasibility of the final
and proposed PELs. To summarize, OSHA concludes that the final PEL of 5
[mu]g/m\3\ is technologically and economically feasible for the
affected industries. On the other hand, the proposed PEL of 1 [mu]g/
m\3\ would be technologically or economically infeasible or is of
unproven feasibility in a large number of industries and operations
covered by the standard, including welding, aerospace painting,
chromate pigment production, chromium catalyst production, chromium dye
production, some hard chrome electroplating operations, and
electroplating job shops. These operations affect approximately 312,170
exposed employees, or almost 56% of the total number of employees
occupationally exposed to Cr(VI) (Table VIII-2). This figure includes
270,000 employees in welding, 8,300 employees in aerospace painting
operations, 33,400 employees in electroplating job shops, and 469
employees in the other three industries. (Note that this number does
not include a separate count for employees performing hard chrome
electroplating in order to avoid double counting employees performing
that operation who are employed in the electroplating job shop
category). OSHA did not receive data or recommendations regarding
setting the PEL at any levels between 1 and 5 [mu]g/m\3\.
A Uniform PEL of 5 [mu]g/m\3\ Is Consistent With the Feasibility
Constraint of Section 6(b)(5)
Section 6(b)(5) of the OSH Act requires OSHA to set the standard
which most adequately assures, to the extent feasible * * * that no
employee will suffer material impairment of health." This provision
requires the agency to eliminate or reduce significant risk, to the
extent feasible. See American Textile Mfr. Inst., Inc. v. Donovan,
452 U.S. 490, 506-22(1981). OSHA has always interpreted Section 6(b)(5)
to accord the agency substantial discretion to set the PEL at the lowest
level that is feasible for industries and operations as a whole. OSHA has
not interpreted the provision to require setting multiple PELs based on
the lowest level particular industries or operations could achieve. Because
Congress did not speak to the precise issue in the statute, OSHA has
authority to adopt the reasonable interpretation that it judges will
best carry out the purposes of the Act. Chevron U.S.A. v. Natural
Resources Defense Council, 467 U.S. 837 (1984).
The new Cr(VI) standard meets the requirements of Section 6(b)(5)
because the PEL of 5 [mu]g/m\3\ is the lowest feasible limit for many
operations and sectors employing a large number of covered employees in
fact, a majority of affected employees. In addition, the record does
not afford a basis for any further disaggregation.
OSHA recognizes that, according to the determination made in
Section VII of this preamble, significant risk remains at a PEL of 5
[mu]g/m\3\. As indicated in Table VII-3 in the Significance of Risk
section, the remaining risk for a worker exposed at the PEL throughout
a 45-year working lifetime is comparable to or greater than the
remaining risk in previous OSHA health standards where quantitative
estimates have been presented. Although OSHA anticipates that the
ancillary provisions of the standard will reduce this residual risk,
the Agency realizes that lower PELs might be achievable in some
industries and operations, which would reduce this risk even further.
As explained below, however, OSHA concludes that these benefits would
be offset by the significant disadvantages of attempting to establish
and apply multiple PELs for the diverse group of industries and
operations covered by the standard. See Building & Constr. Trades Dep't
v. U.S. Dep't of Labor, 838 F.2d 1258, 1273 (D.C. Cir. 1988)
(administrative difficulties, if appropriately spelled out, could
justify a decision to select a uniform PEL).
Requiring OSHA to set multiple PELs--taking into account the
feasibility considerations unique to each industry or operation or
group of them--would impose an enormous evidentiary burden on OSHA to
ascertain and establish the specific situations, if any, in which a
lower PEL could be reached. Such an onerous obligation would inevitably
delay, if not preclude, the adoption of important health standards. In
addition, the demanding burden of setting multiple PELs would be
complicated by the difficulties inherent in precisely defining and
clearly distinguishing between affected industries and operations where
the classification determines legal obligations. The definitional and
line-drawing problem is far less significant when OSHA merely uses a
unit of industries and operations for analytical but not compliance
purposes, and when it sets a PEL in the aggregate, i.e., when its
analysis is limited to determining whether a particular PEL is the
lowest feasible level for affected industries as a whole. If OSHA had
to set multiple PELs, and assign industries or operations to those
PELs, the problem would become much more pronounced as the consequences
of imprecise classifications would become much more significant.
The North American Industry Classification System (NAICS), which
has replaced the Standard Industrial Classification (SIC) system as the
standard Federal statistical agencies use in classifying business
establishments, is not an appropriate basis for establishing multiple
PELs. NAICS classifications are based on generally-worded definitions
and it is not always clear which definition best fits a particular
establishment. Moreover, an establishment's NAICS classification is
based on its primary activity. The establishment may include many other
activities, however, and what is the lowest feasible level for
operations in one activity may not be so for other activities. In
addition, the primary activity in an establishment may change over time
and the NAICS system itself is subject to revision every five years.
Definitional uncertainties, the presence of multiple and changing
business activities, and periodic revisions in individual codes could
have important consequences for enforcement of the standard over time.
For these reasons, OSHA has historically been reluctant to disaggregate
coverage of a standard by SIC classification. See 58 FR 166620-16621
(March 30, 1993) (discussing disaggregation of coverage of lockout/
tagout standard).
Similarly, disaggregation by operation has major practical
disadvantages. In addition to definitional complexities, a significant
problem with the use of operations for disaggregating the PEL is that
many firms have exposures in two or more different categories. Welding,
for example, is widely used in manufacturing operations in general
industry, maritime and construction. So, for instance, setting the PEL
at 5 for welding applications and 1 for other applications would mean
that some firms would have to attain two different PELs for Cr(VI)
exposures within the same workplace, and possibly even for the same
employees. As another example, chromium conversion is a process where a
treated metal surface is converted to a layer containing a complex
mixture of chromium compounds. Unlike electroplating, chromium
conversion is an entirely chemical process, and results in lower Cr(VI)
exposures than are typically associated with chromium electroplating.
Where chromium conversion is performed along with chromium
electroplating in a single establishment, it may be virtually
impossible to distinguish exposures from one source versus the other.
The same workers may even perform both tasks. Exposures from hard
chrome electroplating inevitably affect other nearby workers because
hard chrome plating is often done in the same workplaces or areas and
even at the same time as other operations involving lower Cr(VI)
exposures such as decorative plating and chrome conversion. In fact, in
many circumstances it can be virtually impossible to distinguish the
different sources that contribute to a particular employee's exposure
levels.
These are just a few examples of the many instances reflected in
the record in which individual employers will have Cr(VI) exposures
emanating from two or more different operations (Exs. 38-233, pp. 9-10;
39-52, p. 4; 47-24, p. 2; 39-20, p. 5). If multiple PELs were
established for different operations, employers would be forced to
monitor for compliance with two or more PELs within the same
workplace--a task rendered all the more difficult by the fact that the
exposure of an employee may not be tied exclusively to a single task;
different processes may be performed in close proximity to one another
and each may contribute to the exposure of an individual.
OSHA also believes that a uniform PEL will ultimately make the
standard more effective by making it easier for affected employers to
understand and comply with the standard's requirements. A uniform PEL
also makes it easier for OSHA to provide clear guidance to the
regulated community and to identify non-compliant conditions.
Finally, OSHA is concerned that adopting multiple PELS could result
in a great number of subcategories that would have to be tracked for
enforcement purposes. Apart from welding and electroplating, which
present particularly severe
dissagregation problems, there are over thirty other industry sectors
with exposure to Cr(VI). None of these sectors individually accounts
for more than 6% of the total of exposed employees; in fact, several of
those groups employ fewer than 100 employees.
For these reasons, OSHA has historically interpreted section
6(b)(5) to accord the Agency substantial discretion to set the PEL at
the lowest level feasible for industries or operations as a whole. In
adopting the arsenic standard, for example, OSHA expressly declined to
set different PELS, finding that "[s]uch an approach would be
extremely difficult to implement." 43 FR 19584, 19601 (May 5, 1978).
In that instance, OSHA explained:
The approach OSHA believes appropriate and has chosen for this
and other standards is the lowest level achievable through
engineering controls and work practices in the majority of
locations. This approach is intended to provide maximum protection
without excessively heavy respirator use. Id.
Similarly, when OSHA initially lowered the PEL for benzene from 10 ppm
to 1 ppm, it considered, but rejected, the idea of establishing
additional lower PELs, concluding that "different levels for different
industries would result in serious administrative difficulties." 43 FR
5918, 5947 (Feb. 10, 1978). And when OSHA subsequently reconsidered the
benzene standard after it was remanded for a more specific finding of
significant risk, OSHA considered, but rejected, a PEL of 0.5 ppm,
noting:
The unions have pointed out some situations where controls might
do somewhat better than 1 ppm * * * [but] OSHA believes it has
chosen the correct balance at 1 ppm as the level it can have a high
degree of confidence is generally achievable. 52 FR 34460, 34519
(Sept. 11, 1987).
In the case of cotton dust, where OSHA did set different PELs for
certain discrete groups, the groups involved exposures to different
kinds of cotton dust and different degrees of risk. Even so, OSHA
declined to adopt a unique PEL for every single affected sector. See 43
FR 27350, 37360-61 (June 23, 1978) (OSHA set one PEL for textile
industries and a separate PEL for non-textile industries, but expressly
rejected the option of adopting different exposure limits for each non-
textile industry).
In conclusion, the new PEL is the lowest level that can feasibly be
attained for many industries and operations employing a large number of
covered workers, in fact a majority of employees exposed to hexavalent
chromium. Considering all of the factors outlined above, OSHA finds
that a uniform PEL of 5 [mu]g/m\3\ is consistent with section 6(b)(5)
and that further dissagregation is not warranted.
A Short-term Exposure Limit is Unnecessary. Several commenters
recommended that OSHA establish a short-term exposure limit (STEL) for
Cr(VI) (Exs. 38-219; 38-222; 39-38; 39-50; 40-19). By restricting
potential high magnitude exposures of short duration, a STEL is
intended to protect against health effects associated with relatively
high exposures, as well as to reduce cumulative exposures. The UAW
indicated that the high residual risk of cancer justified a STEL (Ex.
40-19), while NIOSH stated that short-term exposures to high levels of
Cr(VI) can cause severe respiratory effects (40-10-2, p. 17). Other
commenters did not believe a STEL was justified, in some cases noting
that neither NIOSH nor ACGIH recommends a STEL for Cr(VI) (Exs. 38-214;
38-220; 39-19; 39-20; 39-40; 39-41; 39-47; 39-51; 39-52; 39-60; 43-26).
OSHA decided not to include a STEL in the final Cr(VI) standard for
three reasons. First, employers already are required to reduce
exposures to levels at or below the new PEL, which is expected to limit
the occurrence of high exposure excursions. Although it will not
eliminate all risk from peak exposures, the Agency anticipates that
compliance with the new PEL will substantially reduce the frequency and
magnitude of high exposure excursions, and thereby minimize the
likelihood of adverse health effects resulting from peak exposures.
Second, although in theory imposing a STEL might further lower
cumulative exposures to Cr(VI), there is little record evidence
supporting this supposition. Third, in some application groups, such as
plastic colorant producers, employees are typically exposed to Cr(VI)
not only for short durations but also intermittently. The industry has
estimated that only 5% of pigments used contain Cr(VI) (Ex. 47-24-1).
For these users, compliance with a STEL might require the expenditure
of considerable resources without providing much additional protection
to workers. These resources could more effectively be allocated to
other forms of worker protection.
Without better justification, OSHA does not consider establishment
of a STEL to be reasonably necessary or appropriate. OSHA has concluded
that a STEL would provide at most a de minimis health benefit.
(d) Exposure Determination
Paragraph (d) of the final rule sets forth requirements for
determining employee exposures to Cr(VI). The requirements are issued
pursuant to Section 6(b)(7) of the OSH Act (29 U.S.C. 655) which
mandates that any standard promulgated under section 6(b) shall, where
appropriate, "provide for monitoring or measuring of employee exposure
at such locations and intervals, and in such manner as may be necessary
for the protection of employees."
The purpose of requiring an assessment of employee exposures to
Cr(VI) includes: determination of the extent and degree of exposure at
the worksite; identification and prevention of employee overexposure;
identification of the sources of exposure to Cr(VI); collection of
exposure data so that the employer can select the proper control
methods to be used; and evaluation of the effectiveness of those
selected methods. Assessment enables employers to meet their legal
obligation to ensure that their employees are not exposed to Cr(VI) in
excess of the permissible exposure level and to notify employees of
their exposure levels, as required by section 8(c)(3) of the Act. In
addition, the availability of exposure data enables the PLHCP
performing medical examinations to be informed of the extent of
occupational exposures.
The final requirements have been revised from those proposed in
response to comments received. In the proposed general industry
standard, OSHA included a requirement for initial exposure monitoring
in all workplaces covered by the rule, unless monitoring had been
performed in the previous 12 months, or the employer had data to
demonstrate that exposures would be below the action level. Periodic
monitoring was required at intervals determined by monitoring results
(i.e., at least every 6 months if exposures were at or above the action
level, at least every 3 months if exposures were above the PEL), and
additional monitoring was required when changes in the workplace
resulted in new or additional exposures to Cr(VI). These requirements
are similar to requirements for monitoring found in previous OSHA
substance-specific health standards, such as those for methylene
chloride (29 CFR 1910.1052) and 1,3-butadiene (29 CFR 1910.1051).
The proposed standards for construction and shipyards did not
include provisions for exposure monitoring. OSHA did not propose
specific exposure monitoring requirements for construction and
shipyards because operations in these sectors are often of short
duration, and are performed under varying environmental conditions.
In omitting exposure monitoring requirements from the proposed
standards for construction and shipyards, OSHA intended to provide
construction and shipyard employers with the flexibility to assess
Cr(VI) exposures in any manner they considered appropriate. It was not
the Agency's intent that employers ignore substantial exposures to
Cr(VI). Because the obligation to comply with the PEL would remain, the
employer would have to accurately characterize Cr(VI) exposures in
order to determine if they were in compliance. At the time of the
proposal, OSHA considered this performance-oriented approach a
reasonable way to determine employee exposures to Cr(VI) while avoiding
the more infeasible requirements of a scheduled monitoring approach
that might not be useful in construction and shipyard workplaces. This
performance-based approach was consistent with OSHA's standard for air
contaminants (29 CFR 1910.1000), which establishes PELs for over 400
substances but does not include specific requirements for exposure
monitoring.
Construction and shipyard employers who expressed an opinion on the
issue generally supported the absence of specific exposure monitoring
requirements (e.g., Exs. 38-220; 38-235; 38-244). In addition to those
operations that involved changing conditions, employers argued that
periodic monitoring requirements were unnecessary when conditions did
not change (Exs. 38-124; 38-213, 38-215; 38-189, 38-191). For example,
the U.S. Navy stated:
The prescriptive schedule of required air sampling has not
proved beneficial in assessing risks in shipyards * * * where there
has been virtually no change in conditions, yet costs for consistent
air sampling have been incurred on an annual basis without
informational benefit or added protection for workers. The
performance-based sampling approach * * * is protective, efficient,
and logical (Ex. 38-220).
A number of employers also supported a performance oriented approach
for exposure determination in general industry workplaces (Exs. 38-189;
38-191; 38-213; 38-215; 39-48). Some of these commenters argued that
Cr(VI) exposures in their workplaces were intermittent, variable, and
of short duration, and therefore similar to those found in construction
and shipyards (Exs. 38-203; 38-254; 39-19; 39-48; 39-56). Other
comments focused on requirements for periodic monitoring that were
considered to be excessive (e.g., Exs. 38-124; 38-189; 38-191; 38-213;
38-215; 38-233). For example, the Color Pigments Manufacturers
Association stated:
OSHA continues to require repeated monitoring at great cost in
general industry under circumstances where no change in procedure,
process, equipment or exposure has occurred to warrant repeated
exposure monitoring. This requirement is unnecessary and punitive.
It forces general industry to expend valuable resources on continual
monitoring without reason (Ex. 38-205).
Some employers, while maintaining that periodic monitoring requirements
were not warranted, indicated that initial exposure monitoring or an
initial hazard assessment would be appropriate (Exs. 38-214; 38-245-1).
Other commenters, including unions, Public Citizen, and NIOSH,
supported explicit requirements for exposure assessment (Exs. 38-199-1;
38-222; 40-10-2; 47-23, p. 16). These parties argued that employers
will not know whether or not they are in compliance with the standard
without mandated exposure monitoring. For example, the Building and
Construction Trades Department, AFL-CIO, stated:
If OSHA indeed intends construction employers to conduct an
exposure assessment, this requirement must be explicitly stated in
the regulation. To suggest that employers will attempt to
characterize exposure routinely without an explicit requirement in
the regulation is ludicrous (Ex. 38-219).
Even where controls are implemented, it was argued, exposure assessment
is still necessary to ensure that those controls are adequately
protective (Ex. 38-219). NIOSH suggested that OSHA might want to
consider developing alternative means for assessing exposures, such as
the use of interim protection provisions in construction for certain
tasks until exposure monitoring could be done (see the lead standard,
29 CFR 1926.62(d)) and the use of grouped tasks and grouping job types
into classes based on exposure potential (see the asbestos standard, 29
CFR 1926.1101) (Ex. 40-10-2, p. 19).
After considering the evidence and arguments advanced by rulemaking
participants, OSHA is convinced that requirements for scheduled initial
and periodic Cr(VI) exposure monitoring are not appropriate in all
circumstances. In particular, OSHA believes that the evidence in this
rulemaking, as discussed earlier in this section in paragraph (c),
permissible exposure limit, demonstrates the varied nature of Cr(VI)
exposures across a number of different work operations. However, OSHA
also believes that valid concerns have been raised regarding the
adequacy of exposure assessments that would be performed in the absence
of explicit requirements. The Agency is therefore including in the
final rule two alternative options for all affected employers to follow
for determining employee exposures to Cr(VI). The first option,
referred to as the "scheduled monitoring option", consists of
requirements for initial monitoring and periodic monitoring at
intervals based on monitoring results. This approach is similar to that
proposed for general industry in this rulemaking and with exposure
assessment requirements in previous OSHA substance-specific standards.
The second option, referred to as the "performance-oriented option",
allows employers to use any combination of air monitoring data (i.e.,
data obtained from initial and periodic monitoring performed in
accordance with the requirements of the Cr(VI) standard), historical
monitoring data, or objective data to determine employee exposures to
Cr(VI), as long as the data are sufficient to accurately characterize
exposures.
OSHA believes that by including explicit requirements for exposure
determination in the standards for general industry, construction, and
shipyards, the Agency makes clear the obligation of employers to
accurately assess employee exposures to Cr(VI) in all sectors. By
offering two options for achieving this goal, the final rule provides a
framework that is familiar to many employers and has been successfully
applied in the past, as well as flexibility for employers who are able
to characterize employee exposures through alternative methods.
OSHA has chosen not to use the task-based approaches suggested by
NIOSH (Ex. 40-10-2) that the Agency has used in several previous health
standards covering construction. While OSHA believes that these
approaches are effective in certain construction settings, there was
not sufficient information in this rulemaking record for OSHA to
develop classes of exposures that would apply across the many varied
work operations with Cr(VI) exposures. While it was not possible to
develop specific classes of operations to apply across all industries,
OSHA believes that an individual employer, with specific information
about the work processes at his worksite, may be able to use such an
approach in using the performance-based option allowed by this final
rule.
Paragraph (d)(2) contains requirements for employers who choose the
scheduled monitoring option. Employers who select this option must
conduct initial monitoring to determine employee exposure to Cr(VI).
OSHA has not established a separate compliance date for initial
monitoring to allow employers flexibility in scheduling this activity.
However, employers must allow sufficient time after initial monitoring
is performed to achieve compliance (e.g., establish regulated areas,
provide appropriate respiratory protection) by the start-up dates specified
in paragraph (n) (paragraph (l) for construction and shipyards). Monitoring to
determine employee exposures must represent the employee's time-
weighted average exposure to airborne Cr(VI) over an eight-hour
workday. Samples must be taken within the employee's breathing zone
(i.e., "personal breathing zone samples" or "personal samples"),
and must represent the employee's exposure without regard to the use of
respiratory protection.
Employers must accurately characterize the exposure of each
employee to Cr(VI). In some cases, this will entail monitoring all
exposed employees. In other cases, monitoring of "representative"
employees is sufficient. Representative exposure sampling is permitted
when a number of employees perform essentially the same job under the
same conditions. For such situations, it may be sufficient to monitor a
fraction of these employees in order to obtain data that are
"representative" of the remaining employees. Representative personal
sampling for employees engaged in similar work with Cr(VI) exposure of
similar duration and magnitude is achieved by monitoring the
employee(s) reasonably expected to have the highest Cr(VI) exposures.
For example, this may involve monitoring the Cr(VI) exposure of the
employee closest to an exposure source. This exposure result may then
be attributed to the remaining employees in the group.
Exposure monitoring should include, at a minimum, one full-shift
sample taken for each job function in each job classification, in each
work area, for each shift. These samples must consist of at least one
sample characteristic of the entire shift or consecutive representative
samples taken over the length of the shift. Where employees are not
performing the same job under the same conditions, representative
sampling will not adequately characterize actual exposures, and
individual monitoring is necessary.
Employers who have workplaces covered by the standard must
determine if any of their employees are exposed to Cr(VI) at or above
the action level. Further obligations under the standard are based on
the results of this assessment. These may include obligations for
periodic monitoring, establishment of regulated areas, implementation
of control measures, and provision of medical surveillance.
Requirements for periodic monitoring depend on the results of
initial monitoring. If the initial monitoring indicates that employee
exposures are below the action level, no further monitoring is required
unless changes in the workplace result in new or additional exposures.
If the initial determination reveals employee exposures to be at or
above the action level but at or below the PEL, the employer must
perform periodic monitoring at least every six months. If the initial
monitoring reveals employee exposures to be above the PEL, the employer
must repeat monitoring at least every three months.
The scheduled monitoring option also includes provisions to adjust
the frequency of periodic monitoring based on monitoring results. If
periodic monitoring results indicate that employee exposures have
fallen below the action level, and those results are confirmed by
consecutive measurements taken at least seven days apart, the employer
may discontinue monitoring for those employees whose exposures are
represented by such monitoring. Similarly, if periodic monitoring
measurements indicate that exposures are at or below the PEL but at or
above the action level, the employer may reduce the frequency of the
monitoring to at least every six months.
OSHA recognizes that exposures in the workplace may fluctuate.
Periodic monitoring provides the employer with assurance that employees
are not experiencing higher exposures that may require the use of
additional control measures. In addition, periodic monitoring reminds
employees and employers of the continued need to protect against the
hazards associated with exposure to Cr(VI).
Because of the fluctuation in exposures, OSHA believes that when
initial monitoring results equal or exceed the action level but are at
or below the PEL, employers should continue to monitor employees to
ensure that exposures remain at or below the PEL. Likewise, when
initial monitoring results exceed the PEL, periodic monitoring allows
the employer to maintain an accurate profile of employee exposures. If
the employer installs or upgrades controls, periodic monitoring will
demonstrate whether or not controls are working properly. Selection of
appropriate respiratory protection also depends on adequate knowledge
of employee exposures.
In general, the more frequently periodic monitoring is performed,
the more accurate the employee exposure profile. Selecting an
appropriate interval between measurements is a matter of judgment. OSHA
believes that the frequency of six months for subsequent periodic
monitoring for exposures at or above the action level but at or below
the PEL, and three months for exposures above the PEL, provides
intervals that are both practical for employers and protective for
employees. This belief is supported by OSHA's experience with
comparable monitoring intervals in other standards, including those for
cadmium (29 CFR 1910.1027), methylenedianiline (29 CFR 1910.1050),
methylene chloride (29 CFR 1910.1052), and formaldehyde (29 CFR
1910.1048).
OSHA recognizes that monitoring can be a time-consuming, expensive
endeavor and therefore offers employers the incentive of discontinuing
monitoring for employees whose sampling results indicate exposures are
below the action level. The Agency does not believe that periodic
monitoring is generally necessary when monitoring results show that
exposures are below the action level because there is a low probability
that the results of future samples would exceed the PEL. Therefore the
final rule provides an incentive for employers to control their
employees' exposures to Cr(VI) below the action level to minimize their
exposure monitoring obligations while maximizing the protection of
employees' health.
Under the scheduled monitoring option, employers are to perform
additional monitoring when there is a change in production process, raw
materials, equipment, personnel, work practices, or control methods,
that may result in new or additional exposures to Cr(VI). For example,
if an employer has conducted monitoring for an electroplating operation
while using fume suppressants, and the use of fume suppressants is
discontinued, then additional monitoring would be necessary to
determine employee exposures under the modified conditions. In
addition, there may be other situations which can result in new or
additional exposures to Cr(VI) which are unique to an employer's work
situation. For instance, a welder may move from an open, outdoor
location to an enclosed or confined space. Even though the task
performed and materials used may remain constant, the changed
environment could reasonably be expected to result in higher exposures
to Cr(VI). In order to cover those special situations, OSHA requires
the employer to perform additional monitoring whenever the employer has
any reason to believe that a change has occurred which may result in
new or additional exposures. This additional monitoring is necessary to
ensure that monitoring results accurately represent existing exposure
conditions. This information will enable the employer to take appropriate
action to protect exposed employees, such as instituting additional engineering
controls or providing appropriate respiratory protection. On the other
hand, additional monitoring is not required simply because a change has
been made, if the change is not reasonably expected to result in new or
additional exposures to Cr(VI). For example, monitoring may be
conducted in an establishment when welding was performed on steel with
15% Cr content. If the establishment switches to a steel with 10% Cr
content without changing any other aspect of the work operation, then
additional exposures to Cr(VI) would not reasonably be expected, and
additional monitoring would not be required.
The performance-oriented option allows the employer to determine
the 8-hour TWA exposure for each employee on the basis of any
combination of air monitoring data, historical monitoring data, or
objective data sufficient to accurately characterize employee exposure
to Cr(VI). This option is intended to allow employers flexibility in
assessing the Cr(VI) exposures of their employees. Where the employer
elects to follow this option, the exposure determination must be
performed prior to the time the work operation commences, and must
provide the same degree of assurance that employee exposures have been
correctly characterized as air monitoring would. The employer is
expected to reevaluate employee exposures when there is any change in
the production process, raw materials, equipment, personnel, work
practices, or control methods that may result in new or additional
exposures to Cr(VI).
When using the term "air monitoring data" in this paragraph, OSHA
refers to initial and periodic Cr(VI) monitoring conducted to comply
with the requirements of this standard, including the prescribed
accuracy and confidence requirements. Historical monitoring data refers
to Cr(VI) monitoring data that was obtained prior to the effective date
of the final rule, where the data were obtained during work operations
conducted under workplace conditions closely resembling the processes,
types of material, control methods, work practices, and environmental
conditions in the employer's current operations, and where that
monitoring satisfies all other requirements of this section, including
the accuracy and confidence requirements described below.
Objective data means information such as air monitoring data from
industry-wide surveys or calculations based on the composition or
chemical and physical properties of a substance demonstrating employee
exposure to Cr(VI) associated with a particular product or material or
a specific process, operation, or activity. The data must reflect
workplace conditions closely resembling the processes, types of
material, control methods, work practices, and environmental conditions
in the employer's current operations. Objective data demonstrate the
Cr(VI) exposures associated with a work operation or product under the
range of expected conditions of use. For example, data collected by a
trade association from its members may be used to determine exposures
to Cr(VI) provided the data meet the definition of objective data in
the standard.
Previous OSHA substance-specific health standards have usually
allowed employers to use objective data to characterize employee
exposures, but have generally limited its use to demonstrating that
exposures would be below the action level (e.g., the Cadmium standard,
29 CFR 1910.1027(d)(2)(iii)). Likewise, use of historical monitoring
data has typically been allowed, but has usually been limited to data
obtained within the previous 12 months (e.g., the Methylene Chloride
standard, 29 CFR 1910.1052(d)(2)(ii)). In this instance, OSHA does not
place these limitations on the use of historical monitoring data or
objective data. However, the burden is on the employer to show that the
data comply with the requirements of this section. For example,
historical monitoring data obtained 18 months prior to the effective
date of the standard could be used to determine employee exposures, but
only if the employer could show that the data were obtained during work
operations conducted under workplace conditions closely resembling the
processes, types of material, control methods, work practices, and
environmental conditions in the employer's current operations, and that
the monitoring satisfies all other requirements of this section,
including the accuracy and confidence requirements. OSHA's intent is to
allow employers the greatest possible flexibility in methods used to
determine employee exposures to Cr(VI), but to ensure that the methods
used are accurate in characterizing employee exposures.
Under paragraph (d)(4) of the final rule, employers covered by the
general industry standard must notify each affected employee within 15
working days if the exposure determination indicates that employee
exposure exceeds the PEL. In construction and shipyards, employers must
notify each affected employee as soon as possible but not more than 5
working days after the exposure determination indicates that employee
exposure exceeds the PEL. A shorter time period for notification is
provided in construction and shipyards in recognition of the often
short duration of operations and employment in particular locations in
these sectors. The time allowed for notification is consistent with the
harmonized notification times established for these sectors in Phase II
of OSHA's Standards Improvement Project (70 FR 1112 (1/5/05)). Where
the employer follows the scheduled monitoring option, the 15 (or 5)
working day period commences when monitoring results are received. For
employers following the performance-oriented option, the 15 (or 5)
working day period commences when the determination is made (i.e.,
prior to the time the work operation commences, and when exposures are
reevaluated).
When using the term "affected employees" in this provision, OSHA
is referring to all employees considered to be above the PEL. This
would include employees who are not actually subject to personal
monitoring, but are represented by an employee who is sampled. Affected
employees also include employees whose exposures have been deemed to be
above the PEL on the basis of historical or objective data. The
employer shall either notify each affected employee in writing or post
the monitoring results in an appropriate location accessible to all
affected employees. In addition, whenever the PEL has been exceeded,
the written notification must contain a description of the corrective
action(s) being taken by the employer to reduce the employee's exposure
to or below the PEL. The requirement to inform employees of the
corrective actions the employer is taking to reduce the exposure level
to or below the PEL is necessary to assure employees that the employer
is making efforts to furnish them with a safe and healthful work
environment, and is required under section 8(c)(3) of the Act.
Paragraph (d)(5) of the final rule requires the employer to use
monitoring and analytical methods that can measure airborne levels of
Cr(VI) to within an accuracy of plus or minus 25% (25%) and
can produce accurate measurements to within a statistical confidence
level of 95% for airborne concentrations at or above the action level.
Many laboratories presently have methods to measure Cr(VI) at the action
level with at least the required degree of accuracy. One example of an
acceptable method of monitoring and analysis is OSHA method ID215, which
is a fully validated analytical method used by the Agency. (See Chapter III
of the FEA for a discussion of issues regarding methods of sampling and
analysis). Rather than specifying a particular method that must be
used, OSHA allows the employer to use any method as long as the chosen
method meets the accuracy specifications. This is consistent with the
general performance approach favored in the OSH Act.
Paragraph (d)(6) requires the employer to provide affected
employees or their designated representatives an opportunity to observe
any monitoring of employee exposure to Cr(VI), whether the employer
uses the scheduled monitoring option or the performance-oriented
option. When observation of monitoring requires entry into an area
where the use of protective clothing or equipment is required, the
employer must provide the observer with that protective clothing or
equipment, and assure that the observer uses such clothing or equipment
and complies with all other required safety and health procedures.
The requirement for employers to provide employees or their
representatives the opportunity to observe monitoring is consistent
with the OSH Act. Section 8(c)(3) of the OSH Act mandates that
regulations developed under Section 6 provide employees or their
representatives with the opportunity to observe monitoring or
measurements. Also, Section 6(b)(7) of the OSH Act states that where
appropriate, OSHA standards are to prescribe suitable protective
equipment to be used in dealing with hazards. The provision for
observation of monitoring and protection of the observers is also
consistent with OSHA's other substance-specific health standards such
as those for cadmium (29 CFR 1910.1027) and methylene chloride (29 CFR
1910.1052).
(e) Regulated Areas
Paragraph (e) of the final rule requires general industry employers
to establish regulated areas wherever an employee's exposure to
airborne concentrations of Cr(VI) is, or can reasonably be expected to
be, in excess of the PEL. Regulated areas are to be demarcated from the
rest of the workplace in a manner that adequately establishes and
alerts employees to the boundaries of these areas. Access to regulated
areas is to be limited to persons authorized by the employer and
required by work duties to be present in the regulated area; any person
entering the regulated area to observe monitoring procedures; or any
person authorized by the OSH Act or regulations issued under it to be
in a regulated area.
The purpose of a regulated area is to ensure that the employer
makes employees aware of the presence of Cr(VI) at levels above the
PEL, and to limit Cr(VI) exposure to as few employees as possible. The
establishment of a regulated area is an effective means of limiting the
risk of exposure to substances known to have carcinogenic effects.
Because of the potentially serious results of exposure and the need for
persons exposed above the PEL to be properly protected, the number of
persons given access to the area must be limited to those employees
needed to perform the job. Limiting access to regulated areas also has
the benefit of reducing the employer's obligation to implement
provisions of this standard to as few employees as possible.
In keeping with the performance orientation of this standard, OSHA
has not specified how employers are to demarcate regulated areas. OSHA
proposed that warning signs be posted at all approaches to regulated
areas, and set forth specific language in paragraph (1) of the proposed
standard to be included on the warning signs. However, OSHA has
determined that other means of demarcation such as barricades, lines
and textured flooring, or signs using other language can be equally
effective in identifying the boundaries of regulated areas and
notifying employees of associated hazards, the need to restrict access
to such areas, and protective measures to be implemented. The specific
language for warning signs included in paragraph (1) of the proposal,
and the reference to that language in this provision, have therefore
been deleted from the final rule.
In the final rule, OSHA thus has provided employers with the
flexibility to use the methods of demarcation that are most appropriate
for identifying regulated areas in their workplace. Factors that the
Agency believes are appropriate for employers to consider in
determining how to mark their areas include the configuration of the
area, whether the regulated area is permanent, the airborne Cr(VI)
concentration, the number of employees in adjacent areas, and the
period of time the area is expected to have exposure levels above the
PEL. Permitting employers to choose how best to identify and limit
access to regulated areas is consistent with OSHA's belief that
employers are in the best position to make such determinations, based
on their knowledge of the specific conditions of their workplaces.
Whatever methods are chosen, the demarcation must effectively warn
employees not to enter the area unless they are authorized, and then
only if they are using the proper personal protective equipment.
Allowing employers to demarcate and limit access to the regulated areas
as they choose is consistent with OSHA's two most recent substance-
specific health standards, addressing occupational exposure to
methylene chloride (29 CFR 1910.1052(e)) and 1,3-butadiene (29 CFR
1910.1051(e)).
Access to the regulated area is restricted to "authorized
persons." For the purposes of this standard, these are persons
required by their job duties to be present in the area, as authorized
by the employer. This may include maintenance and repair personnel,
management, quality control engineers, or other personnel if job duties
require their presence in the regulated area. In addition, persons
exercising the right to observe monitoring procedures are allowed to
enter regulated areas when exposure monitoring is being conducted.
Persons authorized under the OSH Act, such as OSHA compliance officers,
are also allowed access to regulated areas.
In the final rule, OSHA has not included a requirement for
regulated areas in construction and shipyard workplaces, due to the
expected practical difficulties of establishing regulated areas for
operations in these sectors. OSHA raised the issue of requiring
regulated areas for these workplaces and received comments and
testimony from a variety of sources. A number of commenters supported
not requiring regulated areas in construction and shipyards (Exs. 38-
214; 38-220; 38-235; 38-236; 38-244; 39-37; 39-20; 39-40; 39-48; 39-64;
39-65). The National Association of Home Builders, for example,
indicated that regulated areas are not feasible on residential
construction jobsites because the area where exposures would exceed the
PEL could not be accurately determined, stating:
Because of the fluid nature of construction work and the ever-
changing work environment, a regulated area could never be
accurately determined due to the fact that construction areas are
mostly exposed to the ambient environment. Factors such as shifting
winds, tight work areas and multiple operations adjacent to the
regulated area would create changes in air movement and would make
establishment of a regulated area unattainable (Ex. 38-244).
Associated Builders and Contractors concurred with this assessment, and
maintained that establishment of regulated areas could interfere with
construction operations:
The nature of construction sites makes it extremely difficult to
close off certain areas from others without shutting down or
interfering with significant construction activities (Ex. 39-65).
Some commenters maintained that certain activities should not be
subject to requirements for regulated areas (Exs. 38-7, p. 5; 38-124;
38-203; 38-205; 38-228; 38-233; 38-238; 38-254; 39-19; 39-56; 39-62).
The Office of Advocacy of the Small Business Administration, for
example, stated that requirements for regulated areas should be limited
to industries and processes where they would likely reduce exposures,
arguing that establishment of regulated areas would have the effect of
requiring respirators or other controls for more employees than
necessary (Ex. 38-7). Because regulated areas are required only where
exposures exceed the PEL, OSHA considers that these requirements are
limited to situations where they can reduce exposures. As mentioned
previously, making employees aware of potential exposures in excess of
the PEL and limiting the number of employees present in regulated areas
will effectively reduce exposures to Cr(VI). Moreover, establishment of
regulated areas will not result in additional requirements for
respirators or other controls, because requirements for these other
control measures are not directly related to the establishment of
regulated areas. Simply entering a regulated area, for example, does
not trigger a requirement for use of respiratory protection.
Other commenters maintained that certain general industry
activities, or general industry as a whole, should not be subject to
the proposed requirements for regulated areas. Alabama Power, for
example, indicated that the same rationale used to justify the absence
of regulated area requirements in construction and shipyards also
applied to general industry environments such as power plants (Exs. 38-
254; 38-203). Others argued that regulated areas were not appropriate
for specific activities such as welding (Ex. 38-124), job shop
fabrication (Exs. 38-238; 39-62), or glass manufacturing (Ex. 38-228).
Other commenters expressed support for regulated area requirements,
arguing that they were a feasible and useful means of protecting
workers, and should apply to construction and shipyards as well as
general industry workplaces (Exs. 38-199-1; 38-219; 38-222; 39-38; 39-
71; 40-10-2; 47-28). For example, NIOSH indicated that regulated areas
help minimize exposures to bystanders in construction and shipyard
worksites:
* * * regulated areas are important on construction and shipyard
worksites because of the potential for "bystander" exposures given
that it is common for employees from different trades to work in
close proximity. For construction, bystander employees may work for
different employers, thus complicating control efforts (Ex. 40-10-
2).
Regulated areas, it was argued, are not unduly burdensome. Dr. Franklin
Mirer of the United Auto Workers, when asked if he foresaw problems
with requirements for regulated areas, stated:
* * * you put a sign [up] and you tell people who don't have to
be there not to be there * * * what's burdensome about that? It's
like * * * putting up a sign on the ladies room. Certain people
can't go in that regulated area (Tr. 837).
OSHA believes, however, that Dr. Mirer oversimplifies the
situation. The difficulty is not with the mere physical act of putting
up a sign at a regulated area, but rather with determining where, when,
and for how long a duration to establish a regulated area. Making these
determinations is very problematic given the varied and changing nature
of the operations involving Cr(VI) exposures at construction and
shipyard worksites. Moreover, areas where employees are exposed above
the PEL might change on a daily or even hourly basis and may occur at
different sites on the worksite than they did the day before, making it
unreasonably difficult to keep up with the posting (and removal) of
signs, barricades or other warning in a manner that would effectively
let employees know about the hazard.
OSHA has concluded that requirements for regulated areas are
appropriate for general industry, but not for construction and
shipyards, because the work sites and conditions and other factors,
such as environmental variability normally present in construction and
shipyard employment, differ substantially from those typically found in
general industry. Construction and shipyard tasks are often of
relatively short duration; are commonly performed outdoors, sometimes
under adverse environmental conditions (e.g., wind, rain); and are
often performed at non-fixed workstations or work sites. Collectively,
these factors make establishment of regulated areas impracticable for
many construction and shipyard operations.
These difficulties are particularly evident with regard to welding
operations in construction and shipyard workplaces. Welding is the
predominant source of Cr(VI) exposures in these sectors, accounting for
over 82% of employees exposed above the PEL in construction and over
73% of employees exposed above the PEL in shipyards. Welding operations
in construction and shipyards often involve movement to different
locations during the workday, and welding fumes are highly subject to
changes in air currents, meaning the exposure patterns can shift
rapidly.
In the typical shipyard and construction project involving
exposure, it is difficult to determine appropriate boundaries for
regulated areas because the work and worksite are varied and subject to
environmental influences. Moreover, workers are often moving from place
to place throughout the site on a regular basis. While each employer
has the obligation under the requirements of paragraph (d) of this
final rule to determine Cr(VI) exposures for all employees, accurately
demarcating all areas where Cr(VI) exposures could potentially exceed
the PEL is a separate and potentially much more difficult undertaking.
In general industry environments, which are typically more stable,
likely to be indoors, and usually at a fixed location, this can
generally be accomplished with minimal difficulty. In construction and
shipyard workplaces, for the reasons described above, OSHA has
determined that establishing regulated areas to control exposures to
Cr(VI) can not reasonably be accomplished, and has therefore not
included a requirement for regulated areas for these sectors in the
final rule.
The Agency realizes that in some cases general industry work
operations and work environments may be comparable to those found in
construction and shipyards, and where the general industry employer can
show compliance is not feasible, regulated areas will not have to be
established. However, OSHA believes its longstanding distinction
between these sectors provides an appropriate line for delineating
between those operations where the employer generally is reasonably
able to establish regulated areas where exposures to Cr(VI) exceed the
PEL versus operations where regulated areas are generally not
practicable.
OSHA recognizes that the determination not to include requirements
for regulated areas for construction and shipyards in this final rule
differs from the determinations made in previous rulemakings. The AFL-
CIO pointed out that a number of previous standards including those for
asbestos, cadmium, benzene, 1,2-dibromo-3-chloropropane, ethylene oxide,
methylenedianiline, formaldehyde, and 1,3 butadiene, included provisions
for regulated areas in construction (Exs. 38-222; 47-28-1). It is important
to note, however, that many of these standards such as benzene, 1,2-dibromo-3-
chloropropane, ethylene oxide, methylenedianiline, and formaldehyde
involved relatively few exposures in construction operations. For
example, in the preamble to the final benzene standard OSHA concluded
that while the standard would cover construction, "The standard has
virtually no impact on construction" (52 FR at 34527). Similarly,
requirements for regulated areas in the standard for cadmium in
construction did not pose major problems for employers, because few
workers were expected to be exposed above the PEL and thus subject to
requirements for regulated areas. More importantly, in the cadmium
rulemaking as in others discussed below, regulated areas for
construction were not at issue because so few employees were
potentially exposed above the PEL. Thus, the Agency did not address the
factors that were presented in this rulemaking.
OSHA's standards for lead in construction and asbestos in
construction, on the other hand, affect relatively large numbers of
employers and employees. The standard for lead in construction is a
notable exception to the AFL-CIO's list. OSHA did not include
requirements for regulated areas in that standard (see 29 CFR 1926.62).
While the asbestos construction standard does include requirements for
regulated areas, the classification scheme for asbestos construction
operations (i.e., Class I, II, III and IV) and requirements for
enclosing many work operations makes establishment of regulated areas
easier for employers. (see 29 CFR 1926.1101). The Agency believes that
the broad scope of the Cr(VI) final rule for construction, similar to
the standard covering lead construction operations, would make
application of regulated area requirements substantially more difficult
than is the case for a standard with a much more limited scope, such as
the standards for cadmium or benzene in construction.
Finally, in none of the previous health standards were the
particular difficulties of implementing regulated areas for shipyard
and construction work specifically considered as they have been in this
rulemaking. In this rulemaking, the establishment of regulated areas
was a major issue with a significant volume of comments and testimony,
allowing OSHA to fully consider the matter in light of the specific
nature of Cr(VI) exposures. First, OSHA's proposal did not include
regulated areas in construction and shipyard employment. Secondly, in
the proposal, OSHA included two general questions, numbers 31 and 32,
on modifying the requirements for construction and shipyard employment
and one very specific question, number 47, on whether regulated areas
should be included for construction and shipyard employment (69 FR
59452, 59310). Thus, the public had sufficient notice and OSHA was able
to weigh the evidence, ultimately finding the reasons for excluding
regulated areas from construction and shipyard employment persuasive.
(f) Methods of Compliance
Paragraph (f) of the final rule (paragraph (e) for construction and
shipyards) establishes which methods must be used by employers to
comply with the PEL. It requires that employers institute effective
engineering and work practice controls as the primary means to reduce
and maintain employee exposures to Cr(VI) to levels that are at or
below the PEL unless the employer can demonstrate that such controls
are not feasible. Where the employer demonstrates that such controls
are not feasible, the final rule requires the employer to institute
engineering and work practice controls to reduce exposures to the
lowest feasible level. The employer is then required to supplement
these controls with respiratory protection to achieve the PEL.
A number of commenters supported OSHA's inclusion of the hierarchy
of controls in the final Cr(VI) rule (e.g., Tr. 826, Exs. 38-232; 38-
235; 38-238; 39-20; 39-47; 40-10-2; 47-23; 47-26). For example, NIOSH
endorsed the use of engineering and work practice controls as primary
methods of controlling exposures to Cr(VI) (Ex. 40-10-2). Personal
protective equipment such as respirators was regarded by NIOSH as the
last line of defense, to be used only when engineering controls are not
feasible. Other commenters objected to OSHA's proposed application of
the hierarchy of controls in the Cr(VI) rule, arguing that use of
respiratory protection instead of engineering controls should be
allowed in a variety of different situations (e.g., Exs. 38-204; 38-
215; 38-216-1; 38-218; 38-233; 39-51; 39-66; 43-14; 47-30; 47-31; 47-
32). For example, the National Paint and Coatings Association contended
that respirator use should be permitted in paint and coatings
manufacture:
* * * exposures to hexavalent chromium compounds are limited in
time and place, and their handling is seldom encountered by
other[sic] than a relatively small number of workers, whose use of
respirators would not pose most of the problems OSHA associates with
respirators * * * (Ex. 39-66).
OSHA is requiring primary reliance on engineering controls and work
practices because reliance on these methods is consistent with good
industrial hygiene practice, with the Agency's experience in assuring
that workers have a healthy workplace, and with the Agency's
traditional adherence to a hierarchy of preferred controls. Engineering
controls are reliable, provide consistent levels of protection to a
large number of workers, can be monitored, allow for predictable
performance levels, and can efficiently remove a toxic substance from
the workplace. Once removed, the toxic substance no longer poses a
threat to employees. The effectiveness of engineering controls does not
generally depend to any substantial degree on human behavior, and the
operation of equipment is not as vulnerable to human error as is
personal protective equipment.
Engineering controls can be grouped into three main categories: (1)
Substitution; (2) isolation; and (3) ventilation, both general and
localized. Quite often a combination of these controls can be applied
to an industrial hygiene control problem to achieve satisfactory air
quality. It may not be necessary to apply all these measures to any
specific potential hazard.
Substitution can be an ideal control measure. One of the best ways
to prevent workers from being exposed to a toxic substance is to stop
using it entirely. Although substitution is not always possible,
replacement of a toxic material with a less hazardous alternative
should always be considered.
In those cases where substitution of a less toxic material is not
possible, substituting one type of process for another process may
provide effective control of an air contaminant. For example, process
changes from batch operations to continuous operations will usually
reduce exposures. This is true primarily because the frequency and
duration of workers' potential contact with process materials is
reduced in continuous operations. Similarly, automation of a process
can further reduce the potential hazard.
In addition to substitution, isolation should be considered as an
option for controlling employee exposures to
Cr(VI). Isolation can involve containment of the source of a hazard,
thereby separating it from most workers. Workers can be isolated from
Cr(VI) by working in a clean room or booth, or by placing some other
type of barrier between the source of exposure and the employee.
Employees can also be protected by being placed at a greater distance
from the source of Cr(VI) emissions.
Frequently, isolation enhances the benefits of other control
methods. For example, Cr(VI) compounds may be used in the formulation
of certain paints. If the mixing operation is conducted in a small,
enclosed room the airborne Cr(VI) potentially generated by the
operation could be confined to a small area. By ensuring containment,
local exhaust ventilation is more effective.
Ventilation is a method of controlling airborne concentrations of a
contaminant by supplying or exhausting air. A local exhaust system is
used to remove an air contaminant by capturing the contaminant at or
near its source before it spreads throughout the workplace. General
ventilation (dilution ventilation), on the other hand, allows the
contaminant to spread throughout the work area but dilutes it by
circulating large quantities of air into and out of the area. A local
exhaust system is generally preferred to dilution ventilation because
it provides a cleaner and healthier work environment.
Work practice controls involve adjustments in the way a task is
performed. In many cases, work practice controls complement engineering
controls in providing worker protection. For example, periodic
inspection and maintenance of process equipment and control equipment
such as ventilation systems is an important work practice control.
Frequently, equipment which is in disrepair or near failure will not
perform normally. Regular inspections can detect abnormal conditions so
that timely maintenance can then be performed. If equipment is
routinely inspected, maintained, and repaired or replaced before
failure is likely, there is less chance that hazardous exposures will
occur.
Workers must know the proper way to perform their job tasks in
order to minimize their exposure to Cr(VI) and to maximize the
effectiveness of control measures. For example, if an exhaust hood is
designed to provide local ventilation and a worker performs a task that
generates a contaminant away from the exhaust hood, the control measure
will be of no use. Workers can be informed of proper operating
procedures through information and training. Good supervision further
ensures that proper work practices are carried out by workers. By
persuading a worker to follow proper procedures, such as positioning
the exhaust hood in the correct location to capture the contaminant, a
supervisor can do much to minimize unnecessary exposure.
Employees' exposures can also be controlled by scheduling
operations with the highest exposures at a time when the fewest
employees are present. For example, routine clean-up operations that
involve Cr(VI) releases might be performed at night or at times when
the usual production staff is not present.
Respirators are another important, although less preferred, method
of compliance. However, to be effective, respirators must be
individually selected; fitted and periodically refitted;
conscientiously and properly worn; regularly maintained; and replaced
as necessary. In many workplaces, these conditions for effective
respirator use are difficult to achieve. The absence of any of these
conditions can reduce or eliminate the protection the respirator
provides to some of all of the employees.
Respirator effectiveness ultimately relies on the good work
practices of individual employees. In contrast, the effectiveness of
engineering controls does not rely so routinely on actions of
individual employees. Engineering and work practice controls are
capable of reducing or eliminating a hazard from the workplace as a
whole, while respirators protect only the employees who are wearing
them correctly. Furthermore, engineering and work practice controls
permit the employer to evaluate their effectiveness directly through
air monitoring and other means. It is considerably more difficult to
directly measure the effectiveness of respirators on a regular basis to
ensure that employees are not unknowingly being overexposed. OSHA
therefore considers the use of respirators to be the least satisfactory
approach to exposure control.
In addition, use of respirators in the workplace presents other
safety and health concerns. Respirators can impose substantial
physiological burdens on employees, including the burden imposed by the
weight of the respirator; increased breathing resistance during
operation; limitations on auditory, visual, and odor sensations; and
isolation from the workplace environment. Job and workplace factors
such as the level of physical work effort, the use of protective
clothing, and temperature extremes or high humidity can also impose
physiological burdens on workers wearing respirators. These stressors
may interact with respirator use to increase the physiological strain
experienced by employees.
Certain medical conditions can compromise an employee's ability to
tolerate the physiological burdens imposed by respirator use, thereby
placing the employee wearing the respirator at an increased risk of
illness, injury, and even death. These medical conditions include
cardiovascular and respiratory diseases (e.g., a history of high blood
pressure, angina, heart attack, cardiac arrhythmias, stroke, asthma,
chronic bronchitis, emphysema), reduced pulmonary function caused by
other factors (e.g., smoking or prior exposure to respiratory hazards),
neurological or musculoskeletal disorders (e.g., epilepsy, lower back
pain), and impaired sensory function (e.g., a perforated ear drum,
reduced olfactory function). Psychological conditions, such as
claustrophobia, can also impair the effective use of respirators by
employees and may also cause, independent of physiological burdens,
significant elevations in heart rate, blood pressure, and respiratory
rate that can jeopardize the health of employees who are at high risk
for cardiopulmonary disease.
These concerns about the burdens placed on workers by the use of
respirators were acknowledged in OSHA's revision of its Respiratory
Protection standard, and are the basis for the requirement that
employers provide a medical evaluation to determine the employee's
ability to wear a respirator before the employee is fit tested or
required to use a respirator in the workplace (63 FR 1152, 1/8/98).
Although experience in industry shows that most healthy workers do not
have physiological problems wearing properly chosen and fitted
respirators, nonetheless common health problems can cause difficulty in
breathing while an employee is wearing a respirator.
In addition, safety problems created by respirators that limit
vision and communication must always be considered. In some difficult
or dangerous jobs, effective vision or communication is vital. Voice
transmission through a respirator can be difficult, annoying, and
fatiguing. In addition, movement of the jaw in speaking can cause
leakage, thereby reducing the efficiency of the respirator and
decreasing the protection afforded the employee. Skin irritation can
result from wearing a respirator in hot, humid conditions. Such
irritation can cause considerable distress to workers and can cause
workers to refrain from wearing the respirator, thereby rendering it
ineffective.
Because respirators are less reliable than engineering and work
practice controls and may create additional problems, OSHA believes
that primary reliance on respirators to protect workers is generally
inappropriate when feasible engineering and work practice controls are
available. All OSHA substance-specific health standards have recognized
and required employers to observe the hierarchy of controls, favoring
engineering and work practice controls over respirators. Moreover,
OSHA's enforcement experience with these standards has reinforced the
importance of this concept in the protection of employee health.
The Color Pigment Manufacturers Association suggested that supplied
air respirators provide an acceptable alternative to engineering
controls in many circumstances (Ex. 38-205, p. 44). The American
Foundry Society concurred with this opinion (Ex. 43-14). They claimed
that supplied air hoods do not present the problems and limitations
associated with the use of other respirators and are more reliable and
effective than most engineering controls (Tr. 1713-1717, Exs. 38-205;
43-14). The National Paint and Coatings Association (NPCA) indicated
that Cr(VI) exposures in paint and coatings manufacturing are sporadic
and are limited to a small number of processes and a few workers (Ex.
39-66). NPCA believed these exposures could be effectively controlled
with modern air purifying or supplied air respirators (Ex. 39-66).
While OSHA acknowledges that certain types of respirators may
lessen problems associated with breathing resistance and skin
discomfort, these respirators may still present safety concerns of
their own. OSHA does not believe that respirators provide employees
with a level of protection that is equivalent to engineering controls,
regardless of the type of respirator used. To summarize: engineering
and work practice controls are capable of reducing or eliminating a
hazard from the workplace; respirators only protect the employees who
are wearing them. In addition, the effectiveness of respiratory
protection always depends on the actions of employees, while the
efficacy of engineering controls is generally independent of the
individual.
It is well-recognized that certain types of respirators are
superior to other types of respirators with regard to the level of
protection offered, or impart other advantages. OSHA is currently
evaluating the level of protection offered by different types of
respirators in the Agency's Assigned Protection Factors rulemaking (68
FR 34036, 6/6/03). However, OSHA believes that engineering controls
offer more reliable and consistent protection to a greater number of
workers, and are therefore preferable to any type of respiratory
protection.
Collier Shannon Scott, on behalf of various steel industry groups,
maintained that OSHA should allow use of respiratory protection as a
primary control to achieve the PEL where respiratory protection is
currently used to comply with another OSHA standard (Exs. 38-233; 40-
12). Without such an allowance, it was claimed, employers would have to
add additional controls where employees are already wearing
respirators, which would impose "significant burden and expense on the
employer with no attendant benefit to the employee" (Ex. 38-233, p.
34). If an employer has adopted all feasible engineering controls to
address other workplace exposures (e.g., lead, cadmium), and no other
feasible engineering controls are available to limit Cr(VI) exposures,
the final Cr(VI) rule would not require additional engineering controls
to meet the new Cr(VI) PEL. On the other hand, if additional feasible
engineering controls are available that would reduce Cr(VI) exposures
that exceed the PEL, then these controls would justifiably be required.
OSHA believes these additional engineering controls would better
protect employees. As discussed previously, OSHA considers engineering
controls to be the most effective method of protecting employees and
allows respiratory protection only where such controls have been found
infeasible.
A number of responses to the proposal commented on the possibility
of including separate engineering control air limits, or SECALs, in the
final Cr(VI) rule. Several commenters maintained that SECALs were
unnecessary (Exs. 38-214; 38-220; 39-20). The majority of respondents
who expressed an opinion on this issue supported the use of SECALs (Tr.
373, 1701, 1732, Exs. 38-205; 38-215; 38-216; 38-218; 38-231; 39-43;
47-30). However, it was apparent that these commenters did not have a
common understanding of the basis for establishing SECALs or their
application in the workplace.
SECALs were included in one previous OSHA rule, the Cadmium
standard for general industry (29 CFR 1910.1027). In that rule, SECALs
were based on a two tiered approach to controlling worker exposures. As
described in the preamble to the final rule:
The first tier would be a PEL, set at the level required by the
health science data to protect workers' health. The PEL, in the case
of industries where compliance by means of engineering and work
practice controls was infeasible, could be achieved by any allowable
(e.g., not worker rotation) combination of work practice and
engineering controls and respirators. The second tier would be set
above the PEL at the lowest feasible level that could be achieved by
engineering and work practice controls (57 FR 42389, 9/14/92).
Thus, employers in all industries covered by the cadmium standard were
required to use engineering and work practice controls to the extent
feasible to achieve the PEL. For specified processes in particular
industries, SECALs provided explicit recognition of the lowest exposure
level that could feasibly be achieved with engineering and work
practice controls. Respirators could then be used as supplementary
controls to reduce exposures to the PEL.
While the cadmium standard is the only standard to use the term
"SECAL" other standards have adopted the same approach. For example,
although the PEL in the lead standard is set at 50 [mu]g/m\3\ (29 CFR
1910.1025(c)) the brass and bronze ingot manufacture industry sector is
only required to achieve a lead in air concentration of 75 [mu]g/m\3\
through engineering and work practice controls (29 CFR 1910.1025(e)(1)
Table I, n.3). As with all industry sectors, brass and bronze ingot
manufacture must provide respiratory protection to supplement
engineering and work practice controls if they cannot achieve the PEL.
Similarly, the asbestos standard exempts certain specified operations
from meeting the PEL of 0.1 fiber per cubic centimeter of air (0.1
fiber/cm\3\) through engineering controls, but requires such operations
to use such controls to get down to 0.5 fiber/cm\3\ or 2.5 fibers/cm\3\
for short term exposures and to provide supplemental respiratory
protection (29 CFR 1910.1001(f)(1)(iii)).
Public Citizen maintained that SECALs could be used to provide a
more protective PEL. According to Public Citizen, technological
feasibility considerations applicable to a relatively small number of
workers should not form the basis for establishing a PEL. They said
that if OSHA determines that a lower PEL is not feasible in limited
applications through use of engineering and work practice controls, the
Agency should use SECALs to allow for use of respirators in those
applications (Tr. 721, Ex. 47-23). However, SECALs (or equivalent
provisions) can only be applied to discrete operations that can
be distinguished from other sources of Cr(VI) exposure. As discussed
with regard to the PEL in paragraph (c) of this Summary and
Explanation, this is not the case for most operations involving Cr(VI)
exposure. Moreover, and also as discussed with regard to paragraph (c),
the established test for technological feasibility for standards
requires that the PEL be achieved in most operations with engineering
and work practice controls.
On the other hand, a number of commenters supported SECALs in the
belief that they would lessen the burdens imposed on employers. These
parties appeared to believe that SECALs would allow them to circumvent
the hierarchy of controls and use respiratory protection to achieve the
PEL, even when feasible engineering controls were available. This
approach was advocated by Elementis Chromium and the Chrome Coalition
(Exs. 38-216; 38-231).
As discussed previously, OSHA considers engineering and work
practice controls to be superior to respiratory protection for
controlling workplace exposures to Cr(VI). The Agency, therefore, does
not consider it appropriate to allow regular use of respirators to
achieve the PEL when feasible engineering and work practice controls
are available. The scenario envisioned by some commenters, which
apparently involves a SECAL established at some point higher than the
lowest level achievable with engineering and work practice controls,
would therefore compromise worker safety by allowing an inferior method
of control to substitute for a superior and feasible method.
OSHA does recognize, however, that an administrative burden can be
relieved by providing explicit recognition in the final rule of
operations where the PEL cannot be achieved through use of engineering
and work practice controls alone. In these instances, absent
recognition of infeasibility in the standard, the employer would need
to be able to demonstrate that feasible engineering and work practice
controls could not achieve the PEL.
As discussed in Chapter III of the Final Economic Analysis, OSHA
has determined that during certain painting operations in the aerospace
industry, the PEL of 5 [mu]g/m\3\ cannot be achieved with engineering
and work practice controls (Ex. 49). In these operations, the evidence
indicates that employee exposure to Cr(VI) can feasibly be reduced to
25 [mu]g/m\3\ using engineering and work practice controls; respiratory
protection is necessary to supplement these controls to achieve the
PEL. Accordingly, a provision has been added to the final rule
recognizing the limitations of engineering and work practice controls
in controlling Cr(VI) exposures where painting of aircraft or large
aircraft parts is performed in the aerospace industry. In using the
term "aircraft or large aircraft parts" OSHA is referring to the
interior or exterior of whole aircraft, aircraft wings, tail sections,
wing panels and rocket sections, large aircraft body sections, control
surfaces such as rudders, elevators, and ailerons, or comparably sized
aircraft parts. Thus, in these operations employee exposures must be
reduced to 25 [mu]g/m\3\ or less using engineering and work practice
controls. Respiratory protection will then need to be used to achieve
the PEL.
There may even be some situations where the engineering and work
practice controls cannot achieve exposures of 25 [mu]g/m\3\. The final
rule recognizes this and addresses this by permitting the employer to
demonstrate the infeasibility of achieving 25 [mu]g/m\3\ with these
controls. In these limited circumstances the employer would be
permitted to further rely on respirators to protect employees.
OSHA acknowledges that engineering and work practice controls
cannot feasibly achieve the PEL in some specific operations. In
particular, OSHA is aware that the use of engineering and work practice
controls to comply with the PEL is infeasible for some maintenance and
repair operations and during emergency situations. These situations are
recognized in paragraph (g) of the final rule (paragraph (f) for
construction and shipyards), which addresses use of respiratory
protection where employers can demonstrate that engineering and work
practice controls are not feasible. In such situations, the burden of
proof is appropriately placed on the employer to make and support a
claim of infeasibility because the employer has better access to
information specific to the particular operation that is relevant to
the issue of feasibility.
An exception to the general requirement for primary reliance on
engineering and work practice controls is included in the final rule
for employers who do not have employee exposures above the PEL for 30
or more days per year (during 12 consecutive months) in a particular
process or task. Thus, if a particular process or task causes employee
exposures to Cr(VI) that exceed the PEL on 29 or fewer days during any
12 consecutive months, the employer is allowed to use any combination
of controls, including respirators alone, to achieve the PEL. The
obligation to implement engineering and work practice controls to
comply with the PEL is not triggered until a process or task causes
employees to be exposed above the PEL on 30 or more working days during
a year.
The employer may use this exception if he or she can demonstrate
that a process or task will not cause employee exposures above the PEL
for 30 or more days per year (12 consecutive months). The burden of
proof is on the employer to show that exposures do not exceed the PEL
on 30 or more days per year. OSHA believes this provision provides
needed flexibility to employers, while still providing adequate
protection for workers.
Under current exposure conditions, the primary adverse health
effect addressed by this final rule (i.e., lung cancer) is associated
with cumulative exposure to Cr(VI). Thus, assuming stable exposure
levels, the fewer number of days that a worker is exposed, the lower
the risk incurred. Consequently, some exception based on the number of
days of exposure is justified.
OSHA realizes that in some industries (e.g., color pigment
manufacturing), exposure to Cr(VI) is typically infrequent (i.e., fewer
than 30 days, over 12 consecutive months). For example, certain Cr(VI)
processes may occur only several days a year when production of a
particular product is needed. Under such conditions, it may not be cost
effective or very beneficial to workers' health for employers to invest
the monies needed to install engineering controls to control Cr(VI) to
the PEL. Without this exception, employers would be required to
implement feasible engineering controls and work practice controls
wherever employees are exposed to Cr(VI) above the PEL, even if they
are only exposed on one or several days a year. OSHA believes that the
expense of implementing engineering controls in such circumstances is
not reasonable.
A number of commenters expressed general support for this exception
(e.g., Tr. 1426-1427, 1730; Exs. 38-205; 38-218; 38-220; 38-235; 39-19;
39-20; 39-47; 39-51; 40-1; 47-31). For example, the Navy expressed the
view that this provision allowed employers to focus on the most serious
hazards:
This 30-day threshold approach reflects the reality and
challenges of the Maritime Industry and has value in the
shipbuilding and repair industry. The concept allows employers to
focus engineering and work practice controls on those operations
having the potential to result in the greatest cumulative exposure
while providing the flexibility to address lower-exposure operations
based on a hazard assessment approach (Ex. 38-220).
Some commenters requested that the parameters of the exception be
expanded to apply to exposures that occur more frequently, but for
short durations of time (e.g., a few minutes per day), or to a longer
time period (i.e., a greater number of days)(Tr. 558-559, 1807-1809,
Exs. 38-218; 38-205; 47-31). Another commenter argued that, if an
exception was to be included in the final rule, it should be limited to
situations where exposure at any level occurs on fewer than 30 days
(Ex. 39-71).
OSHA believes that the threshold exposure duration of fewer than 30
days per year is appropriate. With this exception, OSHA intends to
provide relief exclusively to employers whose operations result in
employee exposure to Cr(VI) at or above the PEL only for short periods
of time. Because the PEL is expressed as an 8-hour time-weighted
average, it is appropriate to express this exception in terms of a
given number of days. Exposures that occur for short durations of time
during the day are balanced by longer time periods when no exposure
occurs. The PEL therefore already addresses most situations where
exposures occur for only a few minutes during the day. If the brief
exposures are so high that they cause the 8-hour time weighted average
exposure to exceed the PEL, it is appropriate that they be considered
equivalent to other exposure scenarios where the PEL is exceeded.
The question, then, is what number of days should be selected as
the maximum, above which engineering and work practice controls must be
implemented. There is no simple, scientifically definitive answer to
this question. OSHA believes that the choice of 30 or more working days
per year provides a reasonable balance between the preference for the
more reliable engineering and work practice controls, and the desire to
focus resources on those exposures that present the greatest risks to
workers.
The choice of providing the limited exception for exposures on
fewer than 30 working days per year is also consistent with the lead
and cadmium standards, which incorporate a similar exception. Further,
the 30 day exception is congruent with the 30 day exposure trigger for
medical surveillance included in paragraph (k) of this standard
(paragraph (i) for construction and shipyards), which simplifies the
application of these provisions where employee exposures are tied to a
single process or task. For example, if an employer has employees
exposed to Cr(VI) while performing a single process or task, and the
employer determines that exposures do not occur on 30 or more days per
year, the employer has established that (1) any combination of controls
can be used to achieve the PEL; and (2) no medical surveillance is
necessary unless an employee develops signs or symptoms of the adverse
health effects associated with Cr(VI) exposure or is exposed in an
emergency situation. In any event, OSHA believes that the 30 day
designation is reasonable and no other number of days would be a more
appropriate benchmark. The Agency concludes the 30 working day
exclusion will make the standard more flexible in workplaces where
exposure days are limited.
Several commenters did not believe that an exception to the general
requirement for use of engineering and work practice controls should be
included in the final Cr(VI) rule (Tr. 558-559, 766, 1433, 1807, Exs.
38-199; 38-214; 38-219; 39-71; 40-10-2; 40-18-1; 40-19-1). For example,
NIOSH maintained that such a provision would represent a significant
weakening of the requirement for priority of engineering controls in
preference to respirators (Ex. 40-10-2). OSHA agrees that engineering
and work practice controls are generally superior to respirators.
However, as discussed earlier, the Agency believes an exception for a
limited duration of exposure is a reasonable way to focus resources on
areas where the highest exposures are likely to occur and that the
requirement for respirator use in these situations will provide
sufficient protection for these workers.
Several respondents contended that it would be difficult to track
employee exposure days, apparently believing that the exemption would
be based on the exposures of individual workers, rather than the
exposures created by a process or task (e.g., Tr. 1433, Ex. 40-19-1).
OSHA intends for this exception to be process-or task-based: i.e., it
is specific to a process where engineering controls might be
implemented to reduce exposures to or below the PEL. For example, an
employer might have two processes, A and B, where A involves an ongoing
process in the facility with exposures above the PEL for 30 or more
days and another process, B, that results in exposures above the PEL
for 29 or fewer days per year. The fact that the employer has employees
exposed above the PEL for more than 30 days in process A will not be
used to determine that engineering and work practice controls have to
be used for process B. OSHA intends this exception to be similarly
applied by process or task in the construction and shipyard
environments where employees may move from one work site to another.
By basing the exception on the process or task being performed,
OSHA aims to preclude employers from using job rotation as a means of
limiting the number of days individual employees are exposed above the
PEL. Job rotation does not reduce the risk faced by workers, but only
distributes that risk among a larger worker population. Therefore, OSHA
considers the process or task to be the appropriate basis for applying
this exception, rather than basing an exception on the number of days
that an individual worker is exposed.
Some responses to the proposal did not consider the criteria used
to qualify for the exception to be sufficiently clear (Tr. 765, Exs.
39-65; 40-18-1). The proposal indicated that this exception would apply
where the employer "has a reasonable basis for believing that no
employee in a process or task will be exposed above the PEL for 30 or
more days per year." To clarify the Agency's intent, this language has
been modified to indicate that the employer can take advantage of the
exception when he or she "can demonstrate that no employee in a
process or task will be exposed above the PEL for 30 or more days per
year." This revised language makes clear that the employer has the
burden to demonstrate that a process or task does not result in
employee exposures above the PEL for 30 or more days per year. The
burden of proof is placed on the employer because the employer has
access to the necessary information about employee exposure levels and
processes and tasks at the worksite. Where existing information is
inadequate, the employer is also in the best position to develop the
necessary information.
Historical data, objective data, or exposure monitoring data may be
used to demonstrate that employees will not be exposed above the PEL
for 30 or more days per year. Other information, such as production
orders showing that processes involving Cr(VI) exposures are conducted
on fewer than 30 days per year, may also demonstrate that employees
will not be exposed above the PEL for 30 or more days per year. The
obligation to demonstrate that employees in a process or task will not
be exposed above the PEL for 30 or more days per year is the same for
general industry, construction, and shipyard employers.
OSHA has included a provision in the final rule prohibiting the
rotation of employees to different jobs as a means of achieving the
PEL. Although rotation of employees may reduce the risk of
cancer among individual workers, the practice places a larger pool of
workers at risk. Since no threshold has been established for the
carcinogenic effects of Cr(VI), rotation would not be expected to
reduce the risk to the population of workers when considered as a
whole. A prohibition on worker rotation to achieve the PEL was
supported by several responses to the proposal (e.g., Exs. 38-199-1;
40-10-2) and is consistent with good industrial hygiene practice. A
prohibition on worker rotation to achieve the PEL is also consistent
with many OSHA standards regulating carcinogens such as those for 1,3-
butadiene (29 CFR 1910.1051), methylene chloride (29 CFR 1910.1052),
asbestos (29 CFR 1910.1001), and cadmium (29 CFR 1910.1027).
A number of commenters, however, objected to a prohibition on
worker rotation to achieve the PEL (e.g., Exs. 38-205; 38-214; 38-218;
38-228; 38-233; 39-51; 39-60; 47-30-1). For example, the Society for
the Plastics Industry argued that employers should be allowed to
implement employee rotation where it will result in exposure levels
that are not associated with a significant risk of cancer (Ex. 38-218,
pp. 29-30). However, worker rotation to lower the exposures of
individual employees simply distributes exposures among a larger number
of workers. The intent of this final rule is not simply to achieve a
PEL, but to protect the largest number of workers possible from the
adverse health effects of Cr(VI) exposure, particularly lung cancer. If
the exposures of individual employees are reduced, but a corresponding
increase occurs in the total number of employees exposed, then the
intent of the final rule would be undermined.
Several commenters argued that job rotation has been allowed in
previous OSHA health standards such as those for arsenic, formaldehyde,
and lead, and should be allowed in this case as well (e.g., Exs. 38-
218; 38-228; 47-30). With regard to arsenic and formaldehyde, although
worker rotation was not specifically prohibited, the preamble
discussions for each of these final standards indicated that the Agency
did not consider worker rotation to be an appropriate control strategy
(43 FR 19584, 19617(5/5/78); 52 FR 46168, 46263-46264 (12/4/87)).
OSHA's Lead standard was issued in 1978, and was based on a range
of adverse health effects including damage to the nervous, urinary, and
reproductive systems and inhibition of heme synthesis. Based on the
information available at that time, lead was not recognized by OSHA as
a carcinogen, and worker rotation was regarded as "a relatively safe
and effective means of maintaining TWA levels below permissible
limits" (43 FR 52952, (11/14/78)). The preamble to the final lead rule
noted that such practices were unacceptable "when the contaminant is
one for which no effect levels are unknown, e.g., carcinogens" (43 FR
52952, (11/14/78)). The Lead standard therefore does not set a
precedent for allowing worker rotation for a carcinogen such as Cr(VI).
OSHA recognizes that employers rotate workers for a variety of
reasons. For example, an employer may rotate workers in order to
provide cross-training on different tasks, or to allow workers to
alternate physically demanding tasks with less strenuous activities.
OSHA does not place any restrictions on worker rotation when it is
conducted for reasons other than compliance with the PEL. The Agency
does not intend for this provision to be interpreted as a general
prohibition on employee rotation where workers are exposed to Cr(VI).
Some commenters believed that the hierarchy of controls should
apply to dermal as well as inhalation exposures to Cr(VI)(Exs. 38-199-
1; 38-219). OSHA agrees that engineering and work practice controls can
often be useful in controlling dermal Cr(VI) exposures. In fact, the
Agency believes that engineering and work practice controls used to
limit inhalation exposures to or below the PEL will often be effective
in limiting dermal exposures as well. Substitution, isolation, and
ventilation all serve to control dermal as well as inhalation
exposures.
As discussed in section V of this preamble, OSHA recognizes that
dermal exposures to Cr(VI) are capable of causing serious adverse
health effects. However, dermal exposures do not present the same level
of risk as inhalation exposures. Moreover, OSHA does not anticipate
that engineering and work practice controls will eliminate the need for
protective clothing and equipment and hygiene facilities for protection
from dermal hazards. Therefore, due to the limited benefits that would
be expected from such a provision, OSHA does not believe that a
requirement for preferential use of engineering and work practice
controls to reduce dermal exposures is reasonably necessary in this
final rule. This determination is consistent with previous OSHA health
standards, including standards addressing adverse dermal effects (e.g.,
formaldehyde (29 CFR 1910.1048) and 1,2-dibromo-3-chloropropane (29 CFR
1910.1044)).
Several commenters advocated a task-based approach for specifying
required methods of compliance (Exs. 38-219; 38-235; 40-10-2). Others
indicated that they did not see any benefit to this approach (Exs. 38-
220; 39-20). Under a task-based approach, appropriate control measures
would be specified for particular tasks and employers would be required
to implement the specified controls when employees perform that task.
This approach was used in OSHA's standards for exposure to asbestos in
construction (29 CFR 1926.1101) and shipyards (29 CFR 1915.1001).
However, sufficient information is not available in this rulemaking
record to allow OSHA to establish the specific and detailed
requirements that would be necessary to address the various tasks
covered under the rule.
In the standards for asbestos in construction and shipyards, OSHA
was able to divide the vast majority of activities involving asbestos
exposure into four classes, and to identify control measures that were
generally appropriate for each of the four classes of work. The Agency
is unable to make comparable categorizations for the types of work
covered in this rulemaking. For example, welding operations may involve
substantially different potential Cr(VI) exposures depending upon the
chromium content of the steel being welded and consumables used, the
type of welding being performed, and the environment where the welding
takes place. Appropriate control measures will vary based on these
factors. Because OSHA is unable to specify generally applicable
controls for common tasks involving exposure to Cr(VI), the Agency
considers the performance-oriented approach used in this final rule to
be the only reasonable approach for methods of compliance to control
exposures to Cr(VI). The approach used in this rule is consistent with
most other OSHA substance-specific health standards, including those
for cadmium in construction (29 CFR 1926.1127) and lead in construction
(29 CFR 1926.62).
OSHA has not included a requirement for a written compliance
program in the final rule. In some previous standards, the Agency has
required that employers prepare a written document detailing the
measures used to achieve compliance. This document typically was
required to include a description of operations that result in
exposure; specific methods used to control exposures; a detailed
implementation schedule; a work practice program; a plan for
emergencies; and other information. The purpose of requiring an
employer to establish a written compliance program is to promote
compliance with the standard. Some urged OSHA to include a provision for
a written compliance program in the Cr(VI) standard (Ex. 38-199-1; 39-71; 40-19-1).
OSHA has not included a provision for compliance plans in the
Cr(VI) standard in order to limit the amount of paperwork employers
would be required to complete. The Paperwork Reduction Act of 1995 (44
U.S.C. 3501 et seq.) requires agencies to minimize paperwork burdens
imposed on the public. Preparation of written compliance plans would be
classified as paperwork under that Act. Although a written program may
be useful to some employers, OSHA does not believe that the lack of a
written compliance program will substantially reduce the effectiveness
of the standard. This finding is consistent with OSHA health standards
such as those for formaldehyde (29 CFR 1910.1048) and methylene
chloride (29 CFR 1910.1052). Compliance with this standard will be
promoted through outreach, which OSHA has concluded will be effective
in assisting employers and employees to comply.
(g) Respiratory Protection
Paragraph (g) of the general industry standard (paragraph (f) for
construction and shipyards) establishes the final rule's requirements
for use of respiratory protection. Employers are required to provide
employees with respiratory protection when engineering controls and
work practices cannot reduce employee exposure to Cr(VI) to within the
PEL. Specifically, respirators are required during the installation and
implementation of engineering and work practice controls; during work
operations where engineering and work practice controls are not
feasible; when all feasible engineering and work practice controls have
been implemented, but are not sufficient to reduce exposure to or below
the PEL; during work operations where employees are exposed above the
PEL for fewer than 30 days per year, and the employer has elected not
to implement engineering and work practice controls to achieve the PEL;
and during emergencies. Where respirator use is required, the employer
must institute a respiratory protection program in accordance with
OSHA's Respiratory Protection standard (29 CFR 1910.134).
These requirements for the use of respirators are identical to
those proposed and are generally consistent with other OSHA health
standards, such as those for 1,3 butadiene (29 CFR 1910.1051) and
methylene chloride (29 CFR 1910.1052). They reflect the Agency's
determination, discussed in the section on methods of compliance, that
respirators are inherently less reliable than engineering and work
practice controls. OSHA therefore will allow reliance on respirators
only in limited situations.
OSHA received relatively few comments specifically addressing the
proposed respiratory protection requirements. A numbers of comments
focused on the use of respiratory protection in lieu of engineering and
work practice controls (e.g., Exs. 38-199; 38-214; 38-219; 38-220; 38-
231; 38-232; 38-233; 39-47; 39-51; 39-57; 39-60; 39-65; 39-66; 40-1;
40-7; 40-18; 40-19; 47-3; 47-31). This issue is addressed in the
methods of compliance section above.
OSHA recognizes that respirators may be essential to reduce worker
exposure in certain circumstances where engineering and work practice
controls cannot be used to achieve the PEL (e.g., in emergencies, or
during periods when equipment is being installed), or where engineering
controls may not be reasonably necessary (e.g., where employees are
exposed above the PEL for fewer than 30 days per year), and provision
is made for their use as primary controls in these situations. In other
circumstances, where feasible work practices and engineering controls
alone cannot reduce exposure levels to the PEL, respirators must be
used for supplemental protection. In these situations, the burden of
proof is placed on the employer to demonstrate that engineering and
work practice controls are not feasible.
OSHA anticipates that engineering and work practice controls will
generally be in place within four years of the effective date of the
standard, as specified in paragraph (n) of the final rule (paragraph
(l) for construction and shipyards). The Agency realizes that in some
cases employers may commence operations that involve employee Cr(VI)
exposures after that date, may install new or modified equipment, or
make other workplace changes that result in new or additional exposures
to Cr(VI). In these cases, a reasonable amount of time may be needed
before appropriate engineering controls can be installed and proper
work practices implemented and paragraph (g)(1)(i) addresses this
situation. Employers are expected to provide respirators to protect
workers during such periods.
Respiratory protection is also required during work operations
where engineering and work practice controls are not feasible. OSHA
anticipates that there will be few situations where no engineering and
work practice controls are feasible to limit employee exposure to
Cr(VI). However, the Agency recognizes that it may be infeasible to
control Cr(VI) exposure with engineering and work practice controls
during certain work operations, such as maintenance and repair
activities. Respirators are required in these situations. Several
commenters supported allowing the use of respiratory protection in
these circumstances (e.g., Exs. 38-254; 39-47; 39-56).
In other cases, some engineering and work practice controls may be
feasible, but these controls may not be capable of lowering employee
exposures to or below the PEL. For example, OSHA recognizes that in
certain welding operations such as welding stainless steel in confined
spaces, the PEL cannot always be achieved with feasible engineering and
work practice controls. In these cases, the employer must install
engineering controls and implement work practice controls where such
controls are feasible to reduce exposures, even if these controls
cannot reduce exposures to the PEL. Respirators must also be provided
to supplement the engineering and work practices controls to achieve
the PEL.
The requirement to provide respiratory protection when feasible
engineering controls are not sufficient to reduce exposures to within
the PEL also applies in instances where effective engineering controls
have been installed and are being maintained or repaired. In these
situations, controls may not be effective while maintenance or repair
is underway. Where exposures exceed the PEL, the employer is required
to provide respirators.
As discussed earlier with regard to methods of compliance, OSHA is
including an exception from the general requirement for use of
engineering and work practice controls where employee exposures do not
exceed the PEL on 30 or more days per year. Where this exception
applies, the employer is then required to provide respiratory
protection to achieve the PEL.
OSHA also believes that respirators must be used to protect
employees in emergencies. Since an emergency, by definition, involves
or is likely to involve an uncontrolled release of Cr(VI), it is
important for employers to have procedures to protect employees from
the significant exposures that may occur.
Whenever respirators are used to comply with the requirements of
the standard, the employer must implement a comprehensive respiratory
protection program in accordance with the
Agency's Respiratory Protection standard (29 CFR 1910.134). The
respiratory protection program is designed to ensure that respirators
are properly used in the workplace, and are effective in protecting
workers. The program must include procedures for selecting respirators
for use in the workplace; medical evaluation of employees required to
use respirators; fit testing procedures for tight-fitting respirators;
procedures for proper use of respirators in routine and reasonably
foreseeable emergency situations; procedures and schedules for
maintaining respirators; procedures to ensure adequate quality,
quantity, and flow of breathing air for atmosphere-supplying
respirators; training of employees in the proper use of respirators;
and procedures for evaluating the effectiveness of the program. This
provision serves as a reminder to employers covered by the Cr(VI) rule
that they must also comply with the Respiratory Protection standard
when respirators are provided to employees.
OSHA has proposed to revise the Respiratory Protection standard to
include assigned protection factors (APFs) (68 FR 34036 (6/6/03)). The
proposed revision includes a table which indicates the level of
respiratory protection that a given respirator or class of respirators
is expected to provide, and will apply to employers whose employees use
respirators for protection against Cr(VI) when it becomes a final rule
(68 FR 34036, 34115 (6/6/03)).
A number of commenters supported the reference to the Respiratory
Protection standard (e.g., Tr. 1586-1589, Exs. 38-232; 39-38; 39-57;
47-36). For example, the 3M Company stated:
Many of our customers use respirators to help protect workers
from exposures to multiple contaminants and the reference in the
Cr(VI) standard to the requirements of 1910.134 brings uniformity
that will result in better compliance and protection for workers
such as welders that have exposures to other metals besides Cr(VI)
and workers in the pigment industry that may have exposures to both
cadmium and Cr(VI) (Ex. 38-232).
In contrast, the AFL-CIO suggested specific changes to the proposed
respiratory protection requirements. The AFL-CIO recommended that OSHA
require HEPA filters for all air purifying respirators required in the
final rule (Ex. 38-222). They argued that HEPA filters would provide
the highest level of protection, and a requirement to provide HEPA
filters would be consistent with similar provisions in other OSHA
health standards such as those for asbestos, lead, and cadmium.
OSHA does not believe that a specific requirement mandating use of
HEPA filters for air purifying respirators used for protection from
Cr(VI) is justified, and has not included such a requirement in the
final rule. For air-purifying respirators, in addition to the option of
providing a respirator equipped with a filter certified by NIOSH under
30 CFR Part 11 as a HEPA filter, the Respiratory Protection standard
allows employers several alternatives. Under 1910.134 the employer may
also provide either (1) An air-purifying respirator equipped with a
filter certified for particulates by NIOSH under 42 CFR Part 84; or (2)
an air-purifying respirator equipped with any filter certified for
particulates by NIOSH where dealing with contaminants consisting
primarily of particles with mass median aerodynamic diameters (MMAD) of
at least 2 micrometers. OSHA believes these requirements are
appropriate for protection from exposures to Cr(VI).
NIOSH published revised requirements for testing and certification
procedures for non-powered, air-purifying, particulate-filter
respirators and recodified the previous certification standards for
other respirator classes as 42 CFR Part 84 on June 8, 1995. Respirators
certified under Part 84 have passed a more demanding certification test
than was previously required, involving the most penetrating particle
size of 0.3 micrometers. OSHA believes that these testing and
certification requirements ensure that particulate filters certified
under 42 CFR Part 84 are efficient in preventing the penetration of
submicron-sized particles, and recognized this when the Agency's
revised Respiratory Protection standard was issued on January 8, 1998.
OSHA likewise believes that an air-purifying respirator equipped with
any filter certified for particulates by NIOSH will be efficient in
preventing the penetration of particles with diameters of 2 micrometers
or more, because filters will be more efficient in protecting against
particles larger than 0.3 micrometers in diameter. These findings were
established for air contaminants in general during the rulemaking that
revised the Respiratory Protection standard, and OSHA does not find any
basis in this rulemaking record to make an exception for Cr(VI).
The AFL-CIO suggested that the final Cr(VI) rule should prohibit
the use of disposable particulate (filtering facepiece) respirators for
protection against Cr(VI) exposures (Ex. 38-222). The AFL-CIO indicated
that they believed the record for OSHA's APFs rulemaking (Docket H049C)
supports the position that disposable particulate respirators do not
provide the same level of protection as do elastomeric half mask
respirators, and noted that OSHA does not allow the use of disposable
respirators under the Agency's Asbestos standard.
As noted above, OSHA is in the process of establishing respirator
selection provisions in the APFs rulemaking, which will modify the
Agency's Respiratory Protection standard. It is the Agency's intent
that substance-specific standards, such as this final Cr(VI) rule,
should refer to provisions of the Respiratory Protection standard
(including the generic APFs) where possible instead of establishing
their own separate respirator selection requirements. The record for
the Cr(VI) rulemaking contains no evidence to support separate
respirator selection requirements for Cr(VI), such as a prohibition or
restriction on the use of disposable particulate respirators. As no
basis has been established for distinguishing Cr(VI) from other air
contaminants, OSHA believes it is appropriate for employers required to
provide respirators for protection against Cr(VI) to follow the
provisions of the Respiratory Protection standard.
Pinnacle West Capital Corporation, parent company of Arizona Public
Service Company, expressed the view that the respiratory protection
requirements of the proposed rule could conflict with requirements of
the Nuclear Regulatory Commission (NRC). Referring to operations in the
firm's nuclear power plant, Pinnacle West stated:
* * * the potential exists for respiratory requirements under
this rule to be in conflict with Nuclear Regulatory Commission
expectations for keeping radiation exposures "As Low as Reasonably
Achievable" (ALARA). In some cases, the use of a respirator can
increase the stay time in a radioactive area, thus increasing the
time exposed to an external radiation dose. In such cases, ALARA
practice requires that a respirator not be used (Ex. 39-40).
OSHA does not forsee a conflict between the final rule's requirements
for use of respiratory protection and NRC requirements for minimizing
radiation exposure. NRC and OSHA share jurisdiction over occupational
safety and health at NRC-licensed facilities. With regard to
respiratory protection, NRC standards apply when the hazard is
radiation. However, the NRC standards explicitly recognize in Appendix
A to 10 CFR Part 20 that respirator use must comply with Department of
Labor requirements when chemical or other respiratory hazards exist
instead of, or in addition to, radioactive hazards. The responsibilities
of each agency for worker protection are discussed in a memorandum of
understanding (MOU) between NRC and OSHA (available at
http://www.osha.gov/pls/oshaweb/owadisp.show_document?p_table=MOU&p_id=233).
As NRC's Regulatory Guide 8.15--Acceptable Programs for Respiratory Protection indicates,
"The MOU makes it clear that if an NRC licensee is using respiratory
protection to protect workers against nonradiological hazards, the OSHA
requirements apply" (see http://www.nrc.gov/reading-rm/doc-
collections/reg-guides/occupational-health/active/8-15/ #--1--6). NRC
thus recognizes that respiratory protection for chemical hazards may be
required, and the provisions for respirator use in the final Cr(VI)
rule do not conflict with NRC requirements.
Several commenters expressed the opinion that respiratory
protection should be provided at no cost to employees (e.g., Exs. 38-
219; 38-222; 39-50). OSHA's Respiratory Protection standard explicitly
requires that respirators, as well as associated training and medical
evaluations, be provided at no cost to employees (29 CFR
1910.134(c)(4)). The Agency believes that the Respiratory Protection
standard adequately establishes this requirement; therefore, repetition
of the requirement in this Cr(VI) standard is unnecessary.
(h) Protective Work Clothing and Equipment
Paragraph (h) of the final rule (paragraph (g) for construction and
shipyards) sets forth requirements for the provision of protective
clothing and equipment. The rule requires the employer to provide
appropriate protective clothing and equipment at no cost to employees
where a hazard is present or is likely to be present from skin or eye
contact with Cr(VI). Ordinary street clothing and work uniforms or
other accessories that do not serve to protect workers from Cr(VI)
hazards are not considered protective clothing and equipment under this
standard. The employer is also required to ensure that employees use
the clothing and equipment provided, and follow a number of specified
practices to ensure that protective clothing and equipment is used and
handled in a manner that is protective of employee health.
These requirements are intended to prevent the adverse health
effects associated with dermal exposure to Cr(VI) (described in Section
V.D of this preamble) and the potential for inhalation of Cr(VI) that
would otherwise be deposited on employees' street clothing. The
requirements further serve to minimize exposures to Cr(VI) that may
occur as a result of improper handling of contaminated protective
clothing or equipment. The requirements of this paragraph are based
upon widely accepted principles and conventional practices of
industrial hygiene, and are similar to provisions for protective
clothing and equipment in other OSHA health standards such as those for
cadmium (29 CFR 1910.1027) and methylenedianiline (29 CFR 1910.1050).
The requirements are also consistent with Section 6(b)(7) of the OSH
Act which states that, where appropriate, standards shall prescribe
suitable protective equipment to be used in connection with hazards.
A number of responses to the proposal expressed the view that
requirements for protective clothing and equipment in a final Cr(VI)
standard would duplicate OSHA's existing generic requirements for
personal protective equipment (Tr. 1320-1321, 1389, Exs. 38-124; 38-
127; 38-214; 38-217; 38-218, p. 23; 38-229; 38-233, p. 39; 39-20; 47-
25). OSHA acknowledges that the Agency's generic personal protective
equipment standards (29 CFR 1910.132 for general industry; 29 CFR
1915.152 for shipyards; 29 CFR 1926.95 for construction) currently have
requirements for provision of protective clothing and equipment that
are essentially equivalent to the requirement in this final rule.
However, OSHA believes that the additional requirements contained in
this paragraph which address practices associated with the use of
protective clothing and equipment (e.g., removal and storage, cleaning
and replacement) are necessary and appropriate to provide adequate
protection from the hazards related to Cr(VI) exposure. Because these
additional provisions are closely associated with requirements for
protective clothing and equipment, including the protective clothing
and equipment requirements in this paragraph helps to make the
additional provisions clear and understandable. Also, OSHA believes it
is useful and appropriate for this rule to provide a consolidated set
of requirements for protective clothing and equipment that apply to
Cr(VI) exposures in the workplace, to the extent that this is
reasonably possible and beneficial. This provides an administratively
convenient source of information on these regulatory requirements, will
enable employers to more easily and effectively identify and implement
the measures necessary to protect employees, and will clarify that
additional requirements for protective clothing and equipment in this
standard are linked to the requirements currently in place.
One commenter maintained that OSHA had not shown that dermal
exposures present a significant risk, or that the proposed controls
(including provisions for change rooms and washing facilities included
in a subsequent paragraph of this standard) are reasonably necessary
and appropriate to address that risk (Ex. 38-218). OSHA disagrees.
While there were insufficient data to perform a quantitative risk
assessment on dermatitis, OSHA has established in the preamble
discussion of health effects that Cr(VI) is capable of causing serious
adverse effects to the skin and eyes, resulting in material impairment
of the health of affected individuals. Further, as discussed in regard
to significance of risk (Section VII of this preamble), without
appropriate control measures the effect of dermal exposures could
contribute to the significant risk presented by other workplace
exposures to Cr(VI). Moreover, as discussed below, these provisions are
not only reasonable and necessary but to a great extent reflect
requirements in existing generic standards. This approach is consistent
with other health standards where dermal hazards were present, where
OSHA has included requirements for protective clothing and equipment
(e.g., methylene chloride, formaldehyde).
One commenter suggested that the term "protective clothing and
equipment" be changed to "protective clothing and protective
equipment" (Ex. 39-65). OSHA has retained the term "protective
clothing and equipment" as proposed because the Agency believes it is
sufficiently clear, and is consistent with longstanding use of this
term by the Agency. The term "protective" serves to modify both the
word "clothing" and the word "equipment". When using the term
"protective clothing and equipment" OSHA is referring only to
clothing and equipment that serves to protect workers from Cr(VI)
hazards. Other clothing, work uniforms, tools, or other apparatus that
do not serve to protect workers from Cr(VI) hazards are not considered
protective clothing and equipment under this rule.
The final rule requires the employer to provide appropriate
protective clothing and equipment where a hazard is present or is
likely to be present from skin or eye contact with Cr(VI), but does not
specify criteria to be used for determining when a hazard is present or
is likely to be present. To make this determination, the employer must
evaluate the workplace. This performance-oriented requirement is consistent
with the current requirements of the Agency's standards for use of personal
protective equipment in general industry and shipyards, which require the
employer to assess the workplace to determine if hazards (including hazards
associated with eye and skin contact with chemicals) are present, or
are likely to be present (see, e.g., 29 CFR 1910.132(d)(1)).
To determine whether there is a hazard (or likely to be a hazard)
from skin or eye contact with Cr(VI) in a particular workplace, the
employer should "exercise common sense and appropriate expertise" in
assessing the hazards. (See non-mandatory appendices providing guidance
on hazard assessment in 29 CFR 1910 Subpart I Appendix B; 29 CFR 1915
Subpart I Appendix A). The recommended approach involves a walk-through
survey to identify sources of hazards to workers. Review of injury/
accident data is also recommended. Information obtained during this
process provides a basis for the evaluation of potential hazards.
Several commenters supported this approach to assessing Cr(VI)
hazards to the skin and eyes (Exs. 38-214; 38-220; 38-245-1; 39-19; 39-
20; 39-40; 39-47; 39-48; 39-52). Electric Boat Corporation, for
example, stated:
Electric Boat believes the approach is sound in that the
employer should perform a hazard assessment, like it does for many
other potential hazards in the workplace, and decide if protective
clothing and equipment is necessary to protect from adverse health
effects associated with the skin and eyes (Ex. 38-214).
The U.S. Navy also supported this method, indicating that "It is
appropriate to expect an employer to exercise common sense and
appropriate expertise to determine if a hazard is present or likely to
be present" (Ex. 38-220).
On the other hand, other commenters believed that such a
requirement was vague and subjective, and did not adequately indicate
when personal protective clothing was necessary (Tr. 626, Exs. 38-218;
38-233). One commenter complained that the proposal provided no
objective or quantitative basis for determining when a hazard exists,
and requirements for protective clothing and equipment could be
triggered by exposure to a few particles of dust (Ex. 38-233). Another
commenter requested that OSHA describe the conditions it believes
constitute skin and eye hazards, suggesting the inclusion of
descriptive phrases such as "a light dusting on the skin and work
surfaces" (Ex. 39-51).
One commenter suggested that protective clothing and equipment
should be required for employees exposed above the PEL (Ex. 39-71).
Other commenters argued that a blanket requirement that protective
clothing and equipment be provided for any exposures above the PEL was
not warranted (Exs. 38-214; 38-220; 38-245-1; 39-19; 39-20; 39-40; 39-
47; 39-48; 39-51; 39-52). Still other commenters considered that a
threshold concentration for the Cr(VI) content of mixtures should be
established, below which protective clothing would not be required
(Exs. 39-56; 38-254; 39-60). Establishing a threshold concentration, it
was argued, would help define where and when protective clothing would
be beneficial (Exs. 39-56; 38-254).
OSHA has not established quantitative thresholds for exposure to
Cr(VI) that would trigger the requirement for provision of protective
clothing and equipment. Cr(VI) is present in a large number of
different chemical compounds, each with differing physical and chemical
properties. These compounds themselves can be contained in a wide
variety of mixtures in various concentrations. The characteristics of
these compounds and mixtures can have substantial influence on the
ability of Cr(VI) to elicit adverse health effects to the skin and
eyes. Therefore, it is not possible to specify appropriate thresholds
for dermal or ocular effects from Cr(VI) containing compounds.
Exposures must be evaluated on a case-by-case basis, taking into
account factors such as the acidity or alkalinity of the compound or
mixture as well as the magnitude and duration of exposure. Clearly, the
employer, with knowledge of the workplace, work practices, and Cr(VI)
compounds used, is in the best position to evaluate whether personal
protective clothing or equipment are necessary and appropriate for his
or her workplace exposures.
OSHA is not aware of any evidence that would allow establishment of
a threshold concentration of Cr(VI) below which adverse skin or eye
effects would not occur. Likewise, the Agency does not have sufficient
evidence to demonstrate that a skin or eye hazard will necessarily
occur when exposures exceed the PEL. Therefore, OSHA believes that a
performance-oriented requirement for provision of protective clothing
and equipment is most appropriate for exposures to Cr(VI) covered by
this rule.
As part of this performance-oriented requirement, once a
determination has been made that a hazard is present or likely to be
present in the workplace, the employer must determine what clothing and
equipment are necessary to protect employees. The employer has
flexibility to select the clothing and equipment most suitable for his
or her particular workplace. The type of protective clothing and
equipment needed to protect employees from Cr(VI) hazards will depend
on the potential for exposure and the conditions of use in the
workplace. Examples of protective clothing and equipment that may be
necessary include, but are not limited to gloves, aprons, coveralls,
foot coverings, and goggles.
The employer must exercise reasonable judgment in selecting the
appropriate clothing and equipment to protect employees from Cr(VI)
hazards. In some instances gloves may be all that is necessary to
prevent hazardous Cr(VI) exposure. In other situations, such as when a
worker is performing abrasive blasting on a structure covered with
Cr(VI)-containing paint, more extensive measures such as coveralls,
head coverings, and goggles may be needed. Where exposures to Cr(VI)
are minute, such as in typical welding operations, no protective
clothing or equipment may be necessary. The chemical and physical
properties of the compound or mixture may also influence the choice of
protective clothing and equipment. For example, a chrome plater may
require an apron, gloves, and goggles to protect against possible
splashes of chromic acid that could result in both Cr(VI) exposure and
chemical burns. Other factors such as size, dexterity, and cut and tear
resistance should be considered in the selection process as well (Ex.
40-10-2).
This performance approach is consistent with OSHA's current
standards for provision of personal protective equipment and with
methods currently utilized to select appropriate protective clothing
and equipment. For example, several parties testified that they already
make qualitative determinations or exercise professional judgment in
selecting protective clothing and equipment in their workplaces (Tr.
924-925, 1259-1260, 1414-1416).
The final rule requires employers to provide clothing and equipment
necessary to protect against Cr(VI) hazards at no cost to employees.
Some commenters agreed with this approach (Tr. 1107-1108, 1438-1441,
Exs. 39-50; 38-199-1; 38-219-1; 38-222; 39-71; 40-10-2; 47-26). Others
disagreed, arguing either that the Agency should not include a
provision requiring employer payment or should defer to
the outcome of OSHA's ongoing rulemaking addressing payment for
personal protective equipment in all workplaces (64 FR 15401 (3/31/
99))(e.g., Exs. 38-214, p. 20; 38-244, p.11-12; 39-19; 39-47; 39-60).
OSHA has included a requirement that the employer pay for
protective clothing and equipment in the final rule because the Agency
believes that the employer is generally in the best position to select
and obtain the proper type of protective clothing and equipment for
protection from Cr(VI) hazards and to retain control over them. The
protective clothing and equipment at issue is designed and intended to
protect against Cr(VI) hazards at work. Because of the serious health
hazards associated with Cr(VI) exposure, employees may not remove
contaminated clothing and equipment from the worksite (except for the
employees whose job it is to launder, clean, maintain, or dispose of
such clothing or equipment). The employer is responsible for cleaning
or disposing of the protective clothing and equipment and retains
complete control over it. OSHA believes that by providing and owning
this protective clothing and equipment, the employer will maintain
control over the inventory of these items, conduct periodic
inspections, and, when necessary, repair or replace it to maintain its
effectiveness.
Employer payment for PPE has been a continuing issue for OSHA. OSHA
notes that in the generic rulemaking, the Agency has raised for public
comment, among other issues, whether employers should not be required
to pay for PPE that is personal in nature and used off the job, or that
is a "tool of the trade" typically supplied by the employee and
carried from job site to job site or employer to employer (65 FR 15401,
3/31/1999; 69 FR 41221, 7/8/2004). OSHA has not made a final
determination on any of the issues raised in the generic rulemaking.
The Agency notes that the protective clothing and equipment involved
here do not fall into either of these categories. Employees are not
allowed even to take the contaminated PPE home.
The determination that the protective clothing and equipment
required by the final standard is to be provided at no cost to
employees is specific to this Cr(VI) rule. It reflects the particular
considerations presented by workplace exposures to Cr(VI). The
determination is made without prejudice to the ongoing generic
rulemaking addressing payment for personal protective equipment.
The employer must ensure that protective clothing and equipment
contaminated with Cr(VI) is removed at the completion of the work shift
or at the completion of tasks involving Cr(VI) exposure, whichever
comes first. For example, if employees perform work tasks involving
Cr(VI) exposure for the first two hours of a work shift, and then
perform tasks that do not involve Cr(VI) exposure, they must remove
their protective clothing after the exposure period (in this case, the
first two hours of the shift). If, however, employees are performing
tasks involving Cr(VI) exposure intermittently throughout the day, or
if employees are exposed to other contaminants where protective
clothing and equipment are needed, this provision does not prevent them
from wearing the clothing and equipment until the completion of their
shift. This provision is intended to limit the duration of employees'
exposure, and to prevent contamination from Cr(VI) residues on
protective clothing reaching areas of the workplace where exposures
would not otherwise occur.
To limit exposures outside the workplace, the final rule requires
the employer to ensure that Cr(VI)-contaminated protective clothing and
equipment is removed from the workplace only by those employees whose
job it is to launder, clean, maintain, or dispose of such clothing or
equipment. This provision is intended to ensure that clothing
contaminated with Cr(VI) is not carried to employees' cars and homes,
increasing the worker's exposure as well as exposing other individuals
to Cr(VI) hazards. Furthermore, the standard requires that clothing and
equipment that is to be laundered, cleaned, maintained, or disposed of
be placed in closed, impermeable containers to minimize contamination
of the workplace and ensure employees who later handle these items are
protected. Those cleaning the Cr(VI)-contaminated clothing and
equipment will be further protected by warning labels placed on
containers to inform them of the potential hazards of exposure to
Cr(VI).
The proposed provision addressing labels on containers of
contaminated clothing and equipment has been modified to reference the
requirements of OSHA's Hazard Communication standard (HCS)(29 CFR
1910.1200). Rather than requiring the specific language proposed, the
final rule indicates that bags or containers are to be labeled in
accordance with the requirements of the HCS. As indicated in the
discussion of paragraph (l) of this standard below, OSHA believes that
it is appropriate maintain the labeling requirement but to allow
employers to retain the flexibility provided by the HCS with regard to
the language used on labels. The reference to the HCS is included to
remind employers of their obligation under that standard to label
containers of hazardous chemicals such as Cr(VI).
Several commenters objected to requirements for storage and
transport of contaminated items in impermeable bags or other
impermeable containers, as well as the associated labeling
requirements. The Textile Rental Services Association (TRSA) maintained
that such requirements were not justified, and that no evidence
indicated that laundry workers could be exposed to levels of Cr(VI)
that would be cause for concern (Tr. 1566-1572, Ex. 38-252). TRSA
claimed that the short processing time and minimal handling of garments
limits the potential exposure of laundry workers, and that reduction of
Cr(VI) to Cr(III) over time further limits potential exposure.
Moreover, TRSA argued that labels would cause unwarranted concerns and
lead to unnecessary testing. The Color Pigments Manufacturers
Association contended that the labeling required in the proposal would
lead to commercial laundries refusing to accept items contaminated with
Cr(VI), or accepting them only at significantly increased cost (Ex. 38-
205). Atlantic Marine also believed that laundries would refuse to
accept contaminated clothing (Tr. 926). It was also alleged that
contractors who repair and maintain equipment might refuse to accept
Cr(VI)-contaminated items (Ex. 38-233, p.39).
OSHA believes that the requirements of the final rule for use of
impermeable bags or other impermeable containers for the storage and
transport of Cr(VI)-contaminated items are clearly justified, as are
the requirements for labeling containers in accordance with the HCS. As
discussed previously, this rule requires protective clothing and
equipment when the employer has determined that a skin or eye hazard is
present or is likely to be present from exposure to Cr(VI). Thus,
protective clothing and equipment are only used under this rule in
situations where exposure to Cr(VI) is at least likely to cause a
hazardous exposure. The contamination of protective clothing and
equipment that results from such exposures poses a threat to the health
of workers who handle such clothing and equipment, just as it does to
the workers who use the clothing and equipment. Measures to minimize
the likelihood of hazardous exposures to workers who handle these
items, such as requirements for the use of impermeable
containers, are therefore reasonably necessary and appropriate.
Moreover, OSHA believes it is reasonable to use labels to inform
employers and employees who handle hazardous substances such as Cr(VI)
of the identity of these substances, as well as to provide appropriate
hazard warnings. This provision simply directs the employer's attention
to longstanding labeling requirements of the HCS. When employers and
employees are aware of the presence of Cr(VI) and its potential
hazards, appropriate measures can be implemented to protect employees.
The alternative of leaving those who handle these items in ignorance of
the presence of Cr(VI) discounts the very real possibility that adverse
health effects may occur if proper precautions are not taken. Other
OSHA health standards, such as those for lead (29 CFR 1910.1025),
asbestos (29 CFR 1910.1001), cadmium (29 CFR 1910.1027), and bloodborne
pathogens (29 CFR 1910.1030) include similar labeling requirements.
The final rule requires that the employer clean, launder, repair
and replace protective clothing as needed to ensure that the
effectiveness of the clothing and equipment is maintained. This
provision is necessary to ensure that clothing and equipment continue
to serve their intended purpose of protecting workers. This also
prevents unnecessary exposures outside the workplace from employees
taking contaminated clothing and equipment home for cleaning.
In keeping with the performance-orientation of the final rule, OSHA
does not specify how often clothing and equipment must be cleaned,
repaired or replaced. The Agency believes that appropriate time
intervals may vary widely based on the types of clothing and equipment
used, Cr(VI) exposures, and other circumstances in the workplace. The
obligation of the employer, as always, is to keep the clothing and
equipment in the condition necessary to perform its protective
functions.
Removal of Cr(VI) from protective clothing and equipment by
blowing, shaking, or any other means which disperses Cr(VI) in the air
is prohibited. Such actions would result in increased risk to employees
from unnecessary exposure to airborne Cr(VI) as well as possible dermal
contact.
The standard requires that the employer inform any person who
launders or cleans protective clothing or equipment contaminated with
Cr(VI) of the potentially harmful effects of exposure to Cr(VI), and
the need to launder or clean contaminated clothing and equipment in a
manner that effectively prevents skin or eye contact with Cr(VI) or the
release of airborne Cr(VI) in excess of the PEL. As with the provision
reminding employers of their obligation for labeling under the HCS,
this requirement is intended to ensure that persons who clean or
launder Cr(VI)-contaminated items are aware of the associated hazards
so they can take appropriate protective measures. Where laundry or
cleaning services are performed by third parties, the information
transmitted need not be extensive to accomplish this goal. Appropriate
hazard warnings, as required on labels by the HCS, will be sufficient
to indicate the potentially harmful effects of exposure to Cr(VI). In
addition, the language used in this provision (i.e., the clothing and
equipment should be laundered or cleaned in a manner that minimizes
skin or eye contact with Cr(VI) and effectively prevents the release of
airborne Cr(VI) in excess of the PEL) could be put on a label, thereby
fulfilling the requirements of the provision. The employer is not
expected to specify particular work practices that third parties must
follow to accomplish these objectives.
(i) Hygiene Areas and Practices
Paragraph (i) of the final rule (paragraph (h) for construction and
shipyards) requires employers to provide hygiene facilities and to
assure employee compliance with basic hygiene practices that serve to
minimize exposure to Cr(VI). The rule includes requirements for change
rooms and washing facilities, ensuring that Cr(VI) exposure in eating
and drinking areas is minimized, and a prohibition on certain practices
that may contribute to Cr(VI) exposure. OSHA believes that strict
compliance with these provisions will substantially reduce employee
exposure to Cr(VI).
Several of these provisions are presently required under other OSHA
standards. For example, OSHA's current standard addressing sanitation
in general industry (29 CFR 1910.141) requires that whenever employees
are required by a particular standard to wear protective clothing
because of the possibility of contamination with toxic materials,
change rooms equipped with storage facilities for street clothes and
separate storage facilities for protective clothing shall be provided.
The sanitation standard also includes provisions for washing
facilities, and prohibits storage or consumption of food or beverages
in any area exposed to a toxic material. Similar provisions are in
place for construction (29 CFR 1926.51). The hygiene provisions of this
paragraph are intended to augment the requirements established under
these other standards with additional provisions applicable
specifically to Cr(VI) exposure.
In workplaces where employees must change their clothes to use
protective clothing and equipment, OSHA believes it is essential to
have change rooms with separate storage facilities for street and work
clothing to prevent contamination of employees' street clothes. This
provision will minimize employee exposure to Cr(VI) after the work
shift ends, because it reduces the duration of time they may be exposed
to contaminated work clothes. Potential exposure resulting from
contamination of the homes or cars of employees is also avoided. Change
rooms also provide employees with privacy while changing their clothes.
OSHA intends the requirement for change rooms to apply to all covered
workplaces where employees must change their clothes (i.e., take off
their street clothes) to use protective clothing and equipment. In
those situations where removal of street clothes is not necessary
(e.g., in a workplace where only gloves are used as protective
clothing), change rooms are not required.
This provision reiterates the current requirements for change rooms
found in 29 CFR 1910.141(e) (for general industry and shipyards) and 29
CFR 1926.51(i) (for construction). Several commenters appeared to
interpret this provision to indicate a new obligation for employers to
provide change rooms that were not previously required (Tr. 557-558,
923-924, 1702, Exs. 38-205; 38-218; 38-233). The Agency's intent in
including this provision in the final rule is to provide a consolidated
reference of certain requirements for employers, rather than to
establish new and different requirements for change rooms. Change rooms
that meet the requirements of 29 CFR 1910.141(e) or 29 CFR 1926.51(i)
fulfill the change room requirements of this final Cr(VI) rule.
Paragraph (i)(3) (paragraph (h)(3) of the construction and shipyard
standards) contains requirements for washing facilities. The employer
must provide readily accessible washing facilities capable of removing
Cr(VI) from the skin and ensure that affected employees use these
facilities when necessary. Also, the employer must ensure that
employees who have skin contact with Cr(VI) wash their hands and faces
at the end of the work shift and prior to eating, drinking, smoking,
chewing tobacco or gum, applying cosmetics, or using the toilet. The
value and importance of washing facilities was recognized and supported by a
number of commenters (Tr. 1457, Exs. 38-244; 39-40; 39-41; 40-10-2; 47-
26).
Washing reduces exposure by diminishing the period of time that
Cr(VI) is in contact with the skin. Although use of appropriate
protective clothing and equipment is intended to prevent hazardous skin
and eye contact with Cr(VI) from occurring, OSHA realizes that in some
circumstances these exposures will occur. For example, a worker who
wears gloves to protect against hand contact with Cr(VI) may
inadvertently touch his face with the contaminated glove during the
course of the day. The intent of this provision is to have employees
wash in order to mitigate the adverse effects when skin and eye contact
does occur. At a minimum, employees are to wash their hands and faces
at the end of the shift because washing is needed to remove any
residual Cr(VI) contamination. Likewise, washing prior to eating,
drinking, smoking, chewing tobacco or gum, applying cosmetics or using
the toilet also protects against further Cr(VI) exposure.
The requirements of the final rule for washing facilities are
consistent with existing requirements for washing facilities found in
29 CFR 1910.141(d) (for general industry and shipyards) and 29 CFR
1926.51(f) (for construction). One commenter believed the requirement
for washing facilities to be "vague and subject to interpretation"
(Ex. 38-233). OSHA disagrees. The existing requirements contain
sufficient detail to guide any employer in setting up his or her
washing facilities. Washing facilities that meet the requirements of 29
CFR 1910.141(d) or 29 CFR 1926.51(f) are sufficient to meet these
requirements in this final Cr(VI) rule. In addition, both washing
facility requirements address the traditional stationary workplace and
worksites that are temporary or serviced by mobile crews. Because these
requirements already apply to workplaces covered by the Cr(VI) rule,
interpretation of a requirement for washing facilities should not be an
issue; the facilities should already be provided. Because several
comments on the proposal indicated apparent non-compliance with
existing requirements (e.g., Tr. 1241-1242, 1453-1454), the final rule
reiterates these requirements for washing facilities in order to
clarify the issue and to educate employers and provide a comprehensive
reference of requirements. In addition, the final Cr(VI) rule
supplements the general requirements for provision of washing
facilities with relatively simple, common-sense requirements that the
facilities be used when appropriate to minimize Cr(VI) exposures.
OSHA has not included a requirement for shower facilities in the
final rule. In the preamble to the proposed rule, the Agency requested
comment on the issue of whether or not provisions for showers should be
included in a final Cr(VI) standard. Some comments supported shower
requirements (Exs. 39-71; 40-10-2). NIOSH, for example, indicated a
preference for showers after anything more than limited, minor contact
with Cr(VI) (Ex. 40-10-2). Other commenters did not believe showers
were necessary (Exs. 38-267; 39-52; 39-19; 39-48; 39-40; 39-47; 38-235;
38-244; 38-220; 39-60; 38-214; 38-228; 39-20). OSHA agrees with the
latter group that a requirement for showers is not reasonably necessary
in the final Cr(VI) rule.
OSHA expects that hazardous skin and eye exposures will occur
infrequently with the proper use of appropriate protective clothing and
equipment. In these situations, the Agency believes that washing
facilities will generally be sufficient to allow employees to remove
any Cr(VI) contamination that may occur. Showers may in some situations
be an appropriate industrial hygiene control measure. Wayne Pigment
Corporation, for example, indicated that showers are currently used in
its facility (Ex. 38-204). However, OSHA does not believe that showers
are necessary in all circumstances, and has therefore not included a
requirement for showers in the final rule.
To minimize the possibility of food contamination and to reduce the
likelihood of additional exposure to Cr(VI) through inhalation or
ingestion, OSHA believes it is imperative that employees have a clean
place to eat. Where the employer chooses to allow employees to eat at
the worksite, the final rule requires the employer to ensure that
eating and drinking areas and surfaces are maintained as free as
practicable of Cr(VI). Employers also are required to assure that
employees do not enter eating or drinking areas wearing protective
clothing, unless the protective clothing is properly cleaned
beforehand. This is to further minimize the possibility of
contamination and reduce the likelihood of additional Cr(VI) exposure
from contaminated food or beverages. Employers are given discretion to
choose any method for removing surface Cr(VI) from clothing and
equipment that does not disperse the dust into the air or onto the
employee's body. For example, if a worker is wearing coveralls for
protection against Cr(VI) exposure, thorough HEPA vacuuming of the
coveralls could be performed prior to entry into a lunchroom.
The employer is not required to provide eating and drinking
facilities to employees. Employers may allow employees to consume food
or beverages on or off the worksite. However, where the employer
chooses to allow employees to consume food or beverages at a worksite
where Cr(VI) is present, OSHA intends for the employees to be protected
from Cr(VI) exposures in these areas. To this end OSHA is requiring the
employer to ensure that eating and drinking areas are as free as
practicable of Cr(VI). These provisions are consistent with the current
requirements addressing consumption of food and beverages in the
workplace found at 29 CFR 1910.141(g) and (h) (for general industry and
shipyards) and 29 CFR 1926.51(g) (for construction).
Paragraph (i)(5) (paragraph (h)(5) in the construction and shipyard
standards) specifies certain activities that are prohibited. These
activities include eating, drinking, smoking, chewing tobacco or gum,
or applying cosmetics in regulated areas, or in areas where skin or eye
contact with Cr(VI) occurs. Products associated with these activities,
such as food and beverages, cannot be carried or stored in these areas.
Because the construction and shipyard standards do not include
requirements for regulated areas, reference to regulated areas is
omitted in the regulatory text for these standards. This provision in
the final standard is necessary and appropriate to protect employees
from additional sources of exposure to Cr(VI) not necessary to job
performance.
(j) Housekeeping
The final standard includes housekeeping provisions that require
general industry employers to maintain surfaces as free as practicable
of Cr(VI), promptly clean Cr(VI) spills and leaks, use appropriate
cleaning methods, and properly dispose of Cr(VI)-contaminated waste.
These provisions are important because they minimize additional sources
of exposure that engineering controls generally are not designed to
address. Good housekeeping is a cost effective way to control employee
exposures by removing accumulated Cr(VI) that can become entrained by
physical disturbances or air currents and carried into an employee's
breathing zone, thereby increasing employee exposure. Contact with
contaminated surfaces may also result in dermal exposure to Cr(VI). The
final provisions are generally consistent with housekeeping requirements for
general industry in other OSHA standards, such as those for cadmium (29
CFR 1910.1027) and lead (29 CFR 1910.1025).
Cr(VI) deposited on ledges, equipment, floors, and other surfaces
should be removed as soon as practicable, to prevent it from becoming
airborne and to minimize the likelihood that skin contact will occur.
When Cr(VI) is released into the workplace as a result of a leak or
spill, the standard requires the employer to promptly clean up the
spill. Measures for clean-up of liquids should provide for the rapid
containment of the leak or spill to minimize potential exposures.
Clean-up procedures for dusts must not disperse the dust into the
workplace air. These work practices aid in minimizing the number of
employees exposed, as well as the extent of any potential Cr(VI)
exposure.
The standard requires that, where possible, surfaces contaminated
with Cr(VI) be cleaned by vacuuming or other methods that minimize the
likelihood of Cr(VI) exposure. OSHA believes vacuuming to be a reliable
method of cleaning surfaces on which dust accumulates, but other
effective methods may be used. These methods may include wet methods,
such as wet sweeping or use of wet scrubbers. Dry shoveling, dry
sweeping, and dry brushing are permitted only if the employer can show
that vacuuming or other methods that are usually as efficient as
vacuuming have been tried and found not to be effective under the
particular circumstances in the workplace. The standard also requires
that vacuum cleaners be equipped with HEPA filters to prevent the
dispersal of Cr(VI) into the workplace.
Paragraph (j)(2)(ii) of the final rule differs somewhat from the
proposal in that it differentiates between wet and dry cleaning
methods, indicating that dry shoveling, sweeping, and brushing can be
used only where the employer shows that HEPA-vacuuming or other methods
that minimize the likelihood of exposure to Cr(VI) had been tried and
found not to be effective. The North American Insulation Manufacturers
Association (NAIMA) requested that OSHA recognize wet sweeping as an
acceptable alternative to HEPA-filtered vacuuming (Exs. 38-228-1, p.
21; 47-30, p. 40). The Color Pigments Manufacturers Association (CPMA)
also argued that wet cleaning methods may be more efficient and produce
lower exposures than dry vacuuming (Ex. 38-205, p. 60). OSHA agrees
that wet methods can serve to minimize exposure to Cr(VI), and has
modified the language of the provision to allow wet methods to be
permitted.
The use of compressed air for cleaning is only allowed when used in
conjunction with a ventilation system designed to capture the dust
cloud created by the compressed air, or when no alternative cleaning
method is feasible. This provision is intended to prevent the dispersal
of Cr(VI) into the workplace. The United Auto Workers, International
Brotherhood of Teamsters and the Building Construction Trades
Department, AFL-CIO supported restrictions on the use of compressed air
as a means of minimizing employee exposures to Cr(VI)(Exs. 39-73-2, p.
20; 38-199-1, pp. 41, 46; 38-219-1, p.24).
An allowance for use of compressed air when no alternative method
is feasible was not included in the proposal. This provision was added
in response to arguments by NAIMA that, in some circumstances, no other
cleaning method was available. Specifically, NAIMA indicated that
during furnace rebuilds, tight spaces and hard to reach crevices can
only be effectively cleaned with compressed air (Ex. 38-228-1, p. 21).
In an active furnace area, it was contended that extreme heat limits
use of methods such as vacuuming (Tr. 1207, Ex. 47-30-1, p. 40). Other
examples were also cited (Ex. 47-30-1, p. 40).
Although OSHA agrees that in certain circumstances no alternative
to use of compressed air may be feasible, the Agency anticipates that
these circumstances will be extremely limited. The vast majority of
operations are expected to use preferred methods, such as HEPA-
vacuuming, to remove Cr(VI) contamination from workplace surfaces.
Where compressed air is used without a ventilation system designed to
capture the dust cloud created, the employer must be able to
demonstrate that no alternative cleaning method is feasible.
Cleaning equipment is to be handled in a manner that minimizes the
reentry of Cr(VI) into the workplace. For example, cleaning and
maintenance of HEPA-filtered vacuum equipment must be done carefully to
avoid exposures to Cr(VI). Filters need to be changed as appropriate
and the contents of bags disposed of properly to avoid unnecessary
Cr(VI) exposures.
The final rule requires that items contaminated with Cr(VI) and
consigned for disposal be collected and disposed of in sealed
impermeable bags or other closed impermeable containers. This provision
is intended to prevent dispersal of Cr(VI) into the air or dermal
contact with Cr(VI)-contaminated items during the disposal process.
Some commenters expressed concern about the proposed provision,
indicating that sealed, impermeable bags are impractical for large,
heavy items such as refractory brick (Tr. 1215-1216, Exs. 38-228-1, p.
22; 47-30, pp. 39-40; 47-32). OSHA intends this provision to be
performance-oriented, to allow use of any container so long as that
container prevents release of or contact with Cr(VI). Sealed barrels
could be used to serve this purpose. Other methods, such as palletizing
items and wrapping the pallet in plastic so as to create an impermeable
barrier between workers and the Cr(VI)-contaminated waste, scrap or
debris would also be acceptable.
OSHA proposed that bags or containers of waste, scrap, debris, and
other materials contaminated with Cr(VI) that are consigned for
disposal be labeled, and included specific language in paragraph (l) of
the proposed standard to be included on labels. The purpose of this
provision was to inform individuals who handle these items of the
potential hazards involved. OSHA has retained this requirement in the
final rule, but has modified the provision to require labeling in
accordance with the Agency's Hazard Communication Standard (HCS)(29 CFR
1910.1200). As discussed with regard to paragraph (l), OSHA believes
that it is critically important that employees be made aware of the
hazards associated with potential Cr(VI) exposures. By alerting
employers and employees who are involved in disposal to the potential
hazards of Cr(VI) exposure, they will be better able to implement
protective measures. However, the Agency has determined that the
information required on labels by the HCS, including the chemical
identity and appropriate hazard warnings, is sufficient to make
employees aware of potential Cr(VI) hazards. The specific language for
labels included in paragraph (l) of the proposal, and the reference to
that language in this provision, have therefore been deleted from the
final rule. Reference to the HCS has been added to ensure that
employers are aware of their obligations under the HCS for labeling of
containers containing Cr(VI) contaminated waste.
No housekeeping requirements are included in the final rule for
construction or shipyards. OSHA has determined that the housekeeping
provisions in the general industry standard are not appropriate for
these sectors because of the difficulties of complying with such
requirements in construction and shipyard environments.
OSHA's decision not to include housekeeping requirements in these
industries was supported by a number of commenters (Exs. 38-214, p. 21;
38-244, p. 13; 39-19; 39-20, p. 23; 39-60; 40-1-2, p. 33). The AFL-CIO,
on the other hand, argued that housekeeping requirements should apply
to construction and shipyard workplaces as well as those in general
industry (Ex. 47-28, p. 7). The AFL-CIO maintained that housekeeping
requirements are important measures for protecting worker health, and
noted that housekeeping requirements have been included in previous
OSHA health standards covering construction and shipyards (Ex. 47-28,
p. 7). However in the previous rulemakings that covered substantial
numbers of construction and shipyard workers, such as lead in
construction (29 CFR 1926.62) and asbestos in construction (29 CFR
1926.1101) and shipyards (29 CFR 1915.1001), OSHA did not find
housekeeping provisions to present the difficulties anticipated with
regard to Cr(VI) that are discussed below. OSHA believes these
standards address operations that are generally more amenable to
housekeeping measures. For example, the standards for asbestos in
construction and shipyards include requirements for the use of
dropcloths and barriers to prevent the migration of asbestos from many
areas where asbestos removal operations are performed. These
requirements simplify compliance with housekeeping provisions by
confining asbestos contamination in many cases to discrete and easily
identified areas. Similarly, lead operations in construction are often
enclosed to prevent environmental contamination, easing the burden of
complying with housekeeping requirements.
In previous rulemakings, the issue of excluding these industries
was not specifically raised for comment; here three pertinent questions
were included in the proposal and a record developed. In addition to
two general questions on modifications to the standards that would
better account for the workplace conditions in construction and
shipyards while still providing appropriate protection (Questions 31
and 32), the Agency specifically requested information on its
preliminary determination that housekeeping requirements would likely
be difficult to implement in construction and shipyard environments (69
FR 59310, 59311). OSHA received a number of comments in response and,
although there was not general agreement among them, sufficient
information was presented to allow OSHA to make its conclusions.
OSHA has concluded that there are compelling reasons to exclude
specific requirements for housekeeping for construction and shipyard
worksites in this final rule. In construction and shipyard settings,
operations involving Cr(VI) exposure are often of short duration,
commonly performed outdoors under variable environmental conditions,
and in locations that vary from day to day or even hour to hour within
a shift. Under these circumstances, it is often difficult to
distinguish Cr(VI)-contaminated dusts from other dirt and dusts
commonly found at the worksite (Ex. 39-19). Welding operations present
particular problems in construction and shipyards. Welding is the
predominant source of Cr(VI) exposures in these sectors (see section
VIII). Due to the small particle size of the fumes generated, welding
operations may result in the deposition of Cr(VI) over wide areas when
the welding is performed outdoors. In addition, the deposition may be
highly dependent on environmental conditions (e.g., wind direction and
speed).
These deposited fumes may not be visible to the naked eye, and they
can become intermingled with other dusts commonly found on construction
and shipyard worksites so that they are unrecognizable. Therefore, it
is unreasonable to believe that employers will be able to consistently
and accurately identify Cr(VI)-contamination at construction and
shipyard worksites, or distinguish Cr(VI)-contaminated dusts from soil
or other dusts found at the worksite. For example, if a pipe fitter
welds a section of stainless steel pipe outdoors over open ground, it
is unclear how large an area, if any, would need to be cleaned. In
addition, as noted above, construction and shipyard operations are
often of relatively short duration, and work is often performed at non-
fixed workstations or worksites. These changes in workplace conditions
add to the difficulty of complying with the specific housekeeping
requirements set forth in the final rule for general industry.
The housekeeping measures that apply to general industry are also
impractical on many construction and shipyard worksites. HEPA-filtered
vacuums would likely gather disproportionately large volumes of non-
Cr(IV) dust and debris relative to the volume of Cr(VI) captured,
particularly on open ground. This would result in the continued need to
unclog or replace filters designed for the collection of fine
particulates. Wet or dry sweeping would be unlikely to produce better
results. Disposal of waste, scrap, and debris would be subject to
similar difficulties. For these reasons, OSHA has concluded that
housekeeping requirements are highly impracticable for control of
Cr(VI) exposures in construction and shipyard workplaces and therefore
has not included housekeeping requirements for these industry sectors.
Several commenters expressed the view that many activities in
general industry workplaces are similar to those in construction and
shipyard workplaces, and therefore these activities, or general
industry as a whole, should not be subject to housekeeping requirements
either (Exs. 38-203; 39-47; 39-51, p. 15; 39-56; 40-1-2). Some argued
that housekeeping requirements are inappropriate for welding and
cutting operations (Exs. 38-203; 38-254; 39-47; 39-48; 39-56, 40-1-2).
Some commenters claimed that regardless of whether welding is performed
in construction or general industry, the quantity of settled fume is
insignificant and difficult to identify for housekeeping purposes (Ex.
38-203; 38-254; 39-47; 39-48; 39-56, 40-1-2). Others claimed that steel
mills, rolling mills, and forging operations generate substantial
amounts of dusts that do not contain Cr(VI) (Ex. 38-233, p. 40). These
employers argued that they could not comply with housekeeping
requirements because they would be unable to identify Cr(VI)-
contaminated dusts or keep the facility entirely dust-free (Ex. 38-233,
p. 41). Edison Electric Institute (EEI) alleged that coal-burning power
plants would face similar difficulties with fly ash (Tr. 436, Ex. 40-1-
2, pp. 15-16). ORC Worldwide noted that many general industry work
operations take place in dusty outdoor environments (Ex. 39-51, p. 15).
OSHA has concluded that the housekeeping requirements of the final
rule for general industry are reasonable and appropriate. A large
proportion of the workers covered by the general industry standard are
exposed in operations other than welding. In these operations, Cr(VI)
contamination is generally more easily identified, and housekeeping
measures are more practical and effective. Moreover, in general
industry, welding operations are usually performed in controlled
environments where Cr(VI) contamination can be identified and cleaned
up consistent with the requirements of the housekeeping provisions.
The Agency recognizes that in some cases general industry work
operations and work environments may be comparable to those found in
construction and shipyards. However, certain work conditions and factors
commonly present in construction and shipyard environments differ from
those typically found in general industry. Construction and shipyard
tasks are often relatively short in duration; operations are commonly
performed outdoors, sometimes under adverse environmental conditions
(e.g., wind, rain); and work is often performed at non-fixed workstations
or work sites (Exs. 39-19; 39-60; 38-214). Collectively, these factors
make compliance with the specific housekeeping requirements of the final
rule impractical for typical construction and shipyard operations. OSHA
has thus made a finding, based on the rulemaking record, that for the
majority of construction and shipyard settings, compliance with housekeeping
provisions is impracticable. In contrast, OSHA believes that compliance with
these housekeeping requirements usually does not involve the same practical
difficulties in general industry operations. For the reasons discussed
above, OSHA has determined that it is appropriate to include
housekeeping requirements in the final rule for general industry.
Moreover, paragraph (j)(1)(i) of the final rule only requires surfaces
to be maintained free of the accumulation of Cr(VI) "as practicable".
Thus, the final rule gives sufficient flexibility for the few general
industry situations where the housekeeping provisions are particularly
difficult to implement.
Also, construction and shipyard employers will still need to comply
with the general housekeeping requirements found at 29 CFR 1926.25 (for
construction) for 29 CFR 1915.91 (for shipyards). These standards
include general provision for keeping workplaces clear of debris, but
do not contain the more specific requirements found in the Cr(VI)
standard for general industry (e.g., the obligation to use preferred
cleaning methods).
EEI also cited the Administrative Law Judge (ALJ) decision in
Cincinnati Gas & Elec. Co. Beckjord Station, 2002 CCH OSHD P32,622 (No.
01-711)(ALJ), aff'd on other grounds, 21 BNA OSHC 1057 (2005), that
"the general industry housekeeping standard, 29 CFR 1910.22(a), does
not apply to coal-fired power plants' (Ex. 39-52, p. 13). This is not
correct. The ALJ did not hold that the general housekeeping standard,
29 CFR 1910.22(a), categorically does not apply to coal-fired power
plants; rather, the ALJ found that the Secretary could not cite an
employer under the housekeeping standard at 1910.22 for an explosion
hazard caused by the accumulation of combustible coal dust because this
type of explosion hazard is specifically addressed by 1910.269(v)(11)
of the Electric Power Generation, Transmission, and Distribution
standard. In affirming the decision for different reasons, the
Occupational Safety and Health Review Commission would not " * * *
exclude the possibility that the Secretary could make * * * a showing"
that the general housekeeping standard would not be preempted even with
respect to an explosion hazard by virtue of that standard providing
meaningful protection beyond that afforded by the specific standard.
The Commission concluded, however, that the record before it was not
sufficient to make such a finding. Cincinnati Gas & Elec. Co., 21 BNA
OSHC 1057, 1058 (No.01-0711, 2005). Regardless, the housekeeping
requirements in this section do not protect against explosion hazards;
they protect workers from exposure to a toxic chemical and known
carcinogen and therefore would not be preempted by 1910.269(v)(11).
EEI also claimed that the proposed housekeeping requirements
conflict with the requirements under 1910.269(v)(11) of the Electric
Power Generation, Transmission, and Distribution standard (Ex. 39-52,
p. 22). OSHA does not foresee such a conflict because an employer can
comply with both standards. Section 1910.269(v)(11) requires
controlling ignition sources to abate the explosion hazard, which does
not conflict with the housekeeping provisions of this section that
require all surfaces to be kept as free as practicable from
accumulation of Cr(VI). The housekeeping provisions of this section are
intended to minimize worker exposure to Cr(VI), and nothing suggests
that controlling ignition sources would limit exposures. Thus, the
housekeeping provisions in this standard are necessary to protect
workers.
EEI also believed that housekeeping requirements would conflict
with OSHA's standard addressing occupational exposure to inorganic
arsenic, 29 CFR 1910.1018 (Exs. 39-52, p. 22; 47-25, p. 10). OSHA does
not foresee a conflict between the housekeeping provisions of this rule
and those of the arsenic rule. When housekeeping is performed in
environments where provisions of both standards apply, the employer may
choose methods that comply with both requirements. For example, the
arsenic standard prohibits use of compressed air for cleaning, while
this rule allows use of compressed air for cleaning in extremely
limited circumstances; the arsenic rule does not require HEPA filters
on vacuums used for cleaning, while this rule does. Where both
standards apply, the employer could comply by avoiding the use of
compressed air for cleaning and using HEPA-filtered vacuums.
(k) Medical Surveillance
Paragraph (k) of the final standard (paragraph (i) for construction
and shipyards) sets forth requirements for the provision of medical
surveillance for employees in general industry, construction and
shipyards. This paragraph specifies which employees are to be offered
medical surveillance and at what times. It also specifies the content
of required examinations and material to be provided to and obtained
from the licensed health care professional administering the program.
The purpose of medical surveillance for Cr(VI) is, where reasonably
possible, to determine if an individual can be exposed to the Cr(VI)
present in his or her workplace without experiencing adverse health
effects; to identify Cr(VI)-related adverse health effects so that
appropriate intervention measures can be taken; and to determine the
employee's fitness to use personal protective equipment such as
respirators. This final standard is consistent with Section 6(b)(7) of
the OSH Act which requires that, where appropriate, medical
surveillance programs be included in OSHA health standards to aid in
determining whether the health of workers is adversely affected by
exposure to toxic substances. Almost all other OSHA health standards
have also included medical surveillance requirements.
The final standard requires that each employer covered by this rule
make medical surveillance available at no cost, and at a reasonable
time and place, for all employees meeting the requirements of this
paragraph. As in previous OSHA standards, this final standard is
intended to encourage participation by requiring that medical
examinations be provided by the employer without cost to employees
(also required by section 6(b)(7) of the Act), and at a reasonable time
and place. If participation requires travel away from the worksite, the
employer would be required to bear the cost. Employees would have to be
paid for time spent taking medical examinations, including travel time.
Some commenters questioned the utility of medical surveillance at
construction worksites and recommended that medical surveillance not be
required in the final Cr(VI) standard covering construction. For
example, several commenters representing construction employers noted a
number of particular difficulties in providing medical surveillance on
construction work sites such as the frequent movement of construction
workers from job-to-job and from one employer to another and the difficulty
in finding health care professionals familiar with signs and symptoms of
Cr(VI) exposure (e.g., Exs. 38-236; 38-244; 39-36; and 39-65). More
specifically, the Associated Builders and Contractors (ABC) testified
that "no rationale exists showing such surveillance would likely show
causation or would be feasible" (Ex. 39-65), adding that it was not
possible to demonstrate a cause and effect through exposure monitoring
and medical surveillance (Tr. 1272-1277). Such impracticalities, they
imply, would render medical surveillance in construction settings of
little utility since one would not be able to determine if an exposure
at a particular job site was responsible for the observed signs or
symptoms.
OSHA continues to believe that despite the challenges posed by the
changing nature of work and the mobility of construction workers,
medical surveillance in construction settings serves an important role
just as it does in general industry and shipyard settings. OSHA has
included medical surveillance in other OSHA health standards where
construction has been a primary industry impacted by those rules (e.g.,
lead, asbestos and cadmium) and finds no reason why the Cr(VI) final
standard should be an exception. OSHA disagrees that it will be
difficult to find health care professionals with expertise in Cr(VI)
toxicity. The major effects associated with Cr(VI) exposures include
common ailments such as asthma and dermatitis that would not require
any exceptional expertise in Cr(VI) per se. OSHA believes that it is
important for health care professionals to be familiar with an
employee's work duties and Cr(VI) exposures in order to aid them in
addressing any reported signs or symptoms, and as discussed below
requires important occupational information to be provided to the
selected health care professional. As to ABC's concern about showing
causality, OSHA does not believe that the inability to link a specific
exposure to an individual worker's particular outcome is sufficient
cause not to provide medical surveillance. Cr(VI) exposure, as
discussed previously in the health effects section of this preamble,
may cause non-malignant respiratory effects such as asthma, nasal
ulcerations and perforations, as well as allergic and irritant contact
dermatitis. The fact that an employer may not be able to identify the
specific exposure that caused a particular observed effect does not
negate the value of identifying such effects and making sure that the
affected employee gets the proper medical attention. Moreover, by
questioning the affected employee about his or her work practices and
likely exposures, it may be possible to identify lapses in the
employer's exposure control measures or the employee's work practices
that contributed to the observed effect. Such information will help to
prevent future adverse events for this employee as well as other
employees at the worksite or perhaps even other construction job sites
that have similar types of exposures and operations.
In the proposed standard, OSHA specified that medical surveillance
be provided to those employees who are experiencing signs or symptoms
of the adverse health effects associated with Cr(VI) exposure, or who
are exposed in an emergency. In addition, OSHA proposed that general
industry (but not construction or shipyard) employers be required to
provide medical surveillance for all employees exposed to Cr(VI) at or
above the PEL for 30 or more days a year.
OSHA received a variety of comments regarding the proposed triggers
for determining which employees should be provided medical
surveillance. Some commenters did not support the use of signs and
symptoms to trigger medical surveillance, stating that OSHA had not
provided any definition for what it meant by signs and symptoms and
that symptoms associated with adverse Cr(VI) health effects such as
asthma and dermatitis could also be caused by various other workplace
chemicals, allergies, or sources outside the work environment (e.g.,
Tr. 985-988; Exs. 38-124; 38-205; 47-16; 39-65). In particular, the
Color Pigment Manufacturers Association (CPMA) voiced concern that
employees could simply assert that a symptom had occurred and the
employer, who has no medical expertise to determine if symptoms are a
result of Cr(VI) exposure, would have no choice but to incur the cost
of the medical examination even though that symptom may not have been
the result of a workplace exposure (Ex. 38-205, p. 64). Another
commenter suggested that OSHA use a narrow definition of adverse heath
effects to avoid difficulties with commonplace health effects unrelated
to Cr(VI) exposure (Ex. 39-20).
Others supported the use of signs and symptoms to trigger medical
surveillance (e.g., Exs. 39-20; 38-220; 39-51; 39-71; 39-19; 39-48; 47-
26) but some objected to the sole use of signs and symptoms to trigger
medical surveillance in construction and shipyard settings and felt
that the same triggers required in general industry should be applied
to construction and shipyard settings (e.g., Exs. 38-199; 38-220; 39-
51; 38-219; 40-10-2). Organization Resource Counselors noted that many
workers are reluctant to report medical problems for a variety of
reasons and if medical surveillance is solely dependent on workers
reporting signs and symptoms to their employers, cases may go
undetected until it is too late to take effective action (Ex. 39-51).
NIOSH agreed and voiced concern that shifting the sole responsibility
of medical surveillance to employees to report signs and symptoms of
worker exposure, as they believed the proposal did, was a departure
from long-established public health practice (Tr. 300-301; Ex. 40-10-
2).
While supporting the need to include an airborne exposure trigger
for routine medical surveillance, many commenters did not support
OSHA's use of the PEL as the airborne trigger and argued that OSHA
should use the action level as it has in most of its past health
standards (e.g., Tr. 1117-1118; Exs. 39-73; 39-71; 47-26; 47-23; 40-18-
1; 38-199). NIOSH and the United Auto Workers (UAW) reasoned that given
the remaining significant risk at the PEL, the action level would be a
more appropriate trigger for medical surveillance (Exs. 40-10-2; 39-
73). The UAW also recommended that OSHA remove from the medical
surveillance provisions the 30 day exemption for exposures above the
PEL, arguing that exposures of fewer than 30 days could contribute to
kidney toxicity. Others advocated task-based or hazard assessment-based
approaches, either in conjunction with other triggers or alone, for
determining when employees should be offered medical surveillance
(e.g., Tr. 1442-1443; Exs. 38-199; 38-214; 40-10-2; 38-220). Such task-
based or hazard-assessment approaches could be used, they argued, to
identify high exposure or high risk operations where medical
surveillance might be useful.
Several groups supported triggering medical surveillance after
emergencies (e.g., Exs. 40-10-2; 38-233; 38-219) while some questioned
the value of offering medical surveillance after an emergency event
given that a substance such as Cr(VI) presents chronic hazards (Exs.
39-19, 39-47, 40-1-2). Finally, while some groups were supportive of
OSHA's proposal not to include eye and skin contact as a trigger for
medical surveillance (Exs. 39-72-1, 38-233), NIOSH recommended that OSHA
consider a dermal exposure trigger such as the one OSHA used for its
final standard for methylenedianiline, where medical surveillance was
triggered after dermal exposures of 15 days or more.
OSHA continues to believe, despite the comments offered, that the
observation of signs and symptoms known to be caused by Cr(VI) exposure
serves as a valuable complement to the use of airborne exposure
triggers as a mechanism for initiating medical surveillance. Some
employees may exhibit signs and symptoms of the adverse health effects
associated with Cr(VI) exposure even when not exposed above a specified
air limit for 30 or more days per year. These employees could be
especially sensitive, may have been unknowingly exposed, or may have
been exposed to greater amounts than the exposure assessment suggests.
Therefore in the final rule OSHA has required that employees who
experience signs or symptoms of the adverse health effects associated
with Cr(VI) exposure be included in medical surveillance. OSHA
recognizes that signs and symptoms associated with adverse health
effects such as dermatitis, asthma, and skin ulcerations may be non-
specific (i.e., they may be caused by factors other than Cr(IV)).
However, it is important to realize the context in which signs and
symptoms are expected to be used in medical surveillance. Signs and
symptoms are generally expected to be self-reported by employees and as
such are not intended to serve as a means for diagnosing adverse health
effects or determining their causality. Rather, they serve as a useful
signal that an employee may be suffering from a Cr(VI) exposure-related
health effect or are at the beginning stages of suffering a Cr(VI)-
related adverse health effect. Once these signals are recognized, the
employee can be referred to a PLHCP who can, with sufficient
information about the employee's duties, potential exposures, and
medical and work histories (as required by this standard and discussed
later), make determinations about the Cr(VI)'related effects, provide
medical treatment and recommend work restrictions where necessary. OSHA
believes that employees can be trained, through the required hazard
communication training, to identify signs and symptoms consistent with
Cr(VI) toxicity such as blistering lesions, redness or itchiness of the
skin's exposed areas, shortness of breath and wheezing that worsens at
work, nose bleeds, and whistling during inspiration or expiration.
Viewed in this context, OSHA believes that the inclusion of signs and
symptoms is an important part of the overall medical surveillance
program. Thus, the final standard would protect employees exposed to
Cr(VI) in unusual circumstances even if they don't meet the other
criteria for routine medical surveillance. OSHA acknowledges CPMA's
concern that an employee can simply assert a symptom has occurred and
the employer would be forced to provide medical surveillance and bear
the cost. However, OSHA believes that the overriding concern should be
that appropriate medical attention be provided for workers experiencing
signs and symptoms of effects known to be caused by Cr(VI). By properly
training employees about the signs and symptoms associated with Cr(VI)
and providing appropriate work-related exposure information to the
PHLCP, Cr(VI) work-related health effects can be distinguished from
other non-occupational effects. Once identified as occupationally-
related, many of these outcomes are likely to be subject to state
worker compensation benefits and defray the employer's costs of
providing medical surveillance. Under such a system, OSHA believes
employees will be unlikely to abuse medical surveillance. Nevertheless,
even the possibility that a few bad actors may act irresponsibly should
not be reason to deny worker protection where it is appropriate to
evaluate the employee's condition to determine if exposure to Cr(VI) is
the cause of the condition, and to determine if protective measures are
necessary. In addition, the Agency has found in past rulemakings that
employees generally do not unnecessarily avail themselves of medical
surveillance.
OSHA proposed that in construction and shipyard settings that signs
and symptoms and exposure in emergencies be the sole criteria for
determining which employees to provide with medical surveillance. In
the proposal, only general industry employers were required to use an
airborne trigger for initiating medical surveillance. OSHA is convinced
by comments submitted to the record that it is important that the
triggers for medical surveillance for all industries be the same.
Specifically, OSHA agrees with NIOSH and ORC that having medical
surveillance triggered only by signs and symptoms may miss important
opportunities for detecting adverse effects that may go undetected by
employees. For those reasons, OSHA believes it is appropriate to make
the triggers and the medical surveillance provisions identical across
the general industry, construction and shipyard standards. Even in
situations where the performance-oriented option for exposure
determination is used, OSHA believes that employers using historical or
objective data to characterize airborne exposures will be able to
effectively use that data to determine when to provide routine medical
surveillance.
OSHA had originally proposed that the PEL be used to trigger
medical surveillance. However, based on the comments received on this
issue and the fact that the action level is now higher than the
proposed PEL, OSHA agrees with those urging the action level be used to
trigger medical surveillance. Given the remaining risk at the final
PEL, it is more appropriate to use the action level as the trigger
rather than the PEL. However, OSHA continues to believe that having a
30 day exposure requirement in conjunction with the action level is a
reasonable approach for determining which employees to provide with
medical surveillance. OSHA agrees with the UAW that Cr(VI) metabolizes
differently than cadmium but notes that OSHA has included a similar 30
day exemption for other regulated substances that have different
metabolic half-lives compared to cadmium (e.g., methlyene chloride,
1,3-butadiene, ethylene oxide). OSHA disagrees with the UAW that Cr(VI)
presents a kidney toxicity risk that necessitates medical surveillance
for exposures less than 30 days above the action level. As discussed in
the health effects section of this preamble, OSHA does not believe that
the available scientific studies show a strong correlation between
kidney dysfunction and Cr(VI) exposure. OSHA thus continues to believe
the 30 day trigger is a reasonable benchmark to apply to Cr(VI) for
focusing the provision of medical surveillance to capture effects that
may be strongly influenced by repeated exposure. In cases where adverse
effects occur among workers exposed less than 30 days over the action
level, OSHA believes that these effects will generally present
themselves as signs or symptoms that employees can be trained to
observe and report. Such instances, as discussed above, are covered by
this final rule.
While some commenters recommended that OSHA require a task-based or
hazard-based approach for determining when to provide routine medical
surveillance, OSHA believes that a trigger, based both on the action
level and the number of days an employee is exposed to Cr(VI), is a
reasonable and administratively convenient basis for providing medical
surveillance benefits to Cr(VI)-exposed workers. In addition, it is
consistent with previous OSHA standards. This final standard would not
prohibit employers from augmenting their medical surveillance programs
to include hazard or risk-based approaches where they feel it is
helpful to identify employees who may benefit from medical
surveillance. OSHA always encourages employers to go beyond the minimum
requirements set forth in OSHA standards.
OSHA disagrees with commenters who question the value of requiring
medical surveillance shortly after an emergency has occurred (Exs. 39-
19; 39-47; 40-1-2). While there are chronic effects associated with
Cr(VI) exposure, there are also short term effects such as skin
ulcerations and dermatitis that might result from high exposures
occurring during an emergency. Emergency situations (as defined in the
standard) involve uncontrolled releases of Cr(VI), and OSHA believes
the high exposures that may occur in these situations justify a
requirement for medical surveillance. Thus, OSHA has made a final
determination that medical surveillance must be made available to
employees exposed in an emergency regardless of the airborne
concentrations of Cr(VI) normally found in the workplace. This
requirement for medical examinations after exposure in an emergency in
the final rule is consistent with the provisions of several other OSHA
health standards, including the standards for methylenedianiline (29
CFR 1910.1050), 1,3-butadiene (29 CFR 1910.1051), and methylene
chloride (29 CFR 1910.1052).
OSHA has also made a final determination not to include eye or skin
contact as a basis for medical surveillance. NIOSH suggested that OSHA
use a trigger similar to the one the Agency used in its standard on
methylenedianiline (MDA; 29 CFR 1910.1050). However, it is important to
note that, as discussed in the preamble for the final MDA standard, MDA
is readily absorbed through the skin and contributes to the dose
causing systemic effects from MDA (57 FR 35630, 8/10/92). The Agency
estimated in the final MDA risk assessment that "a 20 fold increase in
risk could be prevented by not allowing dermal exposure to MDA" (57 FR
at 35648). Therefore, using a dermal component to trigger medical
surveillance for MDA was deemed appropriate. This is not the case,
however, for Cr(VI) which is not absorbed into the body but rather
causes its effects by surface contact. Thus, OSHA believes that the MDA
standard does not serve as a useful model for a dermal trigger for
medical surveillance and is not appropriate in the final Cr(VI)
standard. In addition, in previous OSHA standards where the substance
being addressed also caused dermal irritation or sensitization (e.g.,
formaldehyde; 29 CFR 1910.1048 and methylene chloride; 29 CFR
1910.1052), OSHA did not use skin or eye contact in itself with the
substance to trigger medical surveillance. OSHA believes that
compliance with the provisions for protective work clothing and
equipment, hygiene areas and practices, and other protective measures
will minimize the potential for adverse eye and skin effects. When such
health effects occur, OSHA believes that trained employees will be able
to detect these conditions, report them to their employer, and obtain
medical assistance. In such situations, affected employees would be
provided medical surveillance on the basis that they are experiencing
signs or symptoms of Cr(VI)-related health effects.
The required medical surveillance must be performed by or under the
supervision of a physician or other licensed health care professional
(PLHCP). The Agency considers it appropriate to permit any health care
professional to perform medical examinations and procedures provided
under the standard when they are allowed by state law to do so. This
provision provides flexibility to the employer, and reduces cost and
compliance burdens. This requirement is consistent with the approach of
other recent OSHA standards, such as those for methylene chloride (29
CFR 1910.1052), bloodborne pathogens (29 CFR 1910.1030), and
respiratory protection (29 CFR 1910.134). OSHA received comments from
3M that asked the Agency to broaden its application of this provision
to allow a PLHCP who is licensed in one state to be able to provide
medical surveillance in other states where the employer has employees
covered by the rule (Ex. 47-36). As discussed in detail previously in
this summary and explanation section on paragraph (b) definitions, OSHA
has made a final determination not to broaden the definition of a
PHLCP. OSHA continues to believe that issues regarding a PHCLP's scope
of legal practice reside most appropriately with state licensing
boards.
In the proposed standard, OSHA also specified how frequently
medical examinations were to be offered to those employees covered by
the medical surveillance program. OSHA proposed that all employers be
required to provide all covered employees with medical examinations
whenever an employee shows signs or symptoms of Cr(VI) exposure; within
30 days after an emergency resulting in an uncontrolled release of
Cr(VI); and within 30 days after a PLHCP's written medical opinion
recommends an additional examination. In addition, employers in general
industry were to provide covered employees with examinations within 30
days after initial assignment unless the employee has received a
medical examination provided in accordance with the standard within the
past 12 months; annually; and at the termination of employment, unless
an examination has been given less than six months prior to the date of
termination.
OSHA received few comments on the frequency of medical exams. Those
offering comment focused on OSHA's proposed provision for annual
medical exams. Some commenters reported that general medical
surveillance programs were already being offered annually by some
employers (Exs. 38-204; 39-71) implying that an annual requirement for
Cr(VI) medical exams might not be that burdensome. NIOSH supported
OSHA's general approach towards annual medical surveillance but also
recommended that certain tests be done at earlier stages after an
initial baseline assessment (e.g., 3 months after an initial assessment
for a spirometric test, 3 to 6 months after initial assessment for a
chest X-ray) (Ex. 40-10-2). As discussed above, some commenters
expressed concern with the requirement to provide exams within 30 days
after an emergency (Exs. 39-19; 39-47; 40-1-2) and after employees
report signs or symptoms (e.g., Exs. 38-124; 38-205; 47-16; 39-65).
Having received no comments to the contrary, OSHA is maintaining
its requirement for an initial medical exam within 30 days of
assignment to a job with Cr(VI) exposure. The requirement that a
medical examination be offered at the time of initial assignment is
intended to achieve the objective of determining if an individual will
be able to work in the job involving Cr(VI) exposure without adverse
effects. It also serves the useful function of establishing a health
baseline for future reference. Where an examination that complies with
the requirements of the standard has been provided in the past 12
months, that previous examination would serve these purposes, and an
additional examination would not be needed. In keeping with its final
decision to have the triggers for providing medical surveillance
consistent across general industry, construction and shipyard settings,
OSHA is also expanding the requirement for initial medical exams to
construction and shipyard settings.
Similarly, OSHA has made a final determination to expand the
requirement for annual medical exams to construction and shipyard
settings. OSHA believes that the provision of medical surveillance on
an annual basis is an appropriate frequency for screening employees for
Cr(VI)-related diseases. The main goal of periodic medical surveillance
for workers is to detect adverse health effects at an early and
potentially reversible stage. The requirement for annual examinations
is consistent with other OSHA health standards, including those for
cadmium (29 CFR 1910.1027), formaldehyde (29 CFR 1910.1048), and
methylene chloride (29 CFR 1910.1052). Based on the Agency's
experience, OSHA believes that annual medical surveillance would strike
a reasonable balance between the need to diagnose health effects at an
early stage, and the limited number of cases likely to be identified
through surveillance.
Although NIOSH suggested that there are other more frequent
intervals where tests such as spirometric examinations or X-rays might
be useful, OSHA believes that the final Cr(VI) standard's requirement
for employers to provide additional tests when recommended by the PLHCP
is sufficient to address situations where additional procedures might
be useful. OSHA continues to believe that a PLHCP is in the best
position to recommend more frequent evaluations in order to follow
developments in a worker's condition, or to allow for specialized
evaluation. Therefore, OSHA is maintaining in the final standard, the
requirement for the provision of medical examinations within 30 days
after a PLHCP recommends additional testing.
OSHA is also retaining its requirements for medical examinations
within 30 days after an emergency and whenever an employee shows signs
or symptoms of the adverse health effects associated with Cr(VI)
exposure. As discussed earlier in this section, OSHA believes that
despite the non-specificity of some signs and symptoms associated with
Cr(VI)-related effects, it is important to provide an opportunity for
evaluation by a PHLCP after an employee reports signs or symptoms. The
PHLCP can, with work and medical history information, make
determinations as to whether an employee's reported signs and symptoms
are associated with Cr(VI) exposure and recommend appropriate remedies.
Also as discussed previously, OSHA believes that medical examinations
after an emergency also serve an important role because of the nature
of exposures likely to occur in an emergency event and thus retains
this provision in the final standard.
Similar to OSHA's final determination to expand initial and annual
medical examinations to construction and shipyard settings, OSHA is
also extending the requirement for medical examination at the
termination of employment to these sectors. The requirement that the
employer offer a medical examination at the termination of employment
is intended to assure that no employee terminates employment while
carrying an active, but undiagnosed, disease. In situations where a
previous examination, meeting the requirements of paragraph (k),
(paragraph (i) for construction and shipyards) had been provided with 6
months prior to termination, that previous examination would suffice
for this purpose.
In the proposed standard, OSHA specified that the examination to be
provided by the PLHCP was to consist of a medical and work history; a
physical examination of the skin and respiratory tract; and any
additional tests considered appropriate by the PLHCP. Special emphasis
was to be placed on the portions of the medical and work history
focusing on Cr(VI) exposure, health effects associated with Cr(VI)
exposure, and smoking. OSHA did not indicate specific tests that must
be included in the medical examination. This was based on the Agency's
belief that there were not any particular tests generally applicable to
all employees covered by the medical surveillance requirements.
Instead, the proposal required that determinations about the need for
any additional tests be left to the discretion of the PLHCP.
While some commenters agreed that specific tests such as urine
testing should not be included in the content of the required medical
exam (Tr. 2330, Exs. 40-10-2; 38-220; 38-228; 38-235), others
recommended that OSHA include spirometric evaluations, X-rays, and
helical computerized tomography (CT) scans. For example, NIOSH
recommended the addition of baseline and periodic spirometry and
baseline chest X-rays, stating that these are commonly recommended by
various occupational health organizations such as the American Thoracic
Society and the American College of Occupational and Environmental
Medicine and can be useful tools to exclude preexisting abnormalities
when subsequent evaluations are conducted (Tr. 355-360, Ex. 40-10-2)
The AFL-CIO and PACE recommended that OSHA consider adding a
requirement for helical (CT) scans for the purpose of early lung cancer
detection (Tr. 2309, 2317-2333, 2376-2381; Exs. 8-222; 39-71; 44-41.).
Such tests, they stated, have been shown to effectively find early
stage lung cancer that has been curable through surgical intervention.
While PACE acknowledged that the helical CT scan is not yet accepted
medical practice and should be contingent upon employee informed
consent, they argued that the test can be used for high risk factors
based on the results of lung function tests and chest X-rays. Others,
however, supported OSHA's proposal that such tests be provided only
when a licensed health care professional recommends that certain
additional medical tests are necessary. (Exs. 38-203; 38-228; 39-47;
39-56; 39-60). CPMA cautioned that in the "current malpractice
environment", a requirement for any additional examination deemed
necessary by the PLHCP would result in licensed health care
professionals ordering a battery of tests in order to prevent the
possibility of malpractice claims, and the employer would be required
to pay for them (Ex. 38-205).
OSHA acknowledges the value of many of the tests suggested by the
various groups commenting on this issue. However, OSHA continues to
believe that it is more effective to allow the PLHCP the flexibility to
determine when such specific tests might be most useful rather than
requiring them for all employees in the medical surveillance program on
a routine basis. With the basic information gained from the required
medical histories, work histories and a physical examination focusing
on the skin and respiratory tract (the two main targets for Cr(VI)
toxicity), the PLHCPs can use their medical expertise to best determine
what, if any, additional testing is appropriate for any individual
employee. This is especially true for tests such as the helical CT
scan, which although promising, has not been generally proven to be
appropriate on a routine basis. As pointed out by PACE, the helical CT
can be effectively used after identifying high-risk factors. For these
reasons, the final standard does not include any specific tests but
rather includes a physical exam focusing on the skin and respiratory
tract. The physical exam focuses on organs and systems known to be
susceptible to Cr(VI) toxicity. The information obtained will allow the
PLHCP to assess the employee's health status, identify adverse health
effects related to Cr(VI) exposures, and determine if limitations
should be placed on the employee's exposure to Cr(VI). The examining
PLHCP then has the flexibility to determine any additional tests that
might be appropriate for an individual employee.
The proposed standard required the employer to ensure the PLHCP has
a copy of the standard, and to provide a description of the affected
employee's former and current duties as they relate to Cr(VI) exposure;
the employee's former, current, and anticipated exposure level; a
description of any personal protective equipment used or to be used by
the employee, including when and for how long the employee has used
that equipment; and information from records of employment-related
medical examinations previously provided to the affected employee,
currently within the control of the employer.
OSHA received few comments regarding information to be supplied to
the PLHCP. CPMA felt that providing the required information to the
PLHCP would be burdensome and would be of little relevance to the
medical professional and OSHA should instead require that employers
only provide information as warranted by the health care professional
(Ex. 38-205). Ameren Corporation also expressed concerns about the
burden of providing results from previous examinations and suggested
that information gained from the medical and work histories required by
the Cr(VI) standard would suffice (Ex. 39-47).
OSHA disagrees. OSHA believes that making the required information
available to the PLHCP will aid in the evaluation of the employee's
health and have extreme relevance to the medical professional.
Especially in the case where the PLHCP is evaluating the signs and
symptoms of potential Cr(VI)-related health effects, information on the
employee's exposures to Cr(VI), the employee's use of personal
protective equipment and the results of previous examinations, where
possible, will provide important information that can be used in
conjunction with information gained from the required medical and work
histories, in determining whether the observed symptoms are a result of
Cr(VI) exposure. This information will also aid in the PLHCP's
evaluation of the employee's health in relation to assigned duties and
fitness to use personal protective equipment, when necessary. OSHA does
not believe that providing such information to the PLHCP would be
unduly burdensome. Much of this information is already being collected
by the employer for other reasons and therefore the employer is not
likely to have to expend additional energies in providing such
information to the PLHCP. With regard to providing the PLHCP results of
previous examinations, one commenter appears to believe that
extraordinary efforts would be necessary to locate and provide such
information to the PLHCP (Ex. 39-47). However, OSHA has made it
explicit in this provision that it is only requiring those records that
are currently within the control of the employer to be made available
to the PLHCP. Given that they are in control of the employer, this
information should not be overly burdensome to produce. For these
reasons, OSHA is retaining the proposed provisions detailing
information to be provided to the PLHCP in the final standard.
In addition to providing certain information to the PLHCP, the
proposed standard also would have required employers to obtain from the
examining PLHCP a written opinion containing the results of the medical
examination with regard to Cr(VI) exposure, the PLHCP's opinion as to
whether the employee would be placed at increased risk of material
health impairment as a result of exposure to Cr(VI), and any
recommended limitations on the employee's exposure or use of personal
protective equipment. The PLHCP would also need to state in the written
opinion that these findings were explained to the employee.
Few comments were received regarding information to be provided to
the employer by the PLHCP. The UAW argued that OSHA should prohibit the
PLHCP from revealing any information to the employer, and that the
written opinion should only go to the employee or the designated
employee representative (Ex. 39-73-2, Tr. 793-795). Ameren Corporation
objected to limiting the written opinion to only diagnoses related to
Cr(VI) exposure and argued that the PLHCP will likely be evaluating
exposure to other OSHA regulated substances such as lead, asbestos,
cadmium and arsenic and it would be burdensome to have the PLHCP write
separate opinions for each substance for any individual employee (Ex.
39-47). They suggested the following language: "The PLHCP shall not
reveal to the employer specific findings or diagnosis unrelated to
exposure to occupational contaminants".
The purpose of requiring the PLHCP to supply a written opinion to
the employer is to provide the employer with a medical basis to aid in
the determination of placement of employees and to assess the
employee's ability to use protective clothing and equipment. If OSHA
were to deny this information to the employer, as requested by the UAW,
this would diminish one of the main benefits of the medical
surveillance requirements of this standard. Employers must be aware of
this information to effectively place employees and select appropriate
protective equipment. Medical findings unrelated to Cr(VI) exposure,
however, are not necessary information for the employer. Under the
final standard, the PLHCP would not be allowed to include findings or
diagnoses which are unrelated to Cr(VI) exposure in the written opinion
provided to the employer. OSHA has included this provision to reassure
employees participating in medical surveillance that they will not be
penalized or embarrassed by the employer's obtaining information about
them not directly pertinent to Cr(VI) exposure. The employee would be
informed directly by the PLHCP of all results of his or her medical
examination, including conditions of non-occupational origin, but the
employer would only receive information necessary to make decisions
regarding employee placement and protective equipment selection
relative to Cr(VI) exposures. OSHA recognizes that some employees who
are exposed to Cr(VI) may also be exposed to other OSHA regulated
substances where a written opinion is required (e.g., exposures to lead
chromate). It is not the Agency's intent to have the PLHCP write
separate written opinions for an employee who is exposed to more than
one OSHA regulated substance. If the employer has an ongoing medical
surveillance program where a PLHCP is providing a written opinion on
other OSHA regulated substances, the PLHCP can combine the written
opinion for an individual employee for all covered substances. The
intent of this requirement is to assure that personal medical
information not necessary for making determinations about employee
placement and selection of personal protective equipment is not shared
with the employer. Sharing personal medical information unrelated to
workplace Cr(VI) exposures is prohibited by the final standard. OSHA
does not believe that it is necessary to change the language of this
requirement as suggested by Ameren Corporation to convey this message.
The employer is also required to provide a copy of the PLHCP's
written opinion to the employee within two weeks after receiving it, to
ensure that the employee has been informed of the result of the
examination in a timely manner. The employer must obtain the
written opinion within 30 days of the examination; OSHA believes this
will provide the PLHCP sufficient time to receive and consider the
results of any tests included in the examination, and allow the
employer to take any necessary protective measures in a timely manner.
The requirement that the opinion be in written form is intended to
ensure that employers and employees have the benefit of this
information.
The proposed rule did not include a provision for medical removal
protection (MRP) because OSHA made a preliminary determination that MRP
was not reasonably necessary or appropriate for Cr(VI)-related health
effects. The Supreme Court has held that OSHA does not have authority
to adopt wage and benefit guarantee provisions unless it can make a
finding that such a requirement is "related to the achievement of a
safe and healthful work environment." American Textile Mfr. Inst.,
Inc. v. Donovan, 452 U.S. 490, 538 (1981). Consistent with this
decision, OSHA has taken the position that it "must always ascertain
that MRP is needed for health reasons" before adopting provisions for
medical removal wage and benefit protection (52 FR 34460, 34557 (Sept.
11, 1987)).
The need for MRP can vary from health standard to health standard
and is dependent on the nature of the hazard, health effects, and
medical surveillance program involved, and the record evidence obtained
during each rulemaking. Although virtually every previous OSHA health
standard includes provisions for medical surveillance, OSHA has found
MRP necessary for only six of those standards. They are lead,
1910.1025; cadmium, 1910.1027; benzene, 1910.1028; formaldehyde,
1910.1048; methylenedianiline (MDA), 1910.1050; and methylene chloride,
1910.1052.
Upon consideration of this rulemaking record, relevant court
decisions, and the criteria OSHA has previously applied to determine
when MRP is necessary, OSHA is unable to find that an MRP provision is
reasonably necessary or appropriate for the Cr(VI) standard.
The purpose of the medical removal protection OSHA has included in
some health standards is to assure employees they will not suffer wage
or benefit loss if they are temporarily removed from further exposure
as a result of findings made in the course of medical surveillance, and
thereby to encourage the employees to participate in the medical
surveillance program. As discussed below, OSHA has determined not to
include MRP in the Cr(VI) standard for the principal reason that the
agency does not anticipate that a significant number of employees will
need to be temporarily removed from their jobs as a result of medical
surveillance. In addition, the Cr(VI) standard's medical surveillance
program is less dependent on employee action than the programs in some
other health standards that include MRP, such as lead and formaldehyde,
and other considerations that have led OSHA to use MRP in the past are
inapplicable in the context of Cr(VI).
Most of the comments OSHA received regarding MRP were about the
pros and cons of MRP provisions generally, and not about the specific
need, or lack thereof, for MRP in the context of the proposed Cr(VI)
standard. Some of the groups representing workers advocated the
inclusion of MRP with provisions for multiple physician review on the
basis that MRP is generally necessary to encourage worker participation
in medical surveillance programs (Tr. 793-795, 803-806, 2314-2315,
2345, Exs. 38-219-1; 39-71; 39-73-2; 40-10-2; 40-19-1; 47-28;). Some
comments came out against the need for MRP, suggesting, for example,
that MRP was unnecessary in this standard because there are few
instances in which temporary removal from Cr(VI) exposures would be
beneficial. Those commenters noted the permanent nature of the adverse
health effects of Cr(VI) exposure, such as allergic asthma, allergic
dermatitis, and lung cancer (Tr. 629, Exs. 38-220-1; 39-228-1; 39-235;
39-19; 39-47; 40-1-2).
In its proposal, OSHA preliminarily concluded that MRP appeared
unnecessary because it did not anticipate many circumstances in which
employees would be removed from their jobs under the new standard. The
Agency reasoned that an MRP provision was unnecessary because Cr(VI)-
related health effects generally fall into one of two categories:
either they are chronic conditions that temporary removal from exposure
will not improve or remedy (e.g., lung cancer, respiratory or dermal
sensitization), or they are conditions that can be addressed through
proper application of control measures and do not require removal from
exposure (e.g., irritant dermatitis). The evidence submitted during the
rulemaking has led OSHA to conclude that its preliminary reasoning was
correct and that for the reasons stated in the proposal there will be
few, if any, instances where temporary removal from Cr(VI) exposures
would improve employee health (Tr. 629, Exs. 38-220-1; 39-228-1; 39-
235; 39-19; 39-47; 40-1-2)
OSHA has declined to adopt MRP provisions in other health standards
under similar circumstances. In the final standard for Ethylene Oxide
(EtO), for example, OSHA did not include MRP provisions, concluding
that "the effects of exposure to EtO are not highly reversible, as
evidenced by the persistence of chromosomal aberrations after the
cessation of exposure, and the record contains insufficient evidence to
indicate that temporary removal would provide long-term employee health
benefits" (49 FR at 25788, 6/22/1984). Similarly, the more recent 1,3
butadiene standard, which primarily addresses irreversible effects such
as cancer, does not include MRP provisions (61 FR 56746, 11/4/96).
OSHA expects that the overall number of medical removals under the
new standard will be very low. OSHA recognizes that a small number of
employees may be removed from their jobs due to the health effects of
Cr(VI) exposure, but the health effects evidence suggests many of the
Cr(VI)-related effects are permanent and thus any such removals are
likely to be permanent, not temporary. OSHA has historically viewed MRP
as a tool for dealing with temporary removals only, as reflected in the
agency's decisions not to adopt MRP in the EtO and 1,3 butadiene
standards discussed above. Workers' compensation is the appropriate
remedy when permanent removal from exposures is required.
When the D.C. Circuit reviewed OSHA's initial decision not to
include MRP in its formaldehyde standard, it remanded the case for OSHA
to consider the appropriateness of MRP for permanently removed workers.
UAW v. Pendergrass, 878 F.2d 389, 400 (D.C. Cir. 1989). OSHA ultimately
decided to adopt an MRP provision for formaldehyde. However, the agency
did not rely on a need to protect workers permanently unable to return
to their jobs. Indeed, OSHA expressly rejected that rationale for MRP,
noting that "[t]he MRP provisions [were] not designed to cover
employees * * * determined to be permanently sensitized to
formaldehyde" (see 57 FR 22290, 22295 (May 27, 1992)).
Permanent wage and benefit protection would be extremely costly and
is far beyond the scope of the MRP programs OSHA has required. Given
that MRP provides benefits only for a temporary period, it is logical
that eligibility be limited to those who have only a temporary need for
removal. (See, e.g., 1910.1027(l)(12) (MRP benefits available for up to
a maximum of eighteen (18) months); 1910.1028(i)(9) (capping MRP
benefits at six (6) months); 1910.1052(j)(12) (MRP benefits limited to
a maximum of six (6) months)). The purpose of MRP--to alleviate fear of
economic loss--can only be fulfilled for employees who are concerned about
being removed temporarily. An employee worried that he may be permanently
removed from his job if he participates in medical surveillance is unlikely
to be persuaded by the prospect of a few months protection. In addition,
an important objective of MRP is to prevent permanent health effects
from developing by facilitating employee removal from exposure at a
point when the effects are reversible, and that objective has no
application where the effects are already permanent.
The evidence in the record does not demonstrate that affected
employees are unlikely to participate in medical surveillance absent
wage and benefit protection. In fact, given the small number of
removals anticipated under the new standard, any economic disincentive
to participate would likely be minimal. In any event, the medical
surveillance programs required under the new Cr(VI) standard are less
dependent on employee action than are the medical surveillance programs
required under some of OSHA's other health standards. For example, OSHA
adopted an MRP provision in the formaldehyde standard because that
standard "does not provide for periodic medical examinations for
employees exposed at or above the action level" and instead relies on
"the completion of annual medical questionnaires, coupled with * * *
employees' reports of signs and symptoms"--an approach completely
dependent "on a high degree of employee participation and
cooperation" (see 57 FR at 22293). Unlike under the formaldehyde
standard, Cr(VI) medical surveillance programs are not entirely
dependent on employee reports of signs and symptoms. The Cr(VI)
standard requires regular medical examinations and mandates that those
exams include an evaluation of the employee's skin and respiratory
tract. OSHA expects that independent of any subjective symptoms that
may or may not be reported by the employee, practitioners conducting
these examinations can make necessary medical findings based on the
required objective evaluations of the employee's physical condition.
In the lead standard, OSHA adopted an MRP provision in part due to
evidence that employees were "desperate * * * to avoid economic loss
no matter what the consequences to * * * [their] health" and were
therefore using chelating agents to "effect a rapid, short term
reduction in blood lead levels." (see 43 FR 54354, 54446 (Nov. 21,
1978)). In that case "[t]he success of periodic blood level biological
monitoring depend[ed] * * * on workers refraining from efforts to alter
their blood lead levels." Id. Unlike in the case of lead, OSHA is
unaware of any steps employees can take to mask and prevent the
detection of Cr(VI) related health effects. Therefore, OSHA is not
concerned about economic considerations resulting in employees
intentionally sabotaging their examinations in a way that would
undermine the success of the required medical surveillance programs.
Other reasons OSHA has cited for needing to include MRP in its
health standards are similarly inapplicable to Cr(VI). In lead, for
example, OSHA explained that the new blood lead level removal criteria
for the final lead standard were much more stringent than criteria
currently being used by industry and therefore many more temporary
removals would be expected under the new standard " thereby increasing
the utility of MRP (see 43 FR at 54445-54446). There is insufficient
evidence in the Cr(VI) rulemaking record to indicate that this would be
the case for Cr(VI). As stated above, OSHA anticipates few
circumstances where medical removal will be needed. Furthermore, there
are no criteria in the new standard that are likely to increase the
small number of medical removals that may be occurring.
Finally, one reason OSHA adopted MRP in the lead standard was
because it "anticipate[d] that MRP w[ould] hasten the pace by which
employers compl[ied] with the new lead standard" (43 FR at 54450).
OSHA reasoned that the greater the degree of noncompliance, the more
employees would suffer health effects necessitating temporary medical
removal and the more MRP costs the employer would be forced to incur.
Thus, in that case OSHA thought that MRP would serve as an economic
stimulus for employers to protect workers by complying with the
standard. With respect to Cr(VI), however, there is no evidence in the
record that employees suffering from the health effects of Cr(VI)
exposure need to be removed from their jobs now--when the PEL and
exposures are significantly higher than they will be under the new
standard; OSHA therefore has no reason to believe that so many
employees would need to be removed once the PEL is lowered that
employers' concerns about the costs of MRP would induce more rapid
compliance on the part of employers. In fact, as stated earlier, OSHA
believes that the health effects of Cr(VI) exposures will result in
only a small number of medical removals. MRP is thus unlikely to work
as a financial compliance incentive in this case.
OSHA also notes that there are two health standards that provide
limited medical removal protection under their requirements for
respiratory protection. They are asbestos, 1910.1001(g)(2)(iii); and
cotton dust, 1910.1043(f)(2)(ii). These standards require MRP when a
medical determination is made that an employee who is required to wear
a respirator is not medically able to wear the respirator and must be
transferred to a position below the PEL where respiratory protection is
not required. OSHA has determined that such a provision is unnecessary
for the Cr(VI) standard because OSHA has since promulgated a revised
respiratory protection standard that specifically deals with the
problem of employees who are medically unable to wear negative pressure
respirators (29 CFR 1910.134(e)(6)). The respirator standard addresses
the problem, not through MRP, but by requiring the employer to provide
a powered air-purifying respirator instead of a negative pressure
respirator. In the Cr(VI) standard, OSHA requires employers to comply
with the requirements of 1910.134, including medical evaluations
required under that standard. As discussed earlier in the section of
the preamble addressing respiratory protection, there was much support
for referring all aspects of respiratory protection to OSHA's revised
respiratory protection standard. OSHA sees no reason to supersede
1910.134 in the final Cr(VI) standard.
In sum, OSHA does not expect Cr(VI)-related health exposures to
result in a large number of medical removals, either temporary or
permanent, and because the record shows that any removals that do occur
are likely to be permanent, OSHA concludes that the evidence does not
support a finding that MRP is reasonably necessary or appropriate for
the final Cr(VI) standard. This decision is based on the evidence
obtained during this rulemaking, and is not intended to preclude OSHA
from adopting MRP provisions in the future when it believes that such a
provision would contribute to the well-being of employees.
(1) Communication of Hazards to Employees
Paragraph (1) of the final rule (paragraph (j) for construction and
shipyards) sets forth requirements intended to ensure that the dangers
of Cr(VI) exposure are communicated to employees in accordance with
existing requirements of OSHA's Hazard Communication standard (HCS) (
29 CFR 1910.1200).
In the proposed standard, requirements for communication of hazards
were designed to be substantively as consistent as possible with OSHA's
existing HCS in order to avoid a duplicative administrative burden on
employers who would need to comply with the requirements of both
standards. However, despite this effort, a number of commenters
expressed the view that OSHA's existing HCS requirements are
sufficient, and that hazard communication provisions in this rule are
not warranted (e.g., Exs. 38-203; 38-244; 38-254; 39-19; 39-40; 39-47;
39-48; 39-51; 39-56; 39-64; 39-72-1; 40-1-2). The Color Pigments
Manufacturers Association supported this position, adding that
additional requirements only serve to increase the complexity of an
already complex and lengthy standard (Ex. 38-205). The North American
Insulation Manufacturers Association (NAIMA) claimed that additional
requirements deprive employers of necessary discretion, conflict with
efforts to streamline and simplify hazard communication requirements,
and increase the burden on employers while providing no apparent
benefit (Exs. 38-228; 47-30). Moreover, NAIMA added that relying on the
HCS will, in time, have the added benefit of simplifying implementation
of the Globally Harmonized System of Classification Labeling of
Chemicals (GHS).
Several other commenters supported OSHA's proposed requirements for
communication of hazards (e.g., Exs. 38-199-1; 38-219-1; 40-10-2). For
example, NIOSH considered that the general requirements of the HCS are
useful for all workplace hazards, but Cr(VI)-specific requirements
provide focused and enhanced protection of workers (Ex. 40-10-2). The
Building and Construction Trades Department, AFL-CIO maintained that
the information and training requirements contained in the standard
allow employers to go to a single reference to ensure they are in
compliance, helping employers understand their obligations and
assisting compliance officers assess employer compliance (Ex. 38-219-
1).
In viewing the comments submitted to the record, it is clear that
there is widespread support for the communication of hazards to
employees. OSHA continues to believe, as stated in the proposal, that
informing employees of the hazards to which they are exposed and
associated protective measures is essential to provide employees with
the necessary understanding of the degree to which they themselves can
minimize potential health hazards. As part of an overall hazard
communication program, training serves to explain and reinforce the
information presented on labels and in material safety data sheets.
These written forms of communication will be successful and relevant
only when employees understand the information presented and are aware
of the actions to be taken to avoid or minimize exposures, thereby
reducing the possibility of experiencing adverse health effects.
However, OSHA also continues to believe that it is important for
the requirements for communicating Cr(VI) hazards to be consistent with
the requirements in its existing HCS. To better assure this
consistency, OSHA has made a final determination to remove items from
the final rule that duplicate requirements in the HCS. While certain
proposed items are not being retained in the final Cr(VI) standard, the
obligations to provide communication and training on the issues
addressed in these items are required by the HCS. Thus, their removal
does not represent a lessening in worker protection. OSHA believes such
streamlining will provide better consistency and reduce confusion
between the communication of hazards obligations under the final Cr(VI)
rule and the HCS. OSHA acknowledges the comments of the Building and
Construction Trades Department who felt that retaining these items
allows employers to go to a single reference to ensure they are in
compliance. However, since OSHA requires the HCS to be followed and has
not repeated that standard in its entirety in the Cr(VI) standard,
employers would not be able to rely solely on the Cr(VI) standard as a
single reference for complying with the HCS even if such elements were
retained. Moreover, it is a very rare workplace that has only Cr(VI)
and no other hazardous chemicals. Thus, the vast majority of employers
would have to consult the HCS anyway.
OSHA has retained the proposed provisions requiring that employees
be trained about the contents of the new Cr(VI) final rule and the
purpose and description of the medical surveillance program required
under the final Cr(VI) standard. The final standard also requires that
the employer make a copy of the standard readily available to employees
without cost. These elements are not required to be communicated by the
HCS. However, OSHA believes that it is important for employees to be
familiar with and have access to the final Cr(VI) standard and the
employer's obligations to comply with it. Specifically, with regard to
the purpose and description of the medical surveillance program, OSHA
intends that employees be trained about the signs and symptoms of
Cr(VI)-related adverse health effects. This information, in conjunction
with the training on Cr(VI) hazards required by the HCS, will help to
assure that employees are able to adequately report signs and symptoms
of Cr(VI)-related adverse health effects in order to receive medical
attention from a licensed health care professional (as required by the
medical surveillance section of the final standard and previously
discussed in the preamble).
Like the HCS, OSHA intends that the required training be
performance-oriented. The standard lists the subjects, in addition to
those that are already covered by the HCS, that must be addressed in
training, but not the specific ways that this is to be accomplished.
Hands-on training, videotapes, slide presentations, classroom
instruction, informal discussions during safety meetings, written
materials, or any combination of these methods may be appropriate. Such
performance-oriented requirements are intended to encourage employers
to tailor training to the needs of their workplaces, thereby resulting
in the most effective training program in each specific workplace.
OSHA believes that the employer is in the best position to
determine how the training can most effectively be accomplished. The
Agency has therefore laid out the objectives to be met to ensure that
employees are made aware of the hazards associated with Cr(VI) in their
workplace and how they can help to protect themselves. The specifics
regarding how this is to be achieved are left up to the employer.
The communication of hazards elements proposed, but not included
the final rule, are requirements for:
Warning signs for regulated areas;
Warning labels for Cr(VI)-contaminated work clothing and
equipment and Cr(VI) wastes and debris;
Employees to be provided training and training records;
Initial training;
Training that is understandable;
Certain topics for training; and
Additional training.
As discussed below, OSHA believes that these requirements either
duplicate or are inconsistent with requirements in the HCS and are
therefore not necessary in the final Cr(VI) standard.
Under the proposed standards, OSHA included requirements for
specific language on signs and labels (e.g.,
DANGER; CHROMIUM (VI); CANCER HAZARD; CAN DAMAGE SKIN, EYES, NASAL
PASSAGES, AND LUNGS; AUTHORIZED PERSONNEL ONLY; RESPIRATORS MAY BE
REQUIRED IN THIS AREA.) OSHA is deleting the requirement for specific
language on signs for regulated areas and on labels for containers of
contaminated clothing and equipment and containers of Cr(VI)
contaminated waste and debris consigned for disposal. By deleting these
requirements OSHA is only deleting requirements for special signage. As
discussed earlier in this preamble for paragraph (e), regulated areas,
OSHA maintains in the final Cr(VI) standard requirements that regulated
areas in general industry be demarcated but allows them to be
demarcated in any manner that adequately establishes and alerts
employees of the boundaries of the regulated area. OSHA believes that
it is not necessary to require a prescribed sign in order to adequately
demarcate a regulated area. Any manner of demarcation may suffice to
achieve this goal. Similarly, OSHA has removed the requirements for
specific language for warning labels. As discussed earlier in this
preamble for paragraph (h), protective clothing and equipment
(paragraph (g) for construction and shipyards) and paragraph (j),
housekeeping, labels are still required for containers of Cr(VI)-
contaminated work clothing and equipment and containers of Cr(VI) waste
and debris. However, instead of specific mandated signage, OSHA is only
requiring that those containers be labeled in accordance with OSHA's
HCS. OSHA believes this achieves the same primary goal while providing
flexibility for the employer. Moreover, as pointed out by the NAIMA,
prescribed language may interfere with hazard communication
harmonization under the GHS (Ex. 38-228).
In the proposed rule, OSHA required that training be provided for
all employees who are exposed to airborne Cr(VI) or who have eye or
skin contact with Cr(VI), that employers maintain a record of that
training, and that the training be provided at the time of initial
assignment to a job with potential exposure to Cr(IV). OSHA believes
that these issues are already adequately addressed by the HCS. For
example, paragraph (c) of the HCS defines employee as a worker who may
be exposed to hazardous chemicals under normal operating conditions or
in foreseeable emergencies. Such a definition would encompass those
employees who are exposed to airborne Cr(VI) or who have skin or eye
contact with Cr(VI). In addition, paragraph (e)(1) of the HCS requires
that employers develop and implement a written hazard communication
program that provides for employee training. Finally, paragraph (h)(1)
of the HCS requires that employers provide training at the time of
initial assignment.
The HCS does not require training records to be kept. OSHA finds no
evidence in this record to support requiring training records in the
final Cr(VI) standard or to justify this inconsistency with the HCS.
This issue is discussed in further detail later in this preamble under
paragraph (m), recordkeeping.
The proposed standard required that the employer provide training
that is understandable to the employee. Because the HCS requires
training to be "comprehensible" to employees (see 4/10/88 letter of
interpretation; http://www.osha.gov/pls/oshaweb/owadisp.show--
document?p--table= INTERPRETATIONS&p--id=19651), OSHA does not believe
it is necessary to include this provision in the final Cr(VI) standard.
Nevertheless, OSHA emphasizes that in order for the training to be
effective, the employer must ensure that it is provided in a manner
that the employee is able to understand. Employees have varying
educational levels, literacy, and language skills, and the training
must be presented in a language and at a level of understanding that
accounts for these differences in order to meet the requirement that
individuals being trained understand the specified elements. This may
mean, for example, providing materials, instruction, or assistance in
Spanish rather than English if the workers being trained are Spanish-
speaking and do not understand English. The employer is not required to
provide training in the employee's preferred language if the employee
understands both languages; as long as the employee is able to
understand the language used, the intent of the standard will be met.
OSHA has also removed certain elements addressing topics to be
covered under employee information and training. OSHA believes that the
HCS requires training on such items. The items removed address: the
health hazards associated with Cr(VI) exposure; the location, manner of
use and release of Cr(VI); engineering controls and work practices
associated with the employee's job assignment; the purpose, selection
and use of respirators and protective clothing; emergency procedures;
and measures employees can take to protect themselves. Paragraphs
(h)(2)(ii) and (h)(3)(ii-iii) of the HCS cover these topic areas.
Therefore, OSHA believes that removing these elements from the final
Cr(VI) standard neither removes any employer training requirements nor
diminishes worker protection.
OSHA has also removed the proposed element for training employees
on their rights to access records under 29 CFR 1910.1020(g). Such
information on employees' rights is already required to be transmitted
to employees under paragraph (g)(1) of OSHA's Access to Employee
Medical and Exposure Records standard, 29 CFR 1910.1020. Therefore,
OSHA sees no need to duplicate that requirement in the final Cr(VI)
standard.
Finally, OSHA has removed elements addressing additional training.
The proposed rule would have required that additional training be
provided when necessary to ensure that each employee maintains an
understanding of the safe use and handling of Cr(VI) and when workplace
changes result in an increase in employee exposures. While the HCS does
not have a provision requiring periodic retraining, it has been
interpreted to require that employees "must be aware of the hazards to
which they are exposed . . . and know and follow appropriate work
practice" (see OSHA Compliance Directive, CPL 2-2.38D, Inspection
Procedures for the Hazard Communication Standard) OSHA believes that
since employees are required to be aware of the hazards to which they
are exposed, this would mandate that as new exposures occur because of
changes in the workplace employees must be made aware of them.
Similarly, it would mandate additional training as necessary to
maintain employees' understanding of the safe use and handling of
Cr(VI) as this is critically linked to their awareness of hazards to
which they are exposed.
In summary, although OSHA has removed a number of items under the
communication of hazards in the final rule, the training obligations
imposed by this final standard have not meaningfully changed. OSHA has
only removed those items that are duplicative or inconsistent with the
HCS, while retaining items not covered by the HCS that the Agency
believes are necessary to ensure employees understand this final Cr(VI)
standard and thereby protect employee health.
(m) Recordkeeping
Paragraph (m) of the final rule (paragraph (k) for construction and
shipyards) requires employers to maintain exposure and medical
surveillance records. OSHA proposed a requirement for employers to
maintain records of employees' Cr(VI)-related training. This requirement
has not been included in the final rule. As indicated in the discussion of
paragraph (l) of the standard, OSHA believes that the provisions of the
Agency's Hazard Communication standard (HCS) provide appropriate and
sufficient requirements for training employees who are potentially
exposed to Cr(VI). The HCS does not require retention of training
records, and the addition of such a requirement in this rule would
involve substantial additional paperwork burdens for employers. OSHA
believes that the performance-oriented requirements of the HCS, along
with the requirements of paragraph (l) that employees be able to
demonstrate knowledge of both the Cr(VI) standard and the medical
surveillance program it requires, will be sufficient to ensure that
employees are adequately trained with regard to Cr(VI) hazards and
protective measures. The absence of a requirement for retention of
training records is also consistent with OSHA's two most recent
substance-specific health standards, addressing exposure to methylene
chloride (29 CFR 1910.1052) and 1,3 butadiene (29 CFR 1910.1051).
Relatively few comments addressed the proposed recordkeeping
requirements. However, the final rule's requirements for maintenance of
exposure records have been modified to reflect changes to paragraph (d)
of this section addressing exposure determination. Specifically,
requirements for maintaining exposure data have been added to the
construction and shipyard standards. The requirements for retention of
medical surveillance records are unchanged from the proposal.
The final recordkeeping requirements are in accordance with section
8(c) of the OSH Act, which authorizes OSHA to require employers to keep
and make available records as necessary or appropriate for the
enforcement of the Act or for developing information regarding the
causes and prevention of occupational injuries and illnesses. The
recordkeeping provisions are also consistent with OSHA's access to
employee exposure and medical records rule (29 CFR 1910.1020).
Where the employer performs air monitoring to determine employee
Cr(VI) exposures, records must be kept that identify the monitored
employee and all other employees whose exposure the monitoring
represents, and accurately reflect those exposures. The employer is
required to keep records for each exposure measurement taken.
Specifically, records must include the following information: The date
of measurement for each sample taken; the operation involving exposure
to Cr(VI) that was monitored; sampling and analytical methods used and
evidence of their accuracy; the number, duration, and results of
samples taken; the type of personal protective equipment used; and the
name, social security number, and job classification of all employees
represented by the monitoring, indicating which employees were actually
monitored.
The final rule allows employers the option of relying on historical
monitoring data or objective data to determine employee exposures to
Cr(VI) where appropriate. Historical monitoring data are Cr(VI)
monitoring results obtained prior to the effective date of the standard
that were obtained during work operations conducted under workplace
conditions closely resembling the employer's current operations.
Objective data are information such as air monitoring data from
industry-wide surveys or calculations based on the composition or
chemical and physical properties of a substance demonstrating the
employee exposure to Cr(VI) associated with a particular product or
material or a specific process, operation, or activity. Use of
historical monitoring data and objective data under this final rule is
described in greater detail in the discussion of paragraph (d) above
addressing exposure determination.
Where historical monitoring data are relied upon to meet the
exposure determination requirements of this standard, records of these
data must be maintained. The records of historical monitoring data must
demonstrate that the data were obtained using a method sufficiently
accurate to be allowed under paragraph (d)(5) of the standard. The
records must also show that the work being performed, the Cr(VI)-
containing material being handled, and the environmental conditions at
the time the historical monitoring data were obtained are the same as
those on the job for which exposure is being determined. Other data
relevant to operations, materials, processing, or employee exposures
must also be included in records.
Where objective data are used to satisfy the exposure determination
requirement, the employer must establish and maintain an accurate
record of the objective data upon which he or she relied. This record
must include: The chromium-containing material in question; the source
of the objective data; the testing protocol and results of testing, or
analysis of the material for the release of chromium (VI); a
description of the process, operation, or activity involved and how the
data support the determination; and other data relevant to the process,
operation, activity, material, or employee exposures.
Since historical monitoring data and objective data may be used to
exempt the employer from provisions of the standard or provide a basis
for selection of respirators, it is critical that this determination be
carefully documented. Reliance on historical monitoring data and
objective data is intended to provide the same degree of assurance that
employee exposures have been correctly characterized as air monitoring
would, and records must demonstrate a reasonable basis for the exposure
determination.
These records are also available to employees so that they can
examine the determination made by the employer and assure themselves
they are being protected by the employer. Moreover, compliance with the
requirement to maintain records of exposure data enables the employer
to easily show at least for the duration of the retention of records
that the exposure determination was accurate and conducted in an
appropriate manner.
In addition to records relating to employee exposures to Cr(VI),
the employer must establish and maintain an accurate medical
surveillance record for each employee subject to the medical
surveillance requirements of the standard. OSHA believes that medical
records, like exposure records, are necessary and appropriate for the
protection of employee health, the enforcement of the standard, and to
the development of information regarding the causes and prevention of
occupational illnesses. Good medical records, including the record of
the examination at termination of employment, are important to the
employee in that this information will assist the employee and his or
her PLHCP in making the best health care decisions. Medical records are
necessary for the proper evaluation of the employee's health. The
employer will benefit from knowing when his or her employees have
Cr(VI) health related problems. The employer can then act to address
workplace conditions that have been associated with Cr(VI) exposure.
Finally the records can be useful to the Agency and others in
enumerating illnesses and deaths attributable to Cr(VI), in evaluating
compliance programs, and in assessing the efficacy of the standard.
Medical surveillance records are required to include the following
information: The name, social security number, and job classification
of the employee; a copy of the PLHCP's written opinions; and a copy of the
information provided to the PLHCP. This information includes the
employee's duties as they relate to Cr(VI) exposure, Cr(VI) exposure
levels, and descriptions of personal protective equipment used by the
employee (see paragraph (k)(4) in general industry, paragraph (i)(4) in
shipyards and construction).
Several commenters expressed the view that requiring a copy of the
information provided to the PLHCP would entail creating and maintaining
an unnecessary duplicate copy of medical records (e.g., Exs. 38-203;
38-254; 39-47; 39-56). OSHA believes it is important for the employer
to maintain medical records, even if duplicate information is
maintained by the PLHCP. As mentioned previously, this information is
useful in evaluating health outcomes, and retention by the employer
ensures that complete records are available from a single source even
if different PLHCPs provide examinations.
OSHA does not intend for this provision to be interpreted to
require an employer to maintain multiple copies of records. If records
of previous medical exams are within the control of the employer, that
record is sufficient and does not need to be reproduced. For instance,
where an employer maintains a record of medical exams provided to an
employee, a duplicate record does not need to be created in order to
fulfill recordkeeping requirements for a copy of the information
provided to the PLHCP.
The final rule requires that exposure monitoring and medical
surveillance records include the employee's social security number. The
Color Pigments Manufacturers Association suggested that an employee
identification number be permitted in lieu of a social security number
(Ex. 38-205). OSHA examined alternative forms of identification in
Phase II of the Agency's Standards Improvement Project (70 FR 1112 (1/
5/05)) and did not take any action in that rulemaking concerning the
use of social security numbers, indicating that further investigation
was required.
For purposes of this rule, OSHA does not believe that alternative
forms of identification, such as employee identification numbers,
represent an acceptable alternative to social security numbers. The
Agency understands the privacy concerns raised by this requirement.
However, social security numbers have much wider application, and are
correlated to employee identity in many other types of records. Social
security numbers are therefore a more useful tool since each number is
unique to an individual for a lifetime and does not change as an
employee changes employers. This requirement is consistent with
previous OSHA substance-specific health standards.
The final rule also incorporates the requirement that employers
maintain and provide access to records in accordance with OSHA's
standard addressing access to employee exposure and medical records (29
CFR 1910.1020). The medical and exposure records standard requires that
exposure records be kept for at least 30 years and that medical records
be kept for the duration of employment plus thirty years. It is
necessary to keep these records for extended periods because of the
long latency period commonly associated with cancer. Cancer often
cannot be detected until 20 or more years after first exposure. The
extended record retention period is therefore needed because causality
of disease in employees is assisted by, and in some cases can only be
made by, having present and past exposure data as well as the results
of present and past medical examinations.
(n) Dates
Paragraph (n) of the standard (paragraph (l) for construction and
shipyards) establishes start-up dates for requirements of the standard.
OSHA has extended the effective date from that proposed and provided
more time for employers to comply with most provisions of the final
rule, based on information submitted to the record indicating that
compliance may require additional time (e.g., Exs. 39-19; 39-40; 39-47;
38-202; 38-205; 47-32; 38-233). The dates included in this final rule
are also based on the Agency's experience with other standards
concerning the amount of time required for employers to comply with
similar requirements.
The standard will become effective on May 30, 2006. This date is 90
days from the date of publication in the Federal Register. The proposed
standard had provided that the final rule would become effective 60
days after publication in the Federal Register. The extension of the
interval between the publication date and the effective date of the
standard is in response to comments indicating that some employers will
need more time to comply than the proposed rule would have allowed
(e.g., Exs. 38-214; 38-218; 38-220; 38-235; 38-254; 39-19; 39-40; 39-
47; 39-48; 39-56; 39-60; 40-1-2).
The Agency sets the effective date to allow sufficient time for
employers to obtain the standard, read and understand its requirements,
and undertake the necessary planning and preparation for compliance.
Section 6(b)(4) of the OSH Act provides that the effective date of a
standard may be delayed for up to 90 days from the date of publication
in the Federal Register. Given the concerns expressed by commenters,
OSHA's interest in having employers implement effective compliance
efforts, and the minimal effect of the additional 30 day delay, the
Agency has decided that it is appropriate to set the effective date at
90 days from publication, rather than at 60 days.
The dates for employer compliance with obligations of the final
rule have also been extended from those proposed. Special provision has
been made to account for the needs of small businesses in meeting the
requirements of the new standards. OSHA proposed a requirement that all
employers comply with provisions of the final rule (except those for
engineering controls) 90 days after the effective date. The final rule
requires employers with 20 or more employees to comply with most
requirements 180 days after the effective date. Employers with 19 or
fewer employees must comply with most requirements of the final rule
one year after the effective date. This extension is intended to allow
employers sufficient time to complete initial exposure assessments,
establish regulated areas where required, obtain appropriate protective
work clothing and equipment, and comply with other provisions of the
rule. Several commenters expressed concerns that 90 days did not allow
sufficient time for employers to come into compliance with these
provisions (e.g., Exs. 39-19; 39-40; 39-47; 39-48; 39-51; 39-56; 39-60;
40-1-2). ORC Worldwide expressed this opinion, stating:
OSHA's proposal that all obligations of the standard except the
engineering control requirement would be fulfilled within 90 days
after its effective date is not enough time for the industries that
have not determined their Cr(VI) sources and characterized their
exposures to complete those tasks and be in compliance. Many are
large companies with extensive operations, and finding all potential
Cr(VI) sources will take time. Once these sources are identified,
the task of characterizing exposures will require additional time.
OSHA should allow a start-up date that is at least six months from
the effective date (Ex. 39-51).
The Society for the Plastics Industry (SPI) concurred with the view
that 90 days was an insufficient amount of time for employers to come
into compliance with the rule, claiming in particular that employers
who do not currently have respiratory protection programs in place
will require more than 90 days to develop a respiratory protection
program, obtain respirators, conduct medical evaluations and fit
testing, and provide training. SPI advocated allowing 180 days after
the effective date before respirator use would be required (Ex. 38-
218).
The potential difficulties faced by small businesses in meeting the
requirements of the rule were also noted by SPI and others, who urged
OSHA to allow additional time for employers to comply with the
requirements of the final rule (Exs. 38-218, pp. 34-35; 38-233, pp. 33-
34). SPI stated:
* * * small employers should receive more time to meet the
requirements of the new rule when it becomes effective. Many small
employers in the plastics industry do not have the resources to
provide respirators and implement respirator programs, exposure
monitoring, training and education programs, provide other forms of
protective work clothing and PPE, install warning signs and
regulated areas, and implement medical surveillance programs all
within 90 days of the effective date of the new rule (Ex. 38-218, p.
35).
OSHA believes these concerns regarding the proposed compliance
timetable are reasonable, so the Agency is providing additional time in
order to give employers the ability to comply with these obligations.
Given the large number of small employers covered by the requirements,
and the special problems of many of those employers in identifying and
implementing appropriate control measures, OSHA has decided to permit
these employers a longer time period in which to comply with most
requirements of the standard. OSHA has chosen to specify employment of
19 or fewer employers as the threshold size for allowing additional
time for compliance under the final rule. The Agency believes this is a
reasonable threshold, and is consistent with the threshold applied for
similar requirements in the Methylene Chloride standard (29 CFR
1910.1052). OSHA believes the extended compliance times will allow
affected employers sufficient time to comply with the requirements of
the standard.
In the proposal, OSHA indicated that change rooms would be required
no later than one year after the effective date of the standard. As
explained in the discussion of paragraph (i), this standard does not
impose new requirements for change rooms beyond those found in 29 CFR
1910.141(e) (for general industry and shipyards) and 29 CFR 1926.51(i)
(for construction). Therefore, because change rooms should already be
established, no effective date is necessary and reference to change
rooms in this paragraph has been deleted to avoid potential confusion.
Feasible engineering controls must be in place within four years
after the effective date. This is to ensure that employers are provided
sufficient time to complete the process of designing, obtaining, and
installing the necessary control equipment. This represents an
extension of two years beyond that proposed for engineering controls.
Several commenters contended that substantially more time was needed to
implement engineering controls than had been proposed (e.g., Exs. 38-
202; 38-204; 38-205; 38-228-1; 38-233; 39-49; 39-51; 47-32). For
example, Engelhard Corporation indicated that OSHA had underestimated
the complexity involved in meeting the requirements of the standard,
such as testing of new equipment, obtaining building permits for
process changes, and air permit changes (Ex. 38-202). Steel industry
representatives argued that, in addition to time needed to install
adequate engineering controls, additional time should be provided for
the steel industry and other significantly affected industries to
absorb the costs associated with compliance (Ex. 38-233).
OSHA agrees that additional time may be needed to come into full
compliance with the engineering control requirements of the final rule.
In particular, the Agency is aware that in some cases employers may be
required to reevaluate modified ventilation systems for compliance with
regulations governing discharges of Cr(VI) into the environment (e.g.,
EPA's Emission Standards for Hazardous Air Pollutants (NESHAP)
regulations (40 CFR 63)). OSHA has taken into consideration the need of
many affected employers to coordinate their OSHA compliance efforts
with their other regulatory compliance obligations. The Agency believes
it appropriate to allow sufficient time for modification and
reevaluation of ventilation systems to generally be accomplished during
normal permitting cycles in order to lessen the impact of the standard.
Other employers who may also need additional time for implementing
engineering controls include employers with certain electroplating
operations and welding operations. For example, in electroplating there
are new fume suppressant technologies that can be used to reduce
airborne exposures created in electroplating baths. However, some of
these technologies have not been fully tested in the variety of
electroplating operations that exist and employers must be careful in
applying this technology for a particular operation so that the fume
suppressant does not adversely affect the quality of the item being
electroplated. Additional time for implementing such an engineering
control would allow employers to gain experience with this technology
and learn more effective ways to control exposures for their particular
plating operations.
In addition, as discussed previously in this preamble, many welders
will be able to reduce Cr(VI) exposures by switching from shielded
metal arc welding (SMAW) to gas metal arc welding (GMAW). This switch
is not a simple matter. The employer must first research conditions
where such a switch might be possible taking into account the
configuration of the areas where the welding might take place, the
substrate to be welded and the desired quality of the weld. Since
specifications for the desired weld are important, tests of the new
welding technique may be necessary to make sure those specifications
are met. Additionally, extra time is likely to be needed to buy the
necessary equipment and train the employees who will be required to
perform the new welding method. The final rule thus allows four years
from the effective date for employers to institute engineering controls
to comply with the standard. During the period in which employers are
implementing these controls, respirators may be used to comply with the
new PEL.
The extension of the compliance deadline for implementation of
engineering controls will allow those firms that need extensive
engineering controls time to adequately plan for and implement these
controls. This modification will thus help to ensure adequate
protection for workers. OSHA also believes that the extension will have
the ancillary benefit of limiting the economic impact of the rule by
allowing employers additional time to plan for and absorb the costs
associated with compliance. Based on its review of the rulemaking
record, the Agency has reached the conclusion that employers will be
able to implement engineering controls within the time frame
established in the final rule.
Appendices
OSHA did not include appendices in the proposed standard. While
some of OSHA's previous standards have included non-mandatory
appendices on topics such as the hazards associated with the regulated
substance, health screening considerations, and sampling and analytical
methods, OSHA made a preliminary determination that topics typically
included in appendices could be better addressed with guidance materials.
Various commenters supported guidance materials in conjunction with
the standard (Tr. 1307, 1308, 1309-1312, Exs. 38-214, p. 24; 38-220-1,
p. 35; 39-20, p. 26; 39-60). One commenter noted the utility of OSHA's
compliance assistance tools and preferred the accessibility of those
guidance documents and e-tools to appendices (Ex. 39-60). Others,
however, felt that including appendices as a part of the standard would
make them more directly available for review and determining actions
(Tr. 1099-1100, Exs. 38-218, p. 35; 39-19; 39-60; 40-1-2).
After consideration of these comments, OSHA has made a final
determination not to include non-mandatory appendices in the Cr(VI)
final rule. First, many of the appendices OSHA has included in the past
such as sampling and analytical methods and respiratory protection fit-
testing procedures are already readily available. For example, fit-
testing procedures are an appendix to the respiratory protection
standard (29 CFR 1910.134), and employers using respirators to comply
with OSHA PELs must consult that standard. OSHA's analytical methods
are also available through OSHA's website. Secondly, OSHA believes that
guidance materials in the form of compliance assistance and outreach
tools are a more flexible means for disseminating current information
to employees and employers than appendices due to the fixed nature of
an appendix as a part of the promulgated standard. For example, OSHA
analytical methods are often updated and thus an appendix with such a
method included might easily become outdated. Appendices on medical
surveillance guidance could also become outdated as advancements in
medical science occur. Guidance documents separate from the standard,
however, could be more easily updated. Finally, guidance materials can
be disseminated in several ways and take several forms. OSHA's
experience with its outreach and compliance assistance tools has shown
these methods are very effective in disseminating information and are
well received by both employers and employees. Thus, the final Cr(VI)
standard will not contain appendices, but OSHA will issue compliance
assistance information to cover areas useful to the implementation of
this final rule.
XVI. Authority and Signature
This document was prepared under the direction of Jonathan L.
Snare, Acting Assistant Secretary of Labor for Occupational Safety and
Health, U.S. Department of Labor, 200 Constitution Avenue, NW.,
Washington, DC 20210. The Agency issues the final sections under the
following authorities: Sections 4, 6(b), 8(c), and 8(g) of the
Occupational Safety and Health Act of 1970 (29 U.S.C. 653, 655, 657);
section 107 of the Contract Work Hours and Safety Standards Act (the
Construction Safety Act) (40 U.S.C. 333); section 41, the Longshore and
Harbor Worker's Compensation Act (33 U.S.C. 941); Secretary of Labor's
Order No. 5-2002 (67 FR 65008); and 29 CFR Part 1911.
List of Subjects in 29 CFR Parts 1910, 1915, 1917, 1918, and 1926
Cancer, Chemicals, Hazardous substances, Health, Occupational
safety and health, Reporting and recordkeeping requirements.
Signed at Washington, DC., this 16th day of February, 2006.
Jonathan L. Snare,
Acting Assistant Secretary of Labor.
XVII. Final Standards
0
Chapter XVII of Title 29 of the Code of Federal Regulations is to be
amended as follows:
PART 1910--[AMENDED]
Subpart Z--[Amended]
0
1. The authority citation for Subpart Z of Part 1910 is revised to read
as follows:
Authority: Sections 4, 6, 8 of the Occupational Safety and
Health Act of 1970 (29 U.S.C. 653, 655, 657: Secretary of Labor's
Order No. 12-71 (36 FR 8754), 8-76 (41 FR 25059), 9-83 (48 FR
35736), 1-90 (55 FR 9033), 6-96 (62 FR 111), 3-2000 (65 FR 50017),
or 5-2002 (67 FR 65008), as applicable; and 29 CFR part 1911.
All of subpart Z issued under section 6(b) of the Occupational
Safety and Health Act, except those substances that have exposure
limits listed in Tables Z-1, Z-2, and Z-3 of 29 CFR 1910.1000. The
latter were issued under section 6(a) (29 U.S.C. 655(a)).
Section 1910.1000, Tables Z-1, Z-2 and Z-3 also issued under 5
U.S.C. 553, Section 1910.1000 Tables Z-1, Z-2, and Z-3 but not under
29 CFR part 1911 except for the arsenic (organic compounds),
benzene, cotton dust, and chromium (VI) listings.
Section 1910.1001 also issued under section 107 of the Contract
Work Hours and Safety Standards Act (40 U.S.C. 3704) and 5 U.S.C.
553.
Section 1910.1002 also issued under 5 U.S.C. 553 but not under
29 U.S.C. 655 or 29 CFR part 1911.
Sections 1910.1018, 1910.1029 and 1910.1200 also issued under 29
U.S.C. 653.
Section 1910.1030 also issued under Pub. L. 106-430, 114 Stat.
1901.
0
2-3. In Sec. 1910.1000:
0
a. Table Z-1 is amended by revising "tert-Butyl chromate (as
CrO3)"; by removing "Chromic acid and chromates (as
CrO3)"; and by adding "Chromium (VI) compounds" and new
footnote 5;
0
b. Table Z-2, the entry "Chromic acid and chromates (Z37.7-1971)" is
revised, and a new footnote "c" is added.
The revisions and additions read as follows:
Sec. 1910.1000 Air contaminants.
* * * * *
Table Z-1.--Limits for Air Contaminants
----------------------------------------------------------------------------------------------------------------
Substance CAS No. (c) ppm(a) \1\ mg/m\3\ (b)\1\ Skin designation
----------------------------------------------------------------------------------------------------------------
* * * * * * *
tert-Butyl chromate (as CrO3); 1189-85-1
see 1910.1026.
* * * * * * *
Chromium (VI) compounds; See
1910.1026 \5\.
* * * * * * *
----------------------------------------------------------------------------------------------------------------
\5\ See Table Z-2 for the exposure limits for any operations or sectors where the exposure limits in Sec.
1910.1026 are stayed or are otherwise not in effect."
Table Z-2
----------------------------------------------------------------------------------------------------------------
Acceptable maximum peak above the
acceptable ceiling concentration for
Substance 8-hour time Acceptable ceiling an 8-hr shift
weighted average concentration ---------------------------------------
Concentration Maximum duration
----------------------------------------------------------------------------------------------------------------
* * * * * * *
Chromic acid and chromates .................. 1 mg/10m\3\.......
(Z37.7-1971) (as CrO3)\c\.
* * * * * * *
----------------------------------------------------------------------------------------------------------------
\c\ This standard applies to any operations or sectors for which the Hexavalent Chromium standard, 1910.1026, is
stayed or otherwise is not in effect."
* * * * *
0
4. A new Section 1910.1026 is added, to read as follows:
Sec. 1910.1026 Chromium (VI).
(a) Scope. (1) This standard applies to occupational exposures to
chromium (VI) in all forms and compounds in general industry, except:
(2) Exposures that occur in the application of pesticides regulated
by the Environmental Protection Agency or another Federal government
agency (e.g., the treatment of wood with preservatives);
(3) Exposures to portland cement; or
(4) Where the employer has objective data demonstrating that a
material containing chromium or a specific process, operation, or
activity involving chromium cannot release dusts, fumes, or mists of
chromium (VI) in concentrations at or above 0.5 [mu]g/m\3\ as an 8-hour
time-weighted average (TWA) under any expected conditions of use.
(b) Definitions. For the purposes of this section the following
definitions apply:
Action level means a concentration of airborne chromium (VI) of 2.5
micrograms per cubic meter of air (2.5 [mu]g/m\3\) calculated as an 8-
hour time-weighted average (TWA).
Assistant Secretary means the Assistant Secretary of Labor for
Occupational Safety and Health, U.S. Department of Labor, or designee.
Chromium (VI) [hexavalent chromium or Cr(VI)] means chromium with a
valence of positive six, in any form and in any compound.
Director means the Director of the National Institute for
Occupational Safety and Health (NIOSH), U.S. Department of Health and
Human Services, or designee.
Emergency means any occurrence that results, or is likely to
result, in an uncontrolled release of chromium (VI). If an incidental
release of chromium (VI) can be controlled at the time of release by
employees in the immediate release area, or by maintenance personnel,
it is not an emergency.
Employee exposure means the exposure to airborne chromium (VI) that
would occur if the employee were not using a respirator.
High-efficiency particulate air [HEPA] filter means a filter that
is at least 99.97 percent efficient in removing mono-dispersed
particles of 0.3 micrometers in diameter or larger.
Historical monitoring data means data from chromium (VI) monitoring
conducted prior to May 30, 2006, obtained during work operations
conducted under workplace conditions closely resembling the processes,
types of material, control methods, work practices, and environmental
conditions in the employer's current operations.
Objective data means information such as air monitoring data from
industry-wide surveys or calculations based on the composition or
chemical and physical properties of a substance demonstrating the
employee exposure to chromium (VI) associated with a particular product
or material or a specific process, operation, or activity. The data
must reflect workplace conditions closely resembling the processes,
types of material, control methods, work practices, and environmental
conditions in the employer's current operations.
Physician or other licensed health care professional [PLHCP] is an
individual whose legally permitted scope of practice (i.e., license,
registration, or certification) allows him or her to independently
provide or be delegated the responsibility to provide some or all of
the particular health care services required by paragraph (k) of this
section.
Regulated area means an area, demarcated by the employer, where an
employee's exposure to airborne concentrations of chromium (VI)
exceeds, or can reasonably be expected to exceed, the PEL.
This section means this Sec. 1910.1026 chromium (VI) standard.
(c) Permissible exposure limit (PEL). The employer shall ensure
that no employee is exposed to an airborne concentration of chromium
(VI) in excess of 5 micrograms per cubic meter of air (5 [mu]g/
m3), calculated as an 8-hour time-weighted average (TWA).
(d) Exposure determination. (1) General. Each employer who has a
workplace or work operation covered by this section shall determine the
8-hour TWA exposure for each employee exposed to chromium (VI). This
determination shall be made in accordance with either paragraph (d)(2)
or paragraph (d)(3) of this section.
(2) Scheduled monitoring option. (i) The employer shall perform
initial monitoring to determine the 8-hour TWA exposure for each
employee on the basis of a sufficient number of personal breathing zone
air samples to accurately characterize full shift exposure on each
shift, for each job classification, in each work area. Where an
employer does representative sampling instead of sampling all employees
in order to meet this requirement, the employer shall sample the
employee(s) expected to have the highest chromium (VI) exposures.
(ii) If initial monitoring indicates that employee exposures are
below the action level, the employer may discontinue monitoring for
those employees whose exposures are represented by such monitoring.
(iii) If monitoring reveals employee exposures to be at or above
the action level, the employer shall perform periodic monitoring at
least every six months.
(iv) If monitoring reveals employee exposures to be above the PEL,
the employer shall perform periodic monitoring at least every three
months.
(v) If periodic monitoring indicates that employee exposures are
below the action level, and the result is confirmed by the result of
another monitoring
taken at least seven days later, the employer may discontinue the
monitoring for those employees whose exposures are represented by such
monitoring.
(vi) The employer shall perform additional monitoring when there
has been any change in the production process, raw materials,
equipment, personnel, work practices, or control methods that may
result in new or additional exposures to chromium (VI), or when the
employer has any reason to believe that new or additional exposures
have occurred.
(3) Performance-oriented option. The employer shall determine the
8-hour TWA exposure for each employee on the basis of any combination
of air monitoring data, historical monitoring data, or objective data
sufficient to accurately characterize employee exposure to chromium
(VI).
(4) Employee notification of determination results. (i) Where the
exposure determination indicates that employee exposure exceeds the
PEL, within 15 working days the employer shall either post the results
in an appropriate location that is accessible to all affected employees
or shall notify each affected employee individually in writing of the
results.
(ii) Whenever the exposure determination indicates that employee
exposure is above the PEL, the employer shall describe in the written
notification the corrective action being taken to reduce employee
exposure to or below the PEL.
(5) Accuracy of measurement. Where air monitoring is performed to
comply with the requirements of this section, the employer shall use a
method of monitoring and analysis that can measure chromium (VI) to
within an accuracy of plus or minus 25 percent (+/- 25%) and can
produce accurate measurements to within a statistical confidence level
of 95 percent for airborne concentrations at or above the action level.
(6) Observation of monitoring. (i) Where air monitoring is
performed to comply with the requirements of this section, the employer
shall provide affected employees or their designated representatives an
opportunity to observe any monitoring of employee exposure to chromium
(VI).
(ii) When observation of monitoring requires entry into an area
where the use of protective clothing or equipment is required, the
employer shall provide the observer with clothing and equipment and
shall assure that the observer uses such clothing and equipment and
complies with all other applicable safety and health procedures.
(e) Regulated areas. (1) Establishment. The employer shall
establish a regulated area wherever an employee's exposure to airborne
concentrations of chromium (VI) is, or can reasonably be expected to
be, in excess of the PEL.
(2) Demarcation. The employer shall ensure that regulated areas are
demarcated from the rest of the workplace in a manner that adequately
establishes and alerts employees of the boundaries of the regulated
area.
(3) Access. The employer shall limit access to regulated areas to:
(i) Persons authorized by the employer and required by work duties
to be present in the regulated area;
(ii) Any person entering such an area as a designated
representative of employees for the purpose of exercising the right to
observe monitoring procedures under paragraph (d) of this section; or
(iii) Any person authorized by the Occupational Safety and Health
Act or regulations issued under it to be in a regulated area.
(f) Methods of compliance. (1) Engineering and work practice
controls. (i) Except as permitted in paragraph (f)(1)(ii) and paragraph
(f)(1)(iii) of this section, the employer shall use engineering and
work practice controls to reduce and maintain employee exposure to
chromium (VI) to or below the PEL unless the employer can demonstrate
that such controls are not feasible. Wherever feasible engineering and
work practice controls are not sufficient to reduce employee exposure
to or below the PEL, the employer shall use them to reduce employee
exposure to the lowest levels achievable, and shall supplement them by
the use of respiratory protection that complies with the requirements
of paragraph (g) of this section.
(ii) Where painting of aircraft or large aircraft parts is
performed in the aerospace industry, the employer shall use engineering
and work practice controls to reduce and maintain employee exposure to
chromium (VI) to or below 25 [mu]g/m3 unless the employer
can demonstrate that such controls are not feasible. The employer shall
supplement such engineering and work practice controls with the use of
respiratory protection that complies with the requirements of paragraph
(g) of this section to achieve the PEL.
(iii) Where the employer can demonstrate that a process or task
does not result in any employee exposure to chromium (VI) above the PEL
for 30 or more days per year (12 consecutive months), the requirement
to implement engineering and work practice controls to achieve the PEL
does not apply to that process or task.
(2) Prohibition of rotation. The employer shall not rotate
employees to different jobs to achieve compliance with the PEL.
(g) Respiratory protection. (1) General. The employer shall provide
respiratory protection for employees during:
(i) Periods necessary to install or implement feasible engineering
and work practice controls;
(ii) Work operations, such as maintenance and repair activities,
for which engineering and work practice controls are not feasible;
(iii) Work operations for which an employer has implemented all
feasible engineering and work practice controls and such controls are
not sufficient to reduce exposures to or below the PEL;
(iv) Work operations where employees are exposed above the PEL for
fewer than 30 days per year, and the employer has elected not to
implement engineering and work practice controls to achieve the PEL; or
(v) Emergencies.
(2) Respiratory protection program. Where respirator use is
required by this section, the employer shall institute a respiratory
protection program in accordance with 29 CFR 1910.134.
(h) Protective work clothing and equipment. (1) Provision and use.
Where a hazard is present or is likely to be present from skin or eye
contact with chromium (VI), the employer shall provide appropriate
personal protective clothing and equipment at no cost to employees, and
shall ensure that employees use such clothing and equipment.
(2) Removal and storage. (i) The employer shall ensure that
employees remove all protective clothing and equipment contaminated
with chromium (VI) at the end of the work shift or at the completion of
their tasks involving chromium (VI) exposure, whichever comes first.
(ii) The employer shall ensure that no employee removes chromium
(VI)-contaminated protective clothing or equipment from the workplace,
except for those employees whose job it is to launder, clean, maintain,
or dispose of such clothing or equipment.
(iii) When contaminated protective clothing or equipment is removed
for laundering, cleaning, maintenance, or disposal, the employer shall
ensure that it is stored and transported in sealed, impermeable bags or
other closed, impermeable containers.
(iv) Bags or containers of contaminated protective clothing or
equipment that are removed from change rooms for laundering, cleaning,
maintenance, or disposal shall be labeled in accordance with the
requirements of the Hazard Communication Standard, 29 CFR 1910.1200.
(3) Cleaning and replacement. (i) The employer shall clean,
launder, repair and replace all protective clothing and equipment
required by this section as needed to maintain its effectiveness.
(ii) The employer shall prohibit the removal of chromium (VI) from
protective clothing and equipment by blowing, shaking, or any other
means that disperses chromium (VI) into the air or onto an employee's
body.
(iii) The employer shall inform any person who launders or cleans
protective clothing or equipment contaminated with chromium (VI) of the
potentially harmful effects of exposure to chromium (VI) and that the
clothing and equipment should be laundered or cleaned in a manner that
minimizes skin or eye contact with chromium (VI) and effectively
prevents the release of airborne chromium (VI) in excess of the PEL.
(i) Hygiene areas and practices. (1) General. Where protective
clothing and equipment is required, the employer shall provide change
rooms in conformance with 29 CFR 1910.141. Where skin contact with
chromium (VI) occurs, the employer shall provide washing facilities in
conformance with 29 CFR 1910.141. Eating and drinking areas provided by
the employer shall also be in conformance with Sec. 1910.141.
(2) Change rooms. The employer shall assure that change rooms are
equipped with separate storage facilities for protective clothing and
equipment and for street clothes, and that these facilities prevent
cross-contamination.
(3) Washing facilities. (i) The employer shall provide readily
accessible washing facilities capable of removing chromium (VI) from
the skin, and shall ensure that affected employees use these facilities
when necessary.
(ii) The employer shall ensure that employees who have skin contact
with chromium (VI) wash their hands and faces at the end of the work
shift and prior to eating, drinking, smoking, chewing tobacco or gum,
applying cosmetics, or using the toilet.
(4) Eating and drinking areas. (i) Whenever the employer allows
employees to consume food or beverages at a worksite where chromium
(VI) is present, the employer shall ensure that eating and drinking
areas and surfaces are maintained as free as practicable of chromium
(VI).
(ii) The employer shall ensure that employees do not enter eating
and drinking areas with protective work clothing or equipment unless
surface chromium (VI) has been removed from the clothing and equipment
by methods that do not disperse chromium (VI) into the air or onto an
employee's body.
(5) Prohibited activities. The employer shall ensure that employees
do not eat, drink, smoke, chew tobacco or gum, or apply cosmetics in
regulated areas, or in areas where skin or eye contact with chromium
(VI) occurs; or carry the products associated with these activities, or
store such products in these areas.
(j) Housekeeping. (1) General. The employer shall ensure that:
(i) All surfaces are maintained as free as practicable of
accumulations of chromium (VI).
(ii) All spills and releases of chromium (VI) containing material
are cleaned up promptly.
(2) Cleaning methods. (i) The employer shall ensure that surfaces
contaminated with chromium (VI) are cleaned by HEPA-filter vacuuming or
other methods that minimize the likelihood of exposure to chromium
(VI).
(ii) Dry shoveling, dry sweeping, and dry brushing may be used only
where HEPA-filtered vacuuming or other methods that minimize the
likelihood of exposure to chromium (VI) have been tried and found not
to be effective.
(iii) The employer shall not allow compressed air to be used to
remove chromium (VI) from any surface unless:
(A) The compressed air is used in conjunction with a ventilation
system designed to capture the dust cloud created by the compressed
air; or
(B) No alternative method is feasible.
(iv) The employer shall ensure that cleaning equipment is handled
in a manner that minimizes the reentry of chromium (VI) into the
workplace.
(3) Disposal. The employer shall ensure that:
(i) Waste, scrap, debris, and any other materials contaminated with
chromium (VI) and consigned for disposal are collected and disposed of
in sealed, impermeable bags or other closed, impermeable containers.
(ii) Bags or containers of waste, scrap, debris, and any other
materials contaminated with chromium (VI) that are consigned for
disposal are labeled in accordance with the requirements of the Hazard
Communication Standard, 29 CFR 1910.1200.
(k) Medical surveillance. (1) General. (i) The employer shall make
medical surveillance available at no cost to the employee, and at a
reasonable time and place, for all employees:
(A) Who are or may be occupationally exposed to chromium (VI) at or
above the action level for 30 or more days a year;
(B) Experiencing signs or symptoms of the adverse health effects
associated with chromium (VI) exposure; or
(C) Exposed in an emergency.
(ii) The employer shall assure that all medical examinations and
procedures required by this section are performed by or under the
supervision of a PLHCP.
(2) Frequency. The employer shall provide a medical examination:
(i) Within 30 days after initial assignment, unless the employee
has received a chromium (VI) related medical examination that meets the
requirements of this paragraph within the last twelve months;
(ii) Annually;
(iii) Within 30 days after a PLHCP's written medical opinion
recommends an additional examination;
(iv) Whenever an employee shows signs or symptoms of the adverse
health effects associated with chromium (VI) exposure;
(v) Within 30 days after exposure during an emergency which results
in an uncontrolled release of chromium (VI); or
(vi) At the termination of employment, unless the last examination
that satisfied the requirements of paragraph (k) of this section was
less than six months prior to the date of termination.
(3) Contents of examination. A medical examination consists of:
(i) A medical and work history, with emphasis on: Past, present,
and anticipated future exposure to chromium (VI); any history of
respiratory system dysfunction; any history of asthma, dermatitis, skin
ulceration, or nasal septum perforation; and smoking status and
history;
(ii) A physical examination of the skin and respiratory tract; and
(iii) Any additional tests deemed appropriate by the examining
PLHCP.
(4) Information provided to the PLHCP. The employer shall ensure
that the examining PLHCP has a copy of this standard, and shall provide
the following information:
(i) A description of the affected employee's former, current, and
anticipated duties as they relate to the employee's occupational
exposure to chromium (VI);
(ii) The employee's former, current, and anticipated levels of
occupational exposure to chromium (VI);
(iii) A description of any personal protective equipment used or to
be used by the employee, including when and for how long the employee
has used that equipment; and
(iv) Information from records of employment-related medical
examinations previously provided to the affected employee, currently
within the control of the employer.
(5) PLHCP's written medical opinion. (i) The employer shall obtain
a written medical opinion from the PLHCP, within 30 days for each
medical examination performed on each employee, which contains:
(A) The PLHCP's opinion as to whether the employee has any detected
medical condition(s) that would place the employee at increased risk of
material impairment to health from further exposure to chromium (VI);
(B) Any recommended limitations upon the employee's exposure to
chromium (VI) or upon the use of personal protective equipment such as
respirators;
(C) A statement that the PLHCP has explained to the employee the
results of the medical examination, including any medical conditions
related to chromium (VI) exposure that require further evaluation or
treatment, and any special provisions for use of protective clothing or
equipment.
(ii) The PLHCP shall not reveal to the employer specific findings
or diagnoses unrelated to occupational exposure to chromium (VI).
(iii) The employer shall provide a copy of the PLHCP's written
medical opinion to the examined employee within two weeks after
receiving it.
(l) Communication of chromium (VI) hazards to employees.
(1) General. In addition to the requirements of the Hazard
Communication Standard, 29 CFR 1910.1200, employers shall comply with
the following requirements.
(2) Employee information and training. (i) The employer shall
ensure that each employee can demonstrate knowledge of at least the
following:
(A) The contents of this section; and
(B) The purpose and a description of the medical surveillance
program required by paragraph (k) of this section.
(ii) The employer shall make a copy of this section readily
available without cost to all affected employees.
(m) Recordkeeping. (1) Air monitoring data. (i) The employer shall
maintain an accurate record of all air monitoring conducted to comply
with the requirements of this section.
(ii) This record shall include at least the following information:
(A) The date of measurement for each sample taken;
(B) The operation involving exposure to chromium (VI) that is being
monitored;
(C) Sampling and analytical methods used and evidence of their
accuracy;
(D) Number, duration, and the results of samples taken;
(E) Type of personal protective equipment, such as respirators
worn; and
(F) Name, social security number, and job classification of all
employees represented by the monitoring, indicating which employees
were actually monitored.
(iii) The employer shall ensure that exposure records are
maintained and made available in accordance with 29 CFR 1910.1020.
(2) Historical monitoring data. (i) Where the employer has relied
on historical monitoring data to determine exposure to chromium (VI),
the employer shall establish and maintain an accurate record of the
historical monitoring data relied upon.
(ii) The record shall include information that reflects the
following conditions:
(A) The data were collected using methods that meet the accuracy
requirements of paragraph (d)(5) of this section;
(B) The processes and work practices that were in use when the
historical monitoring data were obtained are essentially the same as
those to be used during the job for which exposure is being determined;
(C) The characteristics of the chromium (VI) containing material
being handled when the historical monitoring data were obtained are the
same as those on the job for which exposure is being determined;
(D) Environmental conditions prevailing when the historical
monitoring data were obtained are the same as those on the job for
which exposure is being determined; and
(E) Other data relevant to the operations, materials, processing,
or employee exposures covered by the exception.
(iii) The employer shall ensure that historical exposure records
are maintained and made available in accordance with 29 CFR 1910.1020.
(3) Objective data. (i) The employer shall maintain an accurate
record of all objective data relied upon to comply with the
requirements of this section.
(ii) This record shall include at least the following information:
(A) The chromium containing material in question;
(B) The source of the objective data;
(C) The testing protocol and results of testing, or analysis of the
material for the release of chromium (VI);
(D) A description of the process, operation, or activity and how
the data support the determination; and
(E) Other data relevant to the process, operation, activity,
material, or employee exposures.
(iii) The employer shall ensure that objective data are maintained
and made available in accordance with 29 CFR 1910.1020.
(4) Medical surveillance. (i) The employer shall establish and
maintain an accurate record for each employee covered by medical
surveillance under paragraph (k) of this section.
(ii) The record shall include the following information about the
employee:
(A) Name and social security number;
(B) A copy of the PLHCP's written opinions;
(C) A copy of the information provided to the PLHCP as required by
paragraph (k)(4) of this section.
(iii) The employer shall ensure that medical records are maintained
and made available in accordance with 29 CFR 1910.1020.
(n) Dates. (1) For employers with 20 or more employees, all
obligations of this section, except engineering controls required by
paragraph (f) of this section, commence November 27, 2006.
(2) For employers with 19 or fewer employees, all obligations of
this section, except engineering controls required by paragraph (f) of
this section, commence May 30, 2007.
(3) For all employers, engineering controls required by paragraph
(f) of this section shall be implemented no later than May 31, 2010.
PART 1915--[AMENDED]
0
5. The authority citation for 29 CFR part 1915 is revised to read as
follows:
Authority: Section 41, Longshore and Harbor Workers'
Compensation Act (33 U.S.C. 941); sections 4, 6, 8, Occupational
Safety and Health Act of 1970 (29 U.S.C. 653, 655, 657); Secretary
of Labor's Order No. 12-71 (36 FR 8754), 8-76 (41 FR 25059), 9-83
(48 FR 35736), 1-90 (55 FR 9033), 6-96 (62 FR 111), 3-2000 (65 FR
50017) or 5-2002 (67 FR 65008), as applicable.
Sections 1915.120, 1915.152 and 1915.1026 also issued under 29
CFR part 1911.
Section 1915.1001 also issued under 5 U.S.C. 553. 1915.1000 Air
contaminants.
* * * * *
0
6. In Sec. 1915.1000, Table Z, the entries for "tert-Butyl chromate
(as CrO3)", and "Chromic acid and chromates (as
CrO3)" are revised to read as follows:
Sec. 1915.1000 Air contaminants.
* * * * *
Table Z.--Shipyards
----------------------------------------------------------------------------------------------------------------
Substance CAS No.\d\ ppm\a*\ mg/m\3\ b * Skin designation
----------------------------------------------------------------------------------------------------------------
* * * * * * *
tert-Butyl chromate (as CrO3); 1189-85-1
see 1915.1026 \n\.
1
* * * * * * *
Chromium (VI) Compounds; see
1915.1026 \o\.
* * * * * * *
* * * * * * *
* * * * * * *
----------------------------------------------------------------------------------------------------------------
\3\ Use Asbestos Limit Sec. 1915.1001.
\*\ The PELS are 8-hour TWAs unless otherwise noted; a (C) designation denotes a ceiling limit. They are to be
determined from breathing-zone air samples.
\a\ Parts of vapor or gas per million parts of contaminated air by volume at 25[deg] C and 760 torr.
\b\ Milligrams of substance per cubic meter of air. When entry is in this column only, the value is exact; when
listed with a ppm entry, it is approximate.
\d\ The CAS number is for information only. Enforcement is based on the substance name. For an entry covering
more than one metal compound, measured as the metal, the CAS number for the metal is given--not CAS numbers
for the individual compounds.
\n\ If the exposure limit in 1915.1026 is stayed or is not otherwise in effect, the TLV is a ceiling of 0.1
[mu]g/m\3\ (as CrO3).
\o\ If the exposure limit in 1915.1026 is stayed or is otherwise not in effect, the TLV is 0.1 [mu]g/m\3\ (as
CrO3) as an 8-hour TWA.
0
7. A new Sec. 1915.1026 is added, to read as follows:
Sec. 1915.1026 Chromium (VI).
(a) Scope. (1) This standard applies to occupational exposures to
chromium (VI) in all forms and compounds in shipyards, marine
terminals, and longshoring, except:
(2) Exposures that occur in the application of pesticides regulated
by the Environmental Protection Agency or another Federal government
agency (e.g., the treatment of wood with preservatives);
(3) Exposures to portland cement; or
(4) Where the employer has objective data demonstrating that a
material containing chromium or a specific process, operation, or
activity involving chromium cannot release dusts, fumes, or mists of
chromium (VI) in concentrations at or above 0.5 [mu]g/m3 as
an 8-hour time-weighted average (TWA) under any expected conditions of
use.
(b) Definitions. For the purposes of this section the following
definitions apply:
Action level means a concentration of airborne chromium (VI) of 2.5
micrograms per cubic meter of air (2.5 [mu]g/m3) calculated
as an 8-hour time-weighted average (TWA).
Assistant Secretary means the Assistant Secretary of Labor for
Occupational Safety and Health, U.S. Department of Labor, or designee.
Chromium (VI) [hexavalent chromium or Cr(VI)] means chromium with a
valence of positive six, in any form and in any compound.
Director means the Director of the National Institute for
Occupational Safety and Health (NIOSH), U.S. Department of Health and
Human Services, or designee.
Emergency means any occurrence that results, or is likely to
result, in an uncontrolled release of chromium (VI). If an incidental
release of chromium (VI) can be controlled at the time of release by
employees in the immediate release area, or by maintenance personnel,
it is not an emergency.
Employee exposure means the exposure to airborne chromium (VI) that
would occur if the employee were not using a respirator.
High-efficiency particulate air [HEPA] filter means a filter that
is at least 99.97 percent efficient in removing mono-dispersed
particles of 0.3 micrometers in diameter or larger.
Historical monitoring data means data from chromium (VI) monitoring
conducted prior to May 30, 2006, obtained during work operations
conducted under workplace conditions closely resembling the processes,
types of material, control methods, work practices, and environmental
conditions in the employer's current operations.
Objective data means information such as air monitoring data from
industry-wide surveys or calculations based on the composition or
chemical and physical properties of a substance demonstrating the
employee exposure to chromium (VI) associated with a particular product
or material or a specific process, operation, or activity. The data
must reflect workplace conditions closely resembling the processes,
types of material, control methods, work practices, and environmental
conditions in the employer's current operations.
Physician or other licensed health care professional [PLHCP] is an
individual whose legally permitted scope of practice (i.e., license,
registration, or certification) allows him or her to independently
provide or be delegated the responsibility to provide some or all of
the particular health care services required by paragraph (i) of this
section.
This section means this Sec. 1915.1026 chromium (VI) standard.
(c) Permissible exposure limit (PEL). The employer shall ensure
that no employee is exposed to an airborne concentration of chromium
(VI) in excess of 5 micrograms per cubic meter of air (5 [mu]g/
m3), calculated as an 8-hour time-weighted average (TWA).
(d) Exposure determination. (1) General. Each employer who has a
workplace or work operation covered by this section shall determine the
8-hour TWA exposure for each employee exposed to chromium (VI). This
determination shall be made in accordance with either paragraph (d)(2)
or paragraph (d)(3) of this section.
(2) Scheduled monitoring option. (i) The employer shall perform
initial monitoring to determine the 8-hour TWA exposure for each
employee on the basis of a sufficient number of personal breathing zone
air samples to accurately characterize full shift exposure on each
shift, for each job classification, in each work area. Where an
employer does representative
sampling instead of sampling all employees in order to meet this
requirement, the employer shall sample the employee(s) expected to have
the highest chromium (VI) exposures.
(ii) If initial monitoring indicates that employee exposures are
below the action level, the employer may discontinue monitoring for
those employees whose exposures are represented by such monitoring.
(iii) If monitoring reveals employee exposures to be at or above
the action level, the employer shall perform periodic monitoring at
least every six months.
(iv) If monitoring reveals employee exposures to be above the PEL,
the employer shall perform periodic monitoring at least every three
months.
(v) If periodic monitoring indicates that employee exposures are
below the action level, and the result is confirmed by the result of
another monitoring taken at least seven days later, the employer may
discontinue the monitoring for those employees whose exposures are
represented by such monitoring.
(vi) The employer shall perform additional monitoring when there
has been any change in the production process, raw materials,
equipment, personnel, work practices, or control methods that may
result in new or additional exposures to chromium (VI), or when the
employer has any reason to believe that new or additional exposures
have occurred.
(3) Performance-oriented option. The employer shall determine the
8-hour TWA exposure for each employee on the basis of any combination
of air monitoring data, historical monitoring data, or objective data
sufficient to accurately characterize employee exposure to chromium
(VI).
(4) Employee notification of determination results. (i) Where the
exposure determination indicates that employee exposure exceeds the
PEL, as soon as possible but not more than 5 working days later the
employer shall either post the results in an appropriate location that
is accessible to all affected employees or shall notify each affected
employee individually in writing of the results.
(ii) Whenever the exposure determination indicates that employee
exposure is above the PEL, the employer shall describe in the written
notification the corrective action being taken to reduce employee
exposure to or below the PEL.
(5) Accuracy of measurement. Where air monitoring is performed to
comply with the requirements of this section, the employer shall use a
method of monitoring and analysis that can measure chromium (VI) to
within an accuracy of plus or minus 25 percent (+/-25%) and can produce
accurate measurements to within a statistical confidence level of 95
percent for airborne concentrations at or above the action level.
(6) Observation of monitoring. (i) Where air monitoring is
performed to comply with the requirements of this section, the employer
shall provide affected employees or their designated representatives an
opportunity to observe any monitoring of employee exposure to chromium
(VI).
(ii) When observation of monitoring requires entry into an area
where the use of protective clothing or equipment is required, the
employer shall provide the observer with clothing and equipment and
shall assure that the observer uses such clothing and equipment and
complies with all other applicable safety and health procedures.
(e) Methods of compliance. (1) Engineering and work practice
controls. (i) Except as permitted in paragraph (e)(1)(ii) of this
section, the employer shall use engineering and work practice controls
to reduce and maintain employee exposure to chromium (VI) to or below
the PEL unless the employer can demonstrate that such controls are not
feasible. Wherever feasible engineering and work practice controls are
not sufficient to reduce employee exposure to or below the PEL, the
employer shall use them to reduce employee exposure to the lowest
levels achievable, and shall supplement them by the use of respiratory
protection that complies with the requirements of paragraph (f) of this
section.
(ii) Where the employer can demonstrate that a process or task does
not result in any employee exposure to chromium (VI) above the PEL for
30 or more days per year (12 consecutive months), the requirement to
implement engineering and work practice controls to achieve the PEL
does not apply to that process or task.
(2) Prohibition of rotation. The employer shall not rotate
employees to different jobs to achieve compliance with the PEL.
(f) Respiratory protection. (1) General. The employer shall provide
respiratory protection for employees during:
(i) Periods necessary to install or implement feasible engineering
and work practice controls;
(ii) Work operations, such as maintenance and repair activities,
for which engineering and work practice controls are not feasible;
(iii) Work operations for which an employer has implemented all
feasible engineering and work practice controls and such controls are
not sufficient to reduce exposures to or below the PEL;
(iv) Work operations where employees are exposed above the PEL for
fewer than 30 days per year, and the employer has elected not to
implement engineering and work practice controls to achieve the PEL; or
(v) Emergencies.
(2) Respiratory protection program. Where respirator use is
required by this section, the employer shall institute a respiratory
protection program in accordance with 29 CFR 1910.134.
(g) Protective work clothing and equipment. (1) Provision and use.
Where a hazard is present or is likely to be present from skin or eye
contact with chromium (VI), the employer shall provide appropriate
personal protective clothing and equipment at no cost to employees, and
shall ensure that employees use such clothing and equipment.
(2) Removal and storage. (i) The employer shall ensure that
employees remove all protective clothing and equipment contaminated
with chromium (VI) at the end of the work shift or at the completion of
their tasks involving chromium (VI) exposure, whichever comes first.
(ii) The employer shall ensure that no employee removes chromium
(VI)-contaminated protective clothing or equipment from the workplace,
except for those employees whose job it is to launder, clean, maintain,
or dispose of such clothing or equipment.
(iii) When contaminated protective clothing or equipment is removed
for laundering, cleaning, maintenance, or disposal, the employer shall
ensure that it is stored and transported in sealed, impermeable bags or
other closed, impermeable containers.
(iv) Bags or containers of contaminated protective clothing or
equipment that are removed from change rooms for laundering, cleaning,
maintenance, or disposal shall be labeled in accordance with the
requirements of the Hazard Communication Standard, 29 CFR 1910.1200.
(3) Cleaning and replacement. (i) The employer shall clean,
launder, repair and replace all protective clothing and equipment
required by this section as needed to maintain its effectiveness.
(ii) The employer shall prohibit the removal of chromium (VI) from
protective clothing and equipment by blowing, shaking, or any other
means that disperses chromium (VI) into the air or onto an employee's
body.
(iii) The employer shall inform any person who launders or cleans
protective clothing or equipment contaminated with chromium (VI) of the
potentially harmful effects of exposure to chromium (VI) and that the
clothing and equipment should be laundered or cleaned in a manner that
minimizes skin or eye contact with chromium (VI) and effectively
prevents the release of airborne chromium (VI) in excess of the PEL.
(h) Hygiene areas and practices. (1) General. Where protective
clothing and equipment is required, the employer shall provide change
rooms in conformance with 29 CFR 1910.141. Where skin contact with
chromium (VI) occurs, the employer shall provide washing facilities in
conformance with 29 CFR 1915.97. Eating and drinking areas provided by
the employer shall also be in conformance with Sec. 1915.97.
(2) Change rooms. The employer shall assure that change rooms are
equipped with separate storage facilities for protective clothing and
equipment and for street clothes, and that these facilities prevent
cross-contamination.
(3) Washing facilities. (i) The employer shall provide readily
accessible washing facilities capable of removing chromium (VI) from
the skin, and shall ensure that affected employees use these facilities
when necessary.
(ii) The employer shall ensure that employees who have skin contact
with chromium (VI) wash their hands and faces at the end of the work
shift and prior to eating, drinking, smoking, chewing tobacco or gum,
applying cosmetics, or using the toilet.
(4) Eating and drinking areas. (i) Whenever the employer allows
employees to consume food or beverages at a worksite where chromium
(VI) is present, the employer shall ensure that eating and drinking
areas and surfaces are maintained as free as practicable of chromium
(VI).
(ii) The employer shall ensure that employees do not enter eating
and drinking areas with protective work clothing or equipment unless
surface chromium (VI) has been removed from the clothing and equipment
by methods that do not disperse chromium (VI) into the air or onto an
employee's body.
(5) Prohibited activities. The employer shall ensure that employees
do not eat, drink, smoke, chew tobacco or gum, or apply cosmetics in
areas where skin or eye contact with chromium (VI) occurs; or carry the
products associated with these activities, or store such products in
these areas.
(i) Medical surveillance. (1) General. (i) The employer shall make
medical surveillance available at no cost to the employee, and at a
reasonable time and place, for all employees:
(A) Who are or may be occupationally exposed to chromium (VI) at or
above the action level for 30 or more days a year;
(B) Experiencing signs or symptoms of the adverse health effects
associated with chromium (VI) exposure; or
(C) Exposed in an emergency.
(ii) The employer shall assure that all medical examinations and
procedures required by this section are performed by or under the
supervision of a PLHCP.
(2) Frequency. The employer shall provide a medical examination:
(i) Within 30 days after initial assignment, unless the employee
has received a chromium (VI) related medical examination that meets the
requirements of this paragraph within the last twelve months;
(ii) Annually;
(iii) Within 30 days after a PLHCP's written medical opinion
recommends an additional examination;
(iv) Whenever an employee shows signs or symptoms of the adverse
health effects associated with chromium (VI) exposure;
(v) Within 30 days after exposure during an emergency which results
in an uncontrolled release of chromium (VI); or
(vi) At the termination of employment, unless the last examination
that satisfied the requirements of paragraph (i) of this section was
less than six months prior to the date of termination.
(3) Contents of examination. A medical examination consists of:
(i) A medical and work history, with emphasis on: past, present,
and anticipated future exposure to chromium (VI); any history of
respiratory system dysfunction; any history of asthma, dermatitis, skin
ulceration, or nasal septum perforation; and smoking status and
history;
(ii) A physical examination of the skin and respiratory tract; and
(iii) Any additional tests deemed appropriate by the examining
PLHCP.
(4) Information provided to the PLHCP. The employer shall ensure
that the examining PLHCP has a copy of this standard, and shall provide
the following information:
(i) A description of the affected employee's former, current, and
anticipated duties as they relate to the employee's occupational
exposure to chromium (VI);
(ii) The employee's former, current, and anticipated levels of
occupational exposure to chromium (VI);
(iii) A description of any personal protective equipment used or to
be used by the employee, including when and for how long the employee
has used that equipment; and
(iv) Information from records of employment-related medical
examinations previously provided to the affected employee, currently
within the control of the employer.
(5) PLHCP's written medical opinion. (i) The employer shall obtain
a written medical opinion from the PLHCP, within 30 days for each
medical examination performed on each employee, which contains:
(A) The PLHCP's opinion as to whether the employee has any detected
medical condition(s) that would place the employee at increased risk of
material impairment to health from further exposure to chromium (VI);
(B) Any recommended limitations upon the employee's exposure to
chromium (VI) or upon the use of personal protective equipment such as
respirators;
(C) A statement that the PLHCP has explained to the employee the
results of the medical examination, including any medical conditions
related to chromium (VI) exposure that require further evaluation or
treatment, and any special provisions for use of protective clothing or
equipment.
(ii) The PLHCP shall not reveal to the employer specific findings
or diagnoses unrelated to occupational exposure to chromium (VI).
(iii) The employer shall provide a copy of the PLHCP's written
medical opinion to the examined employee within two weeks after
receiving it.
(j) Communication of chromium (VI) hazards to employees. (1)
General. In addition to the requirements of the Hazard Communication
Standard, 29 CFR 1910.1200, employers shall comply with the following
requirements.
(2) Employee information and training. (i) The employer shall
ensure that each employee can demonstrate knowledge of at least the
following:
(A) The contents of this section; and
(B) The purpose and a description of the medical surveillance
program required by paragraph (i) of this section.
(ii) The employer shall make a copy of this section readily
available without cost to all affected employees.
(k) Recordkeeping. (1) Air monitoring data. (i) The employer shall
maintain an accurate record of all air monitoring conducted to comply
with the requirements of this section.
(ii) This record shall include at least the following information:
(A) The date of measurement for each sample taken;
(B) The operation involving exposure to chromium (VI) that is being
monitored;
(C) Sampling and analytical methods used and evidence of their
accuracy;
(D) Number, duration, and the results of samples taken;
(E) Type of personal protective equipment, such as respirators
worn; and
(F) Name, social security number, and job classification of all
employees represented by the monitoring, indicating which employees
were actually monitored.
(iii) The employer shall ensure that exposure records are
maintained and made available in accordance with 29 CFR 1910.1020.
(2) Historical monitoring data. (i) Where the employer has relied
on historical monitoring data to determine exposure to chromium (VI),
the employer shall establish and maintain an accurate record of the
historical monitoring data relied upon.
(ii) The record shall include information that reflects the
following conditions:
(A) The data were collected using methods that meet the accuracy
requirements of paragraph (d)(5) of this section;
(B) The processes and work practices that were in use when the
historical monitoring data were obtained are essentially the same as
those to be used during the job for which exposure is being determined;
(C) The characteristics of the chromium (VI) containing material
being handled when the historical monitoring data were obtained are the
same as those on the job for which exposure is being determined;
(D) Environmental conditions prevailing when the historical
monitoring data were obtained are the same as those on the job for
which exposure is being determined; and
(E) Other data relevant to the operations, materials, processing,
or employee exposures covered by the exception.
(iii) The employer shall ensure that historical exposure records
are maintained and made available in accordance with 29 CFR 1910.1020.
(3) Objective data. (i) The employer shall maintain an accurate
record of all objective data relied upon to comply with the
requirements of this section.
(ii) This record shall include at least the following information:
(A) The chromium containing material in question;
(B) The source of the objective data;
(C) The testing protocol and results of testing, or analysis of the
material for the release of chromium (VI);
(D) A description of the process, operation, or activity and how
the data support the determination; and
(E) Other data relevant to the process, operation, activity,
material, or employee exposures.
(iii) The employer shall ensure that objective data are maintained
and made available in accordance with 29 CFR 1910.1020.
(4) Medical surveillance. (i) The employer shall establish and
maintain an accurate record for each employee covered by medical
surveillance under paragraph (i) of this section.
(ii) The record shall include the following information about the
employee:
(A) Name and social security number;
(B) A copy of the PLHCP's written opinions;
(C) A copy of the information provided to the PLHCP as required by
paragraph (i)(4) of this section.
(iii) The employer shall ensure that medical records are maintained
and made available in accordance with 29 CFR 1910.1020.
(l) Dates. (1) For employers with 20 or more employees, all
obligations of this section, except engineering controls required by
paragraph (e) of this section, commence November 27, 2006.
(2) For employers with 19 or fewer employees, all obligations of
this section, except engineering controls required by paragraph (e) of
this section, commence May 30, 2007.
(3) For all employers, engineering controls required by paragraph
(e) of this section shall be implemented no later than May 31, 2010.
PART 1917--[AMENDED]
0
8. The authority citation for 29 CFR Part 1917 is revised to read as
follows:
Authority: Section 41, Longshore and Harbor Workers'
Compensation Act (33 U.S.C. 941); sections 4, 6, 8, Occupational
Safety and Health Act of 1970 (29 U.S.C. 653, 655, 657); Secretary
of Labor's Order Nos. 12-71 (36 FR 8754), 8-76 (41 FR 25059), 9-83
(48 FR 35736), 6-96 (62 FR 111), or 5-2002 (67 FR 65008), as
applicable; and 29 CFR part 1911.
Section 1917.28 also issued under 5 U.S.C. 553.
Section 1917.29 also issued under Sec.29, Hazardous Materials
Transportation Uniform Safety Act of 1990 (49 U.S.C. 1801-1819 and 5
U.S.C. 553).
0
9. New paragraphs (a)(2)(xiii)(E) and (b) are added to Sec. 1917.1, to
read as follows:
Sec. 1917.1 Scope and applicability.
(a) * * *
(2) * * *
(xiii) * * *
(E) Hexavalent chromium Sec. 1910.1026 (See Sec. 1915.1026)
* * * * *
(b) Section 1915.1026 applies to any occupational exposures to
hexavalent chromium in workplaces covered by this Part.
PART 1918--[AMENDED]
0
10. The authority citation for 29 CFR part 1918 is revised to read as
follows:
Authority: Sections 4, 6, 8, Occupational Safety and Health Act
of 1970 (29 U.S.C. 653, 655, 657); section 41, Longshore and Harbor
Workers' Compensation Act (33 U.S.C. 941); Secretary of Labor's
Order Nos. 12-71 (36 FR 8754); 8-76 (41 FR 25059), 9-83 (48 FR
35736); 6-96 (62 FR 111) or 5-2002 (67 FR 65008), as applicable; and
29 CFR part 1911.
Section 1918.90 also issued under 5 U.S.C. 553
Section 1918.100 also issued under Sec. 29, Hazardous Materials
Transportation Uniform Safety Act of 1990 (49 U.S.C. 1801-1819 and 5
U.S.C. 553).
0
11. New paragraphs (b)(9)(v) and (c) are added to Sec. 1918.1 to read
as follows:
Sec. 1918.1 Scope and application.
* * * * *
(b) * * *
(9) * * *
(v) Hexavalent chromium Sec. 1910.1026 (See Sec. 1915.1026)
* * * * *
(c) Section 1915.1026 applies to any occupational exposures to
hexavalent chromium in workplaces covered by this part.
PART 1926--[AMENDED]
Subpart D--[Amended]
0
12. The authority citation for subpart D of 29 CFR part 1926 is revised
to read as follows:
Authority: Section 107, Contract Work Hours and Safety Standards
Act (40 U.S.C. 333); sections 4, 6, 8, Occupational Safety and
Health Act of 1970 (29 U.S.C. 653, 655, 657);5 U.S.C. 553; Secretary
of Labor's Order Nos. 12-71 (36 FR 8754), 8-76 (41 FR 25059), 9-83
(48 FR 35736), 1-90 (55 FR 9033), 6-96 (62 FR 111), 3-2000 (65 FR
50017), or 5-2002 (67 FR 65008), as applicable; and 29 CFR part
1911.
0
13. In Appendix A to Sec. 1926.55, the entries for "tert-Butyl
chromate (as CrO3)" and "Chromic acid and chromates (as
CrO3)" are revised to read as follows:
Sec. 1926.55 Gases, vapors, fumes, dusts, and mists.
* * * * *
Appendix A to Sec. 1926.55.--1970 American Conference of Governmental Industrial Hygienists' Threshold Limit
Values of Airborne Contaminants
[Threshold limit values of airborne contaminants for construction]
----------------------------------------------------------------------------------------------------------------
Substance CAS No.\d\ ppm \a\ mg/m\3\ \b\ Skin designation
----------------------------------------------------------------------------------------------------------------
* * * * * * *
tert-Butyl chromate (as CrO3); 1189-85-1
see 1926.1126n.
* * * * * * *
Chromium (VI) Compounds; See
1926.1126\o\.
* * * * * * *
----------------------------------------------------------------------------------------------------------------
* * * * * * *
\3\ Use Asbestos Limit Sec. 1915.1001
\a\ Parts of vapor or gas per million parts of contaminated air by volume at 25[deg] C and 760 torr.
\b\ Milligrams of substance per cubic meter of air. When entry is in this column only, the value is exact; when
listed with a ppm entry, it is approximate.
\d\ The CAS number is for information only. Enforcement is based on the substance name. For an entry covering
more than one metal compound, measured as the metal, the CAS number for the metal is given--not CAS numbers
for the individual compounds.
\n\ If the exposure limit in 1926.1026 is stayed or is not otherwise in effect, the TLV is a ceiling of 0.1 mg/
m\3\ (as CrO3).
\o\ If the exposure limit in 1926.1026 is stayed or is not otherwise in effect, the TLV is 0.1 mg/m\3\ (as CrO3)
as an 8-hour TWA.
Subpart Z--[Amended]
0
14. The authority citation for subpart Z of 29 CFR part 1926 is revised
to read as follows:
Authority: Section 107, Contract Work Hours and Safety Standards
Act (40 U.S.C. 333); Sections 4, 6, 8, Occupational Safety and
Health Act of 1970 (29 U.S.C. 653, 655, 657); Secretary of Labor's
Order Nos. 12-71 (36 FR 8754), 8-76 (41 FR 25059), 9-83 (48 FR
35736), 1-90 (55 FR 9033), 6-96 (62 FR 111), 3-2000 (65 FR 50017) or
5-2002 (67 FR 65008), as applicable; and 29 CFR part 1911.
Sections 1926.1101 and 1926.1127 also issued under 5 U.S.C. 553.
Section 1926.1102 not issued under 29 U. S. C. 655 or 29 CFR
part 1911; also issued under 5 U.S.C. 553.
0
16. A new section 1926.1126 is added to subpart Z of 29 CFR part 1926
to read as follows:
Sec. 1926.1126 Chromium (VI).
(a) Scope. (1) This standard applies to occupational exposures to
chromium (VI) in all forms and compounds in construction, except:
(2) Exposures that occur in the application of pesticides regulated
by the Environmental Protection Agency or another Federal government
agency (e.g., the treatment of wood with preservatives);
(3) Exposures to portland cement; or
(4) Where the employer has objective data demonstrating that a
material containing chromium or a specific process, operation, or
activity involving chromium cannot release dusts, fumes, or mists of
chromium (VI) in concentrations at or above 0.5 [mu]g/m\3\ as an 8-hour
time-weighted average (TWA) under any expected conditions of use.
(b) Definitions. For the purposes of this section the following
definitions apply:
Action level means a concentration of airborne chromium (VI) of 2.5
micrograms per cubic meter of air (2.5 [mu]g/m\3\) calculated as an 8-
hour time-weighted average (TWA).
Assistant Secretary means the Assistant Secretary of Labor for
Occupational Safety and Health, U.S. Department of Labor, or designee.
Chromium (VI) [hexavalent chromium or Cr(VI)] means chromium with a
valence of positive six, in any form and in any compound.
Director means the Director of the National Institute for
Occupational Safety and Health (NIOSH), U.S. Department of Health and
Human Services, or designee.
Emergency means any occurrence that results, or is likely to
result, in an uncontrolled release of chromium (VI). If an incidental
release of chromium (VI) can be controlled at the time of release by
employees in the immediate release area, or by maintenance personnel,
it is not an emergency.
Employee exposure means the exposure to airborne chromium (VI) that
would occur if the employee were not using a respirator.
High-efficiency particulate air [HEPA] filter means a filter that
is at least 99.97 percent efficient in removing mono-dispersed
particles of 0.3 micrometers in diameter or larger.
Historical monitoring data means data from chromium (VI) monitoring
conducted prior to May 30, 2006, obtained during work operations
conducted under workplace conditions closely resembling the processes,
types of material, control methods, work practices, and environmental
conditions in the employer's current operations.
Objective data means information such as air monitoring data from
industry-wide surveys or calculations based on the composition or
chemical and physical properties of a substance demonstrating the
employee exposure to chromium (VI) associated with a particular product
or material or a specific process, operation, or activity. The data
must reflect workplace conditions closely resembling the processes,
types of material, control methods, work practices, and environmental
conditions in the employer's current operations.
Physician or other licensed health care professional [PLHCP] is an
individual whose legally permitted scope of practice (i.e., license,
registration, or certification) allows him or her to independently
provide or be delegated the responsibility to provide some or all of
the particular health care services required by paragraph (i) of this
section.
This section means this Sec. 1926.1126 chromium (VI) standard.
(c) Permissible exposure limit (PEL). The employer shall ensure
that no employee is exposed to an airborne concentration of chromium
(VI) in excess of 5 micrograms per cubic meter of air (5 [mu]g/m\3\),
calculated as an 8-hour time-weighted average (TWA).
(d) Exposure determination. (1) General. Each employer who has a
workplace or work operation covered by this section shall determine the
8-hour TWA exposure for each employee exposed to chromium (VI). This
determination shall be made in accordance with either paragraph (d)(2) or
paragraph (d)(3) of this section.
(2) Scheduled monitoring option. (i) The employer shall perform
initial monitoring to determine the 8-hour TWA exposure for each
employee on the basis of a sufficient number of personal breathing zone
air samples to accurately characterize full shift exposure on each
shift, for each job classification, in each work area. Where an
employer does representative sampling instead of sampling all employees
in order to meet this requirement, the employer shall sample the
employee(s) expected to have the highest chromium (VI) exposures.
(ii) If initial monitoring indicates that employee exposures are
below the action level, the employer may discontinue monitoring for
those employees whose exposures are represented by such monitoring.
(iii) If monitoring reveals employee exposures to be at or above
the action level, the employer shall perform periodic monitoring at
least every six months.
(iv) If monitoring reveals employee exposures to be above the PEL,
the employer shall perform periodic monitoring at least every three
months.
(v) If periodic monitoring indicates that employee exposures are
below the action level, and the result is confirmed by the result of
another monitoring taken at least seven days later, the employer may
discontinue the monitoring for those employees whose exposures are
represented by such monitoring.
(vi) The employer shall perform additional monitoring when there
has been any change in the production process, raw materials,
equipment, personnel, work practices, or control methods that may
result in new or additional exposures to chromium (VI), or when the
employer has any reason to believe that new or additional exposures
have occurred.
(3) Performance-oriented option. The employer shall determine the
8-hour TWA exposure for each employee on the basis of any combination
of air monitoring data, historical monitoring data, or objective data
sufficient to accurately characterize employee exposure to chromium
(VI).
(4) Employee notification of determination results. (i) Where the
exposure determination indicates that employee exposure exceeds the
PEL, as soon as possible but not more than 5 working days later the
employer shall either post the results in an appropriate location that
is accessible to all affected employees or shall notify each affected
employee individually in writing of the results.
(ii) Whenever the exposure determination indicates that employee
exposure is above the PEL, the employer shall describe in the written
notification the corrective action being taken to reduce employee
exposure to or below the PEL.
(5) Accuracy of measurement. Where air monitoring is performed to
comply with the requirements of this section, the employer shall use a
method of monitoring and analysis that can measure chromium (VI) to
within an accuracy of plus or minus 25 percent (25%) and
can produce accurate measurements to within a statistical confidence
level of 95 percent for airborne concentrations at or above the action
level.
(6) Observation of monitoring. (i) Where air monitoring is
performed to comply with the requirements of this section, the employer
shall provide affected employees or their designated representatives an
opportunity to observe any monitoring of employee exposure to chromium
(VI).
(ii) When observation of monitoring requires entry into an area
where the use of protective clothing or equipment is required, the
employer shall provide the observer with clothing and equipment and
shall assure that the observer uses such clothing and equipment and
complies with all other applicable safety and health procedures.
(e) Methods of compliance. (1) Engineering and work practice
controls. (i) Except as permitted in paragraph (e)(1)(ii) of this
section, the employer shall use engineering and work practice controls
to reduce and maintain employee exposure to chromium (VI) to or below
the PEL unless the employer can demonstrate that such controls are not
feasible. Wherever feasible engineering and work practice controls are
not sufficient to reduce employee exposure to or below the PEL, the
employer shall use them to reduce employee exposure to the lowest
levels achievable, and shall supplement them by the use of respiratory
protection that complies with the requirements of paragraph (f) of this
section.
(ii) Where the employer can demonstrate that a process or task does
not result in any employee exposure to chromium (VI) above the PEL for
30 or more days per year (12 consecutive months), the requirement to
implement engineering and work practice controls to achieve the PEL
does not apply to that process or task.
(2) Prohibition of rotation. The employer shall not rotate
employees to different jobs to achieve compliance with the PEL.
(f) Respiratory protection. (1) General. The employer shall provide
respiratory protection for employees during:
(i) Periods necessary to install or implement feasible engineering
and work practice controls;
(ii) Work operations, such as maintenance and repair activities,
for which engineering and work practice controls are not feasible;
(iii) Work operations for which an employer has implemented all
feasible engineering and work practice controls and such controls are
not sufficient to reduce exposures to or below the PEL;
(iv) Work operations where employees are exposed above the PEL for
fewer than 30 days per year, and the employer has elected not to
implement engineering and work practice controls to achieve the PEL; or
(v) Emergencies.
(2) Respiratory protection program. Where respirator use is
required by this section, the employer shall institute a respiratory
protection program in accordance with 29 CFR 1910.134.
(g) Protective work clothing and equipment. (1) Provision and use.
Where a hazard is present or is likely to be present from skin or eye
contact with chromium (VI), the employer shall provide appropriate
personal protective clothing and equipment at no cost to employees, and
shall ensure that employees use such clothing and equipment.
(2) Removal and storage. (i) The employer shall ensure that
employees remove all protective clothing and equipment contaminated
with chromium (VI) at the end of the work shift or at the completion of
their tasks involving chromium (VI) exposure, whichever comes first.
(ii) The employer shall ensure that no employee removes chromium
(VI)-contaminated protective clothing or equipment from the workplace,
except for those employees whose job it is to launder, clean, maintain,
or dispose of such clothing or equipment.
(iii) When contaminated protective clothing or equipment is removed
for laundering, cleaning, maintenance, or disposal, the employer shall
ensure that it is stored and transported in sealed, impermeable bags or
other closed, impermeable containers.
(iv) Bags or containers of contaminated protective clothing or
equipment that are removed from change rooms for laundering, cleaning,
maintenance, or disposal shall be labeled in accordance with the
requirements of the Hazard Communication Standard, 29 CFR 1910.1200.
(3) Cleaning and replacement. (i) The employer shall clean,
launder, repair and replace all protective clothing and equipment
required by this section as needed to maintain its effectiveness.
(ii) The employer shall prohibit the removal of chromium (VI) from
protective clothing and equipment by blowing, shaking, or any other
means that disperses chromium (VI) into the air or onto an employee's
body.
(iii) The employer shall inform any person who launders or cleans
protective clothing or equipment contaminated with chromium (VI) of the
potentially harmful effects of exposure to chromium (VI) and that the
clothing and equipment should be laundered or cleaned in a manner that
minimizes skin or eye contact with chromium (VI) and effectively
prevents the release of airborne chromium (VI) in excess of the PEL.
(h) Hygiene areas and practices. (1) General. Where protective
clothing and equipment is required, the employer shall provide change
rooms in conformance with 29 CFR 1926.51 Where skin contact with
chromium (VI) occurs, the employer shall provide washing facilities in
conformance with 29 CFR 1926.51. Eating and drinking areas provided by
the employer shall also be in conformance with Sec. 1926.51.
(2) Change rooms. The employer shall assure that change rooms are
equipped with separate storage facilities for protective clothing and
equipment and for street clothes, and that these facilities prevent
cross-contamination.
(3) Washing facilities. (i) The employer shall provide readily
accessible washing facilities capable of removing chromium (VI) from
the skin, and shall ensure that affected employees use these facilities
when necessary.
(ii) The employer shall ensure that employees who have skin contact
with chromium (VI) wash their hands and faces at the end of the work
shift and prior to eating, drinking, smoking, chewing tobacco or gum,
applying cosmetics, or using the toilet.
(4) Eating and drinking areas. (i) Whenever the employer allows
employees to consume food or beverages at a worksite where chromium
(VI) is present, the employer shall ensure that eating and drinking
areas and surfaces are maintained as free as practicable of chromium
(VI).
(ii) The employer shall ensure that employees do not enter eating
and drinking areas with protective work clothing or equipment unless
surface chromium (VI) has been removed from the clothing and equipment
by methods that do not disperse chromium (VI) into the air or onto an
employee's body.
(5) Prohibited activities. The employer shall ensure that employees
do not eat, drink, smoke, chew tobacco or gum, or apply cosmetics in
areas where skin or eye contact with chromium (VI) occurs; or carry the
products associated with these activities, or store such products in
these areas.
(i) Medical surveillance. (1) General. (i) The employer shall make
medical surveillance available at no cost to the employee, and at a
reasonable time and place, for all employees:
(A) Who are or may be occupationally exposed to chromium (VI) at or
above the action level for 30 or more days a year;
(B) Experiencing signs or symptoms of the adverse health effects
associated with chromium (VI) exposure; or
(C) Exposed in an emergency.
(ii) The employer shall assure that all medical examinations and
procedures required by this section are performed by or under the
supervision of a PLHCP.
(2) Frequency. The employer shall provide a medical examination:
(i) Within 30 days after initial assignment, unless the employee
has received a chromium (VI) related medical examination that meets the
requirements of this paragraph within the last twelve months;
(ii) Annually;
(iii) Within 30 days after a PLHCP's written medical opinion
recommends an additional examination;
(iv) Whenever an employee shows signs or symptoms of the adverse
health effects associated with chromium (VI) exposure;
(v) Within 30 days after exposure during an emergency which results
in an uncontrolled release of chromium (VI); or
(vi) At the termination of employment, unless the last examination
that satisfied the requirements of paragraph (i) of this section was
less than six months prior to the date of termination.
(3) Contents of examination. A medical examination consists of:
(i) A medical and work history, with emphasis on: past, present,
and anticipated future exposure to chromium (VI); any history of
respiratory system dysfunction; any history of asthma, dermatitis, skin
ulceration, or nasal septum perforation; and smoking status and
history;
(ii) A physical examination of the skin and respiratory tract; and
(iii) Any additional tests deemed appropriate by the examining
PLHCP.
(4) Information provided to the PLHCP. The employer shall ensure
that the examining PLHCP has a copy of this standard, and shall provide
the following information:
(i) A description of the affected employee's former, current, and
anticipated duties as they relate to the employee's occupational
exposure to chromium (VI);
(ii) The employee's former, current, and anticipated levels of
occupational exposure to chromium (VI);
(iii) A description of any personal protective equipment used or to
be used by the employee, including when and for how long the employee
has used that equipment; and
(iv) Information from records of employment-related medical
examinations previously provided to the affected employee, currently
within the control of the employer.
(5) PLHCP's written medical opinion. (i) The employer shall obtain
a written medical opinion from the PLHCP, within 30 days for each
medical examination performed on each employee, which contains:
(A) The PLHCP's opinion as to whether the employee has any detected
medical condition(s) that would place the employee at increased risk of
material impairment to health from further exposure to chromium (VI);
(B) Any recommended limitations upon the employee's exposure to
chromium (VI) or upon the use of personal protective equipment such as
respirators;
(C) A statement that the PLHCP has explained to the employee the
results of the medical examination, including any medical conditions
related to chromium (VI) exposure that require further evaluation or
treatment, and any special provisions for use of protective clothing or
equipment.
(ii) The PLHCP shall not reveal to the employer specific findings
or diagnoses unrelated to occupational exposure to chromium (VI).
(iii) The employer shall provide a copy of the PLHCP's written
medical opinion to the examined employee within two weeks after
receiving it.
(j) Communication of chromium (VI) hazards to employees. (1)
General. In addition to the requirements of the Hazard Communication
Standard, 29 CFR 1910.1200, employers shall comply with the following
requirements.
(2) Employee information and training. (i) The employer shall
ensure that each employee can demonstrate knowledge of at least the
following:
(A) The contents of this section; and
(B) The purpose and a description of the medical surveillance
program required by paragraph (i) of this section.
(ii) The employer shall make a copy of this section readily
available without cost to all affected employees.
(k) Recordkeeping. (1) Air monitoring data. (i) The employer shall
maintain an accurate record of all air monitoring conducted to comply
with the requirements of this section.
(ii) This record shall include at least the following information:
(A) The date of measurement for each sample taken;
(B) The operation involving exposure to chromium (VI) that is being
monitored;
(C) Sampling and analytical methods used and evidence of their
accuracy;
(D) Number, duration, and the results of samples taken;
(E) Type of personal protective equipment, such as respirators
worn; and
(F) Name, social security number, and job classification of all
employees represented by the monitoring, indicating which employees
were actually monitored.
(iii) The employer shall ensure that exposure records are
maintained and made available in accordance with 29 CFR 1910.1020.
(2) Historical monitoring data. (i) Where the employer has relied
on historical monitoring data to determine exposure to chromium (VI),
the employer shall establish and maintain an accurate record of the
historical monitoring data relied upon.
(ii) The record shall include information that reflects the
following conditions:
(A) The data were collected using methods that meet the accuracy
requirements of paragraph (d)(5) of this section;
(B) The processes and work practices that were in use when the
historical monitoring data were obtained are essentially the same as
those to be used during the job for which exposure is being determined;
(C) The characteristics of the chromium (VI) containing material
being handled when the historical monitoring data were obtained are the
same as those on the job for which exposure is being determined;
(D) Environmental conditions prevailing when the historical
monitoring data were obtained are the same as those on the job for
which exposure is being determined; and
(E) Other data relevant to the operations, materials, processing,
or employee exposures covered by the exception.
(iii) The employer shall ensure that historical exposure records
are maintained and made available in accordance with 29 CFR 1910.1020.
(3) Objective data. (i) The employer shall maintain an accurate
record of all objective data relied upon to comply with the
requirements of this section.
(ii) This record shall include at least the following information:
(A) The chromium containing material in question;
(B) The source of the objective data;
(C) The testing protocol and results of testing, or analysis of the
material for the release of chromium (VI);
(D) A description of the process, operation, or activity and how
the data support the determination; and
(E) Other data relevant to the process, operation, activity,
material, or employee exposures.
(iii) The employer shall ensure that objective data are maintained
and made available in accordance with 29 CFR 1910.1020.
(4) Medical surveillance. (i) The employer shall establish and
maintain an accurate record for each employee covered by medical
surveillance under paragraph (i) of this section.
(ii) The record shall include the following information about the
employee:
(A) Name and social security number;
(B) A copy of the PLHCP's written opinions;
(C) A copy of the information provided to the PLHCP as required by
paragraph (i)(4) of this section.
(iii) The employer shall ensure that medical records are maintained
and made available in accordance with 29 CFR 1910.1020.
(l) Dates. (1) For employers with 20 or more employees, all
obligations of this section, except engineering controls required by
paragraph (e) of this section, commence November 27, 2006.
(2) For employers with 19 or fewer employees, all obligations of
this section, except engineering controls required by paragraph (e) of
this section, commence May 30, 2007.
(3) For all employers, engineering controls required by paragraph
(e) of this section shall be implemented no later than May 31, 2010.
[FR Doc. 06-1589 Filed 2-27-06; 8:45 am]
BILLING CODE 4510-26-P
















































































































